Medical treatment of obesity F Hosseinpanah M D

Medical treatment of obesity F. Hosseinpanah, M. D Obesity Research Center Research Institute for Endocrine sciences Shahid Beheshti University of Medical Sciences November 29, 2019 Tehran

Agenda • • Introduction Orlistat Lorcaserin Combination therapy Liraglutide Individualization of Pharmacotherapy Future Therapies Take home messages

Historical Roots • Dating back to the 1890 s use of sheep thyroid extract (to induce weight loss in euthyroid patients) resulted in reports of cardiac arrhythmias and death • In 1930 s, 2, 4‑dinitrophenol used for weight loss caused fatal hyperthermia, agranulocytosis and the development of cataracts. • In the 1950 s, amphetamine use became popular, especially in combination with other mixed regimens such as diuretics, laxatives and thyroid hormone, but resulted in reports of serious MI and sudden deaths
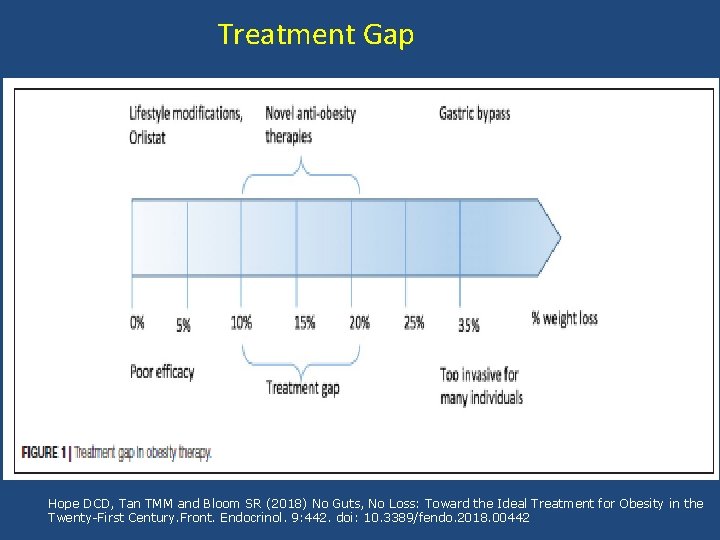
Treatment Gap Hope DCD, Tan TMM and Bloom SR (2018) No Guts, No Loss: Toward the Ideal Treatment for Obesity in the Twenty-First Century. Front. Endocrinol. 9: 442. doi: 10. 3389/fendo. 2018. 00442
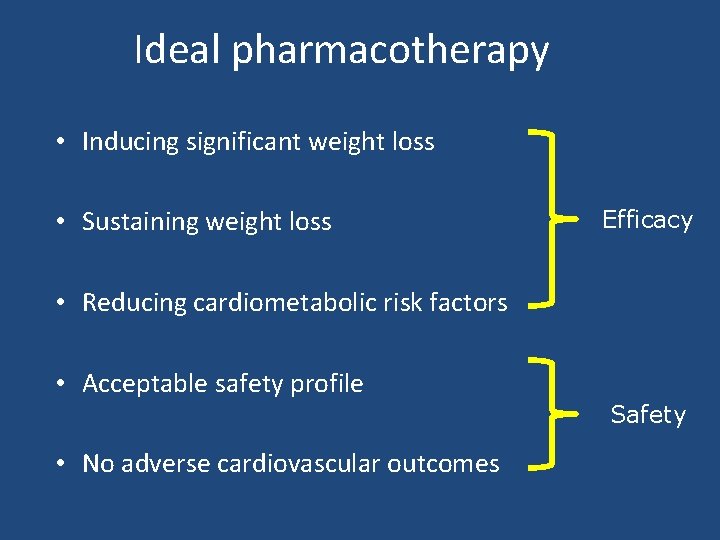
Ideal pharmacotherapy • Inducing significant weight loss • Sustaining weight loss Efficacy • Reducing cardiometabolic risk factors • Acceptable safety profile • No adverse cardiovascular outcomes Safety

Safety Issue • A careful assessment of the safety of antiobesity medications may be more important than for drugs used to treat other conditions , in which the drugs are less liable to misused • Obesity is a chronic condition. In light of this, longer-term data on the effectiveness and safety would be helpful

The story of removal and rejection • Fenfluramine and dexfenfluramine were removed in 1997 (valvular heart disease, pulmonary arterial hypertension) • Rimonabant was removed in 2007 (depression, anxiety. Suicidal ideation) • Sibutramin was removed in 2010 (cardiovascular events) • Lorcaserin was rejected by FDA in 2010, and approved again on June 2012
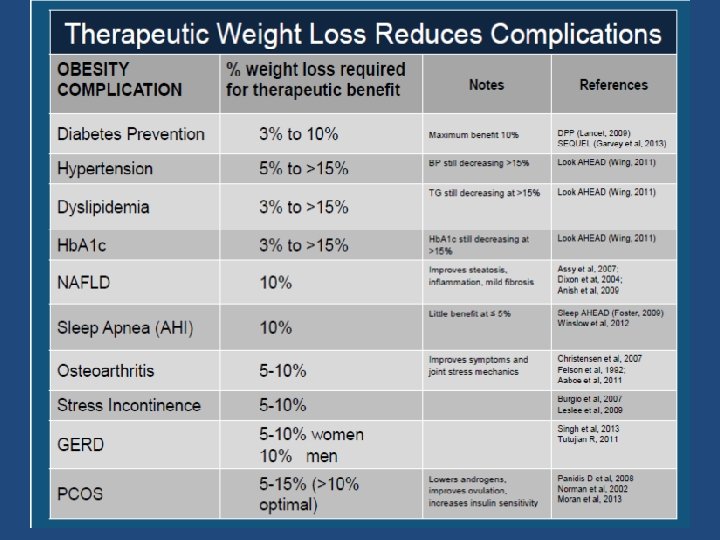

Obesity Guidance: Efficacy Criteria ¨ US FDA criteria at the end of year 1: ¨ Mean placebo-subtracted weight loss > 5% ¨ Proportion of subjects who lose > 5% of baseline body weight is greater in drug- vs. placebo-treated group ¨ EMEA criteria at end of year 1: ¨ mean placebo-subtracted weight loss > 10% ¨ Proportion of patients who lose > 10% of baseline body weight is greater in drug- vs. placebo-treated group
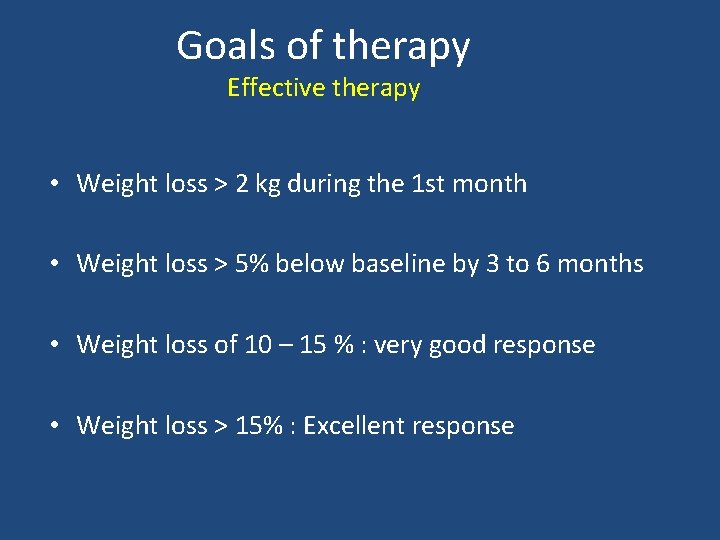
Goals of therapy Effective therapy • Weight loss > 2 kg during the 1 st month • Weight loss > 5% below baseline by 3 to 6 months • Weight loss of 10 – 15 % : very good response • Weight loss > 15% : Excellent response
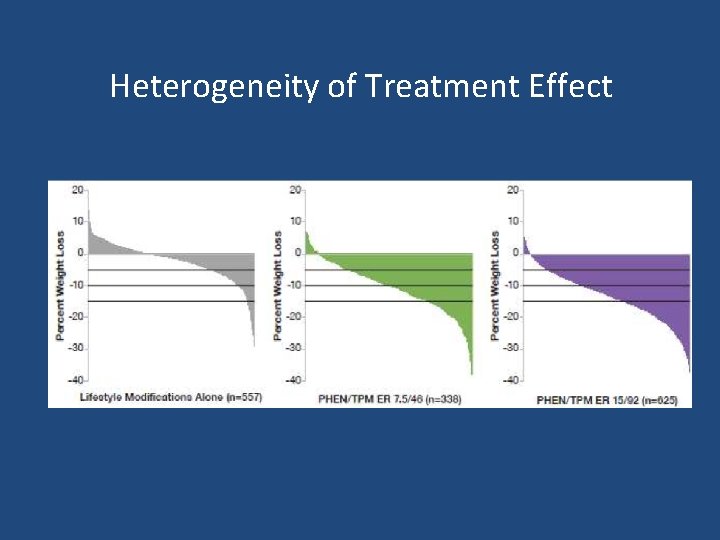
Heterogeneity of Treatment Effect

FDA “Off-Ramp”for obesity pharmacotherapy • If patient has not lost at least 5% (lorcaserin, naltrexone ER/bupropion ER, phentermine/topiramate ER) or 4% (liraglutide 3 mg) of baseline weight by week 12 on full maintenance dose, then discontinue

FDA approved drugs for long term use Orlistat 1997 Lorcaserin 2012 Phentermin plus extended-release topiramate 2012 Naltrexone hydrochloride and bupropion hydrochloride extended-release 2014 • Liraglutide 2014 • •

FDA approved drugs for short term use • Benzphetamine • Diethylpropion • Phendimetrazine • Phentermine 1959

Outcome Measures • • Weight Waist W/H ratio Lipid profile BP , HR Hb A 1 c , c peptide , Insulin Conversion of IGT to DM

Measures of weight loss • Mean weight loss ( Kg ) • Percentage weight loss • Percentage of individuals losing ≥ %5 or ≥ %10 of baseline weight • Maintenance of weight loss during study • Absolute weight loss (i. e. in excess of placebo )
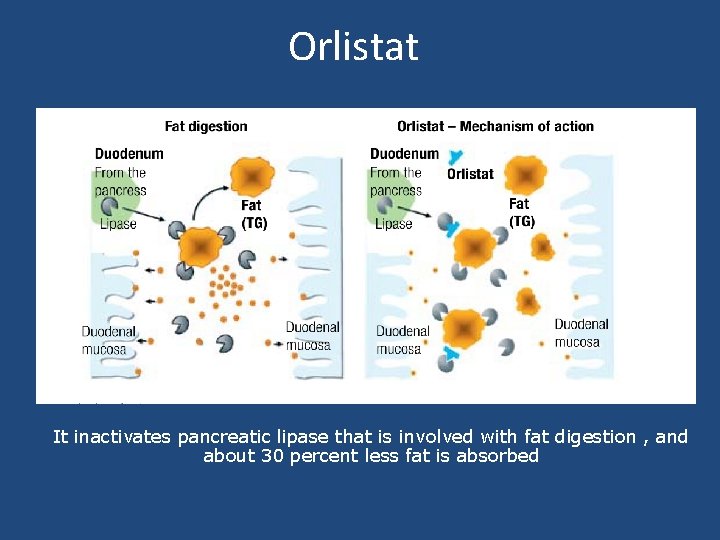
Orlistat It inactivates pancreatic lipase that is involved with fat digestion , and about 30 percent less fat is absorbed
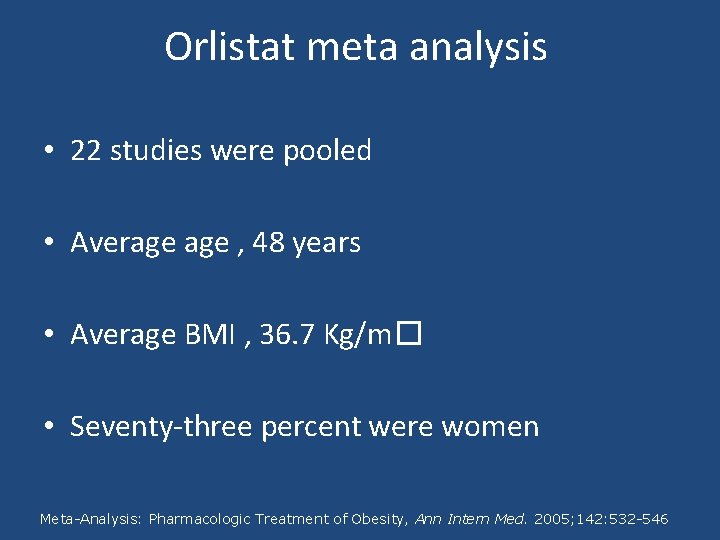
Orlistat meta analysis • 22 studies were pooled • Average , 48 years • Average BMI , 36. 7 Kg/m� • Seventy-three percent were women Meta-Analysis: Pharmacologic Treatment of Obesity, Ann Intern Med. 2005; 142: 532 -546
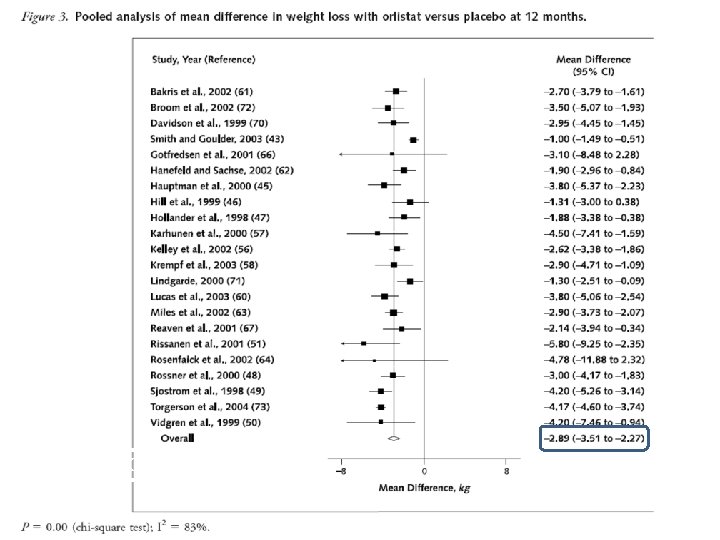
-2. 75 Kg (-3. 31 to -2. 20)

N = 3305 , BMI ≥ 30 F/U = 4 y Diet + placebo VS DIET + orlistat ( 120 mg TDS ) DIABETES CARE, VOLUME 27, NUMBER 1, JANUARY 2004
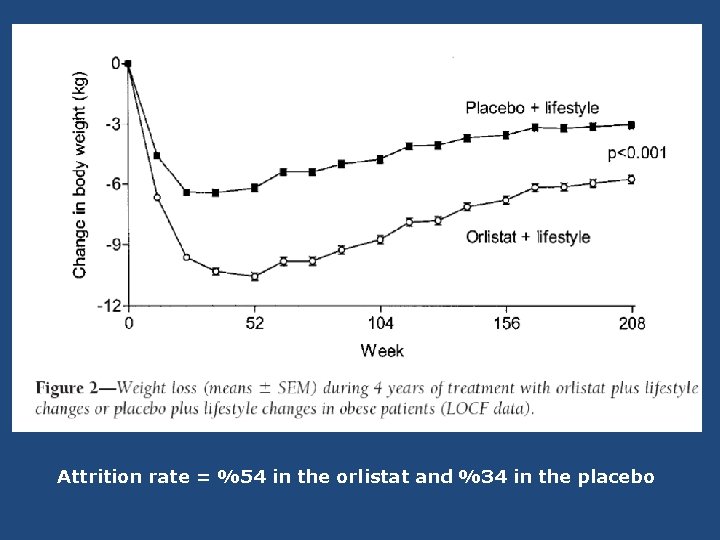
Attrition rate = %54 in the orlistat and %34 in the placebo



Side Effects • Gastrointestinal symptoms – – – Increased flatus Abdominal discomfort Oily spotting Fecal Incontinence More severe if intake of fat is more than 100 grams. • Nutritional concerns – Fat-soluble vitamins may be malabsorbed. – Daily multi-vitamin is recommended



Lorcaserin • Lorcaserin has specific selectivity towards the 5‑HT 2 c receptor • Lorcaserin alleviates risk associated with prior agents of this class, such as fenfluramine, which was found to have affinity for both 5‑HT 2 A (causing hallucinations) and 5‑HT 2 B receptors (causing cardiac valve insufficiency and pulmonary hypertension ) Pharmacol. Exp. Ther. 325, 577– 587(2008)

• • • Double-blind randomized clinical trial Sample size : 3182 (obese or overweight) Intervention : lorcaserin (10 mg) twice daily Length of F/U : 2 years Primary outcomes : weight loss at 1 year and maintenance of weight loss at 2 years N Engl J Med 2010; 363: 245 -56

High attrition rate • The rates of completion of year 1 of the study were 55. 4% in the lorcaserin group and 45. 1% in the placebo group • The overall rate of completion of year 2 of the study was 72. 6% of patients who completed year 1
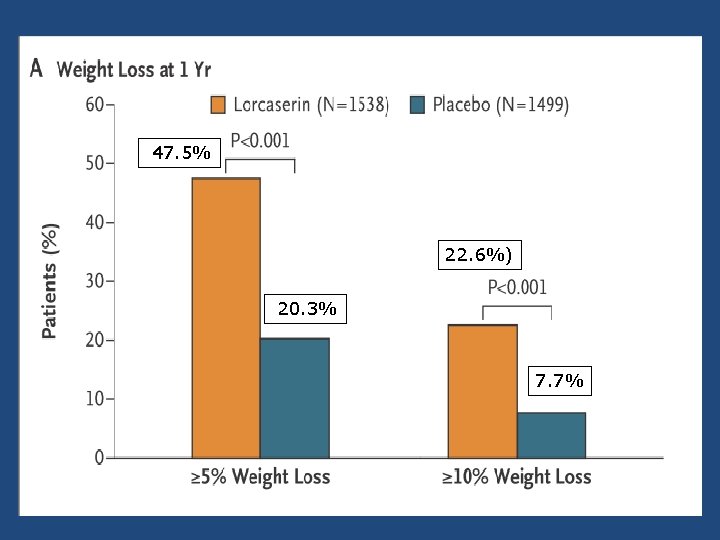
47. 5% 22. 6%) 20. 3% 7. 7%
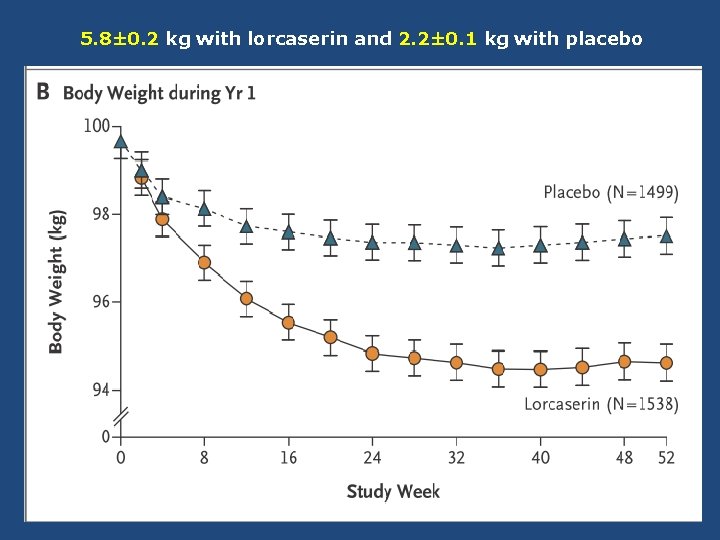
5. 8± 0. 2 kg with lorcaserin and 2. 2± 0. 1 kg with placebo
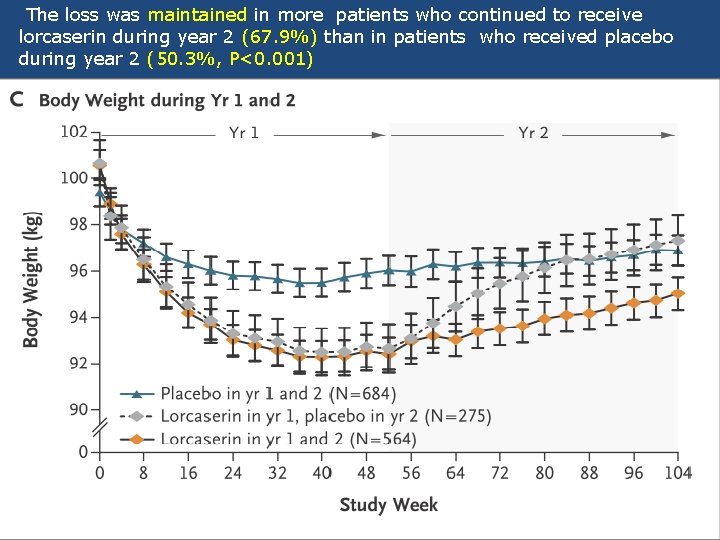
The loss was maintained in more patients who continued to receive lorcaserin during year 2 (67. 9%) than in patients who received placebo during year 2 (50. 3%, P<0. 001)

Adverse events • Rate of cardiac valvulopathy was not increased with the use of lorcaserin • Among the most frequent adverse events re- ported with lorcaserin were headache, dizziness, and nausea • The rates of serious adverse events in the two groups were similar


METHODS • 12, 000 overweight or obese patients with ASCVD or multiple cardiovascular risk factors • lorcaserin (10 mg twice daily) or placebo • Lemght of F/U: 3. 3 years • The primary safety outcome of MACE (a composite of CV death, MI, or stroke) was assessed at an interim analysis to exclude a noninferiority boundary of 1. 4 • If noninferiority was met , primary cardiovascular efficacy outcome (a composite of MACE, heart failure, hospitalization for unstable angina, or coronary revascularization (extended MACE) was assessed for superiority at the end of the trial
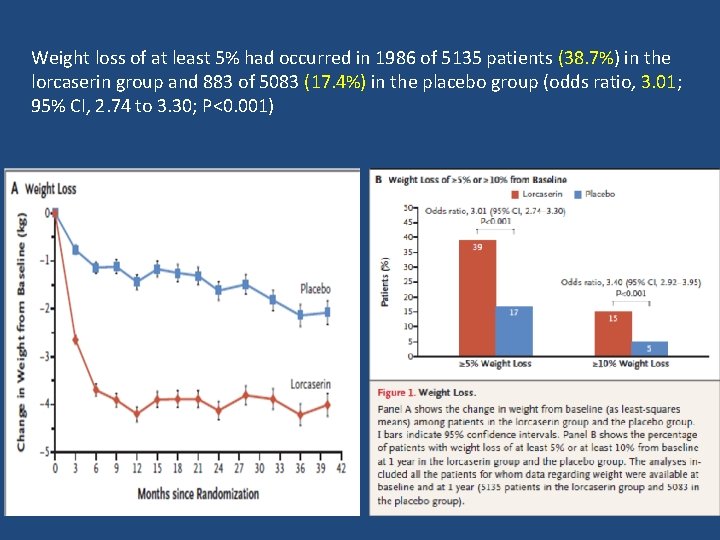
Weight loss of at least 5% had occurred in 1986 of 5135 patients (38. 7%) in the lorcaserin group and 883 of 5083 (17. 4%) in the placebo group (odds ratio, 3. 01; 95% CI, 2. 74 to 3. 30; P<0. 001)
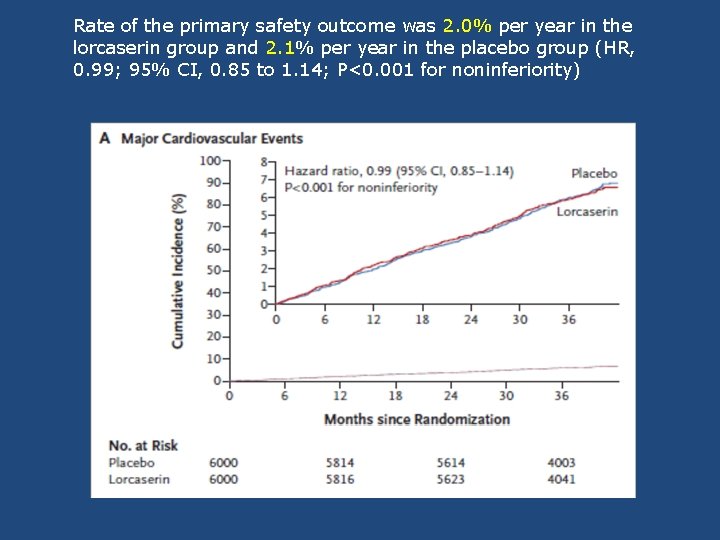
Rate of the primary safety outcome was 2. 0% per year in the lorcaserin group and 2. 1% per year in the placebo group (HR, 0. 99; 95% CI, 0. 85 to 1. 14; P<0. 001 for noninferiority)
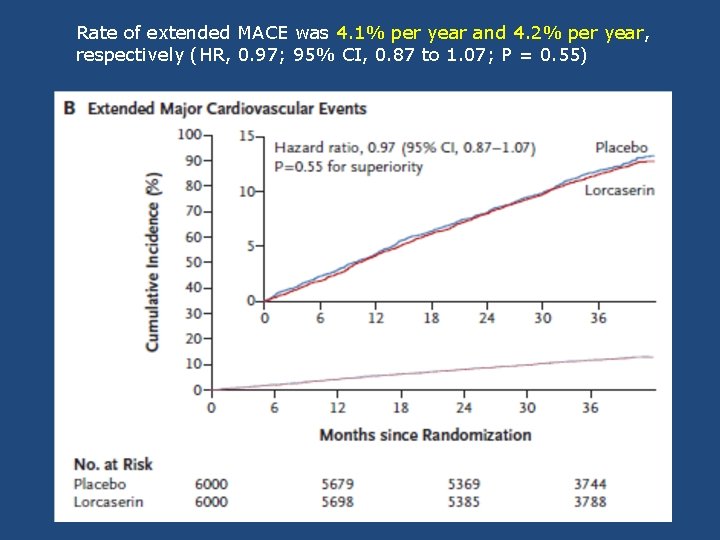
Rate of extended MACE was 4. 1% per year and 4. 2% per year, respectively (HR, 0. 97; 95% CI, 0. 87 to 1. 07; P = 0. 55)


Key message • In a high-risk population of overweight or obese patients, lorcaserin facilitated sustained weight loss without a higher rate of major cardiovascular events than that with placebo.


Methods • Subjects: 9804 overweight or obese subjects, 55 years of age or older, with preexisting cardiovascular disease, type 2 diabetes mellitus. , underwent random assignment to sibutramine (4906 subjects) or placebo (4898 subjects) • F/U: 3. 4 years • Primary end point: nonfatal myocardial infarction, non-fatal stroke, resuscitation after cardiac arrest, or cardiovascular death
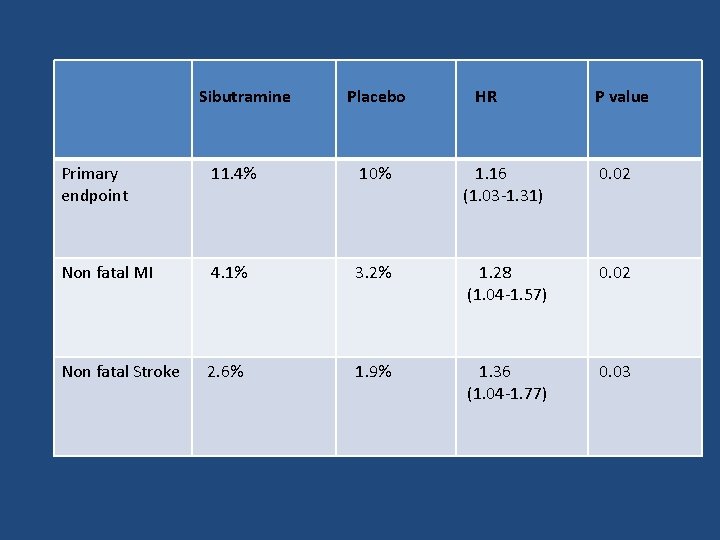
Sibutramine Placebo HR P value Primary endpoint 11. 4% 10% 1. 16 (1. 03 -1. 31) 0. 02 Non fatal MI 4. 1% 3. 2% 1. 28 (1. 04 -1. 57) 0. 02 Non fatal Stroke 2. 6% 1. 9% 1. 36 (1. 04 -1. 77) 0. 03




Key message • Subjects with preexisting cardiovascular conditions who were receiving long-term sibutramine treatment had an increased risk of nonfatal myocardial infarction and nonfatal stroke but not of cardiovascular death or death from any cause. Sibutramin was removed in 2010 (cardiovascular events)

Combinations of drugs that produce weight loss • • Phentermine and Fenfluramine Phentremine with Topiramate Phentremine with Zonisamide Naltrexone with Bupropion

Phentermine/Topiramate ER • Phentermine is a sympathomimetic amine • Release insignificant quantities of dopamine compared with amphetamines and thus has a comparatively lower abuse potential • The exact anorexigenic mechanism of topiramate is not well understood Antel, J. & Hebebrand, J. Weight-reducing side effects of the antiepileptic agents topiramate and zonisamide. Handb. Exp. Pharmacol. 209, 433– 466 (2012)

Phentermine/Topiramate ER • Inhibition of voltage-dependent sodium channels • Inhibition of glutamate receptors and carbonic anhydrase • Potentiation of γ-aminobutyrate activity Antel, J. & Hebebrand, J. Weight-reducing side effects of the antiepileptic agents topiramate and zonisamide. Handb. Exp. Pharmacol. 209, 433– 466 (2012)


Combinations of drugs that produce weight loss Effects of low-dose, controlled-release, Phentermine plus Topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Gadde, K. M. et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet 377, 1341– 1352 (2011).

CONQUER Trial • Overweight or obese adults (aged 18– 70 years), with a BMI of 27 – 45 kg/m 2 and two or more comorbidities (hypertension, dyslipidemia, diabetes or prediabetes, or abdominal obesity) to placebo, once-daily phentermine 7. 5 mg plus topiramate 46. 0 mg, or once-daily phentermine 15. 0 mg plus topiramate 92. 0 mg • In total, 943 (38%) patients discontinued the study drugs— 43% in the placebo group, 31% in the group assigned to phentermine 7. 5 mg plus topiramate 46. 0 mg, and 36% in the group assigned to phentermine 15. 0 mg plus topiramate 92. 0 mg
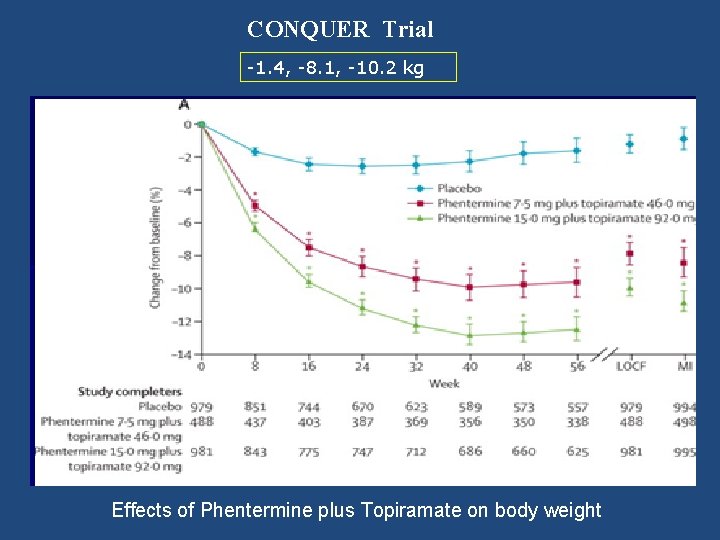
CONQUER Trial -1. 4, -8. 1, -10. 2 kg Effects of Phentermine plus Topiramate on body weight
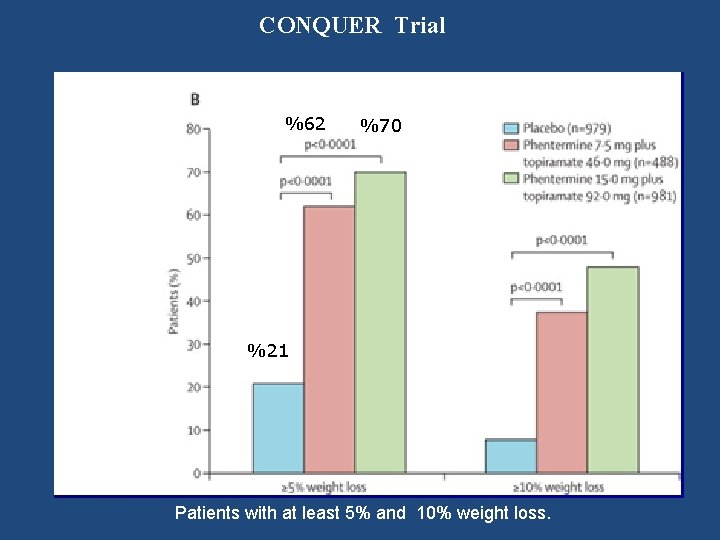
CONQUER Trial %62 %70 %21 Patients with at least 5% and 10% weight loss.

Adverse Effects • • • Dry mouth Paresthesia Constipation Insomnia Dizziness Dysgeusia Depression-related adverse events Anxiety-related adverse Cognitive (eg, disturbance in attention)


Safety Concerns expressed by the FDA included: 1. Teratogenicity (both components of combination drug) 2. Cardiovascular concerns (specific to phentermine component) 3. Cognitive, psychiatric and metabolic acidosis (the last three being specific to topiramate component) Roberts, M. D. Clinical briefing document: Endocrinologic and Metabolic Drugs Advisory Committee Meeting (February 22, 2012). New Drug Application 22580: VI‑ 0521 Qnexa (phentermine/ topiramate). Sponsor: Vivus. FDA

• Aim : To determine the extent to which the rates of MACE in patients using PHEN and TPM (including fixed dose) differed from the MACE rates during unexposed periods. • Design: Retrospective cohort study J Clin Endocrinol Metab 104: 513– 522, 2019
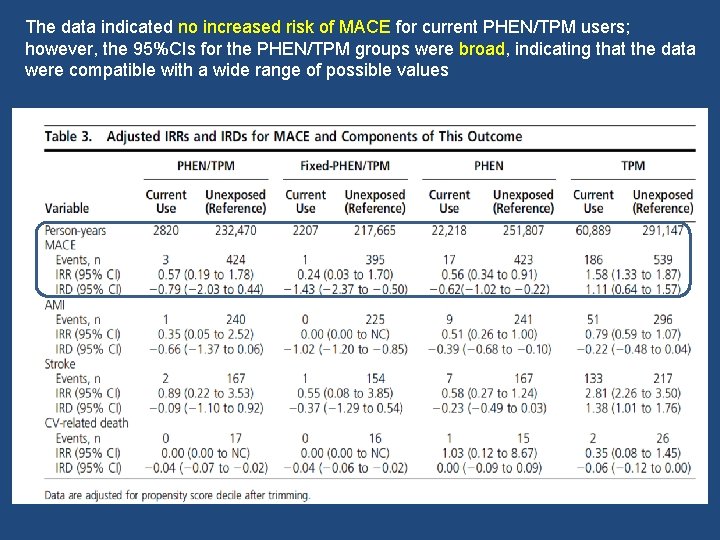
The data indicated no increased risk of MACE for current PHEN/TPM users; however, the 95%CIs for the PHEN/TPM groups were broad, indicating that the data were compatible with a wide range of possible values

Naltrexone SR/bupropion SR • Bupropion reduces food intake by acting on adrenergic and dopaminergic receptors in the hypothalamus • Bupropion stimulates the pro-opiomelanocortin neurons in the hypothalamus to produce proopiomelanocortin, which is further processed to produce both α-melanocyte stimulating hormone (which reduces food intake) and β-endorphin (which stimulates feeding) • Naltrexone blocks this effect of β-endorphin , thus allowing the inhibitory effects of a-melanocyte stimulating hormone to reduce food intake by acting on the melanocortin-4 receptor system

Naltrexone Hydrochloride and Bupropion Hydrochloride extended-release • The effectiveness of NB was evaluated in multiple clinical trials that included approximately 4, 500 obese and overweight patients with and without significant weightrelated conditions treated for one year • Frothy two percent of patients treated with NB lost at least 5 percent of their body weight compared with 17 percent of patients treated with placebo Greenway, F. L. et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR‑I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 376, 595– 605 (2010).


Safety • The combination, NB, did not increase rates of depression and suicidal ideation more than placebo in the clinical trials • Transient nausea, most frequently occurring during the 3‑week dose-escalation period, was the most common adverse effect in approximately one-third of patients. • Other more common adverse events reported with an incidence of at least 2% among patients treated with NB compared with placebo included vomiting , constipation, dry mouth, headaches , dizziness
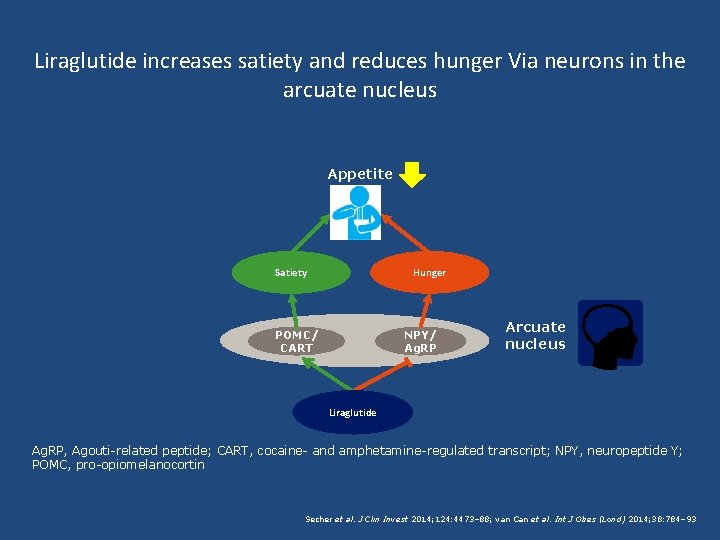
Liraglutide increases satiety and reduces hunger Via neurons in the arcuate nucleus Appetite Hunger Satiety POMC/ CART NPY/ Ag. RP Arcuate nucleus Liraglutide Ag. RP, Agouti-related peptide; CART, cocaine- and amphetamine-regulated transcript; NPY, neuropeptide Y; POMC, pro-opiomelanocortin Secher et al. J Clin Invest 2014; 124: 4473– 88; van Can et al. Int J Obes (Lond) 2014; 38: 784– 93

3731 participants Without • ≥ 18 years prediabetes • Stable BW • BMI ≥ 30 kg/m 2 or With prediabetes 1 ≥ 27 kg/m 2 + comorbidities Liraglutide 3. 0 mg (n=959) Trial information • Placebo (n=487) • Liraglutide 3. 0 mg (n=1528) • Placebo (n=757) • Lifestyle intervention: -500 kcal/day diet + 150 min/week physical activity Dose escalation 0– 4 weeks Treatment duration 52 weeks Randomisation (2: 1) Trial objective • Efficacy and safety of liraglutide 3. 0 mg, as adjunct to D&E, in participants with obesity or overweight plus comorbidities, without diabetes 12 weeks Re-randomisation (1: 1) 2 -week FU June 2011 to March 2013 Randomised controlled double-blind study 191 sites in 27 countries Duration: 56 weeks (with prediabetes), 68 weeks (without prediabetes) EOT Key endpoints • • Three co-primary: BW change, 5% or 10% BW loss Secondary: Changes from baseline in BMI, WC, glycaemic control variables, cardiometabolic risk factors, and HRQo. L 1. ADA. Diabetes Care 2010; 33(Suppl. 1): S 11– 61 BW, body weight; D&E, diet and exercise; EOT, end of treatment; FU, follow-up; HRQo. L, health-related quality of life; WC, waist circumference Pi-Sunyer et al. N Engl J Med 2015; 373: 11– 22

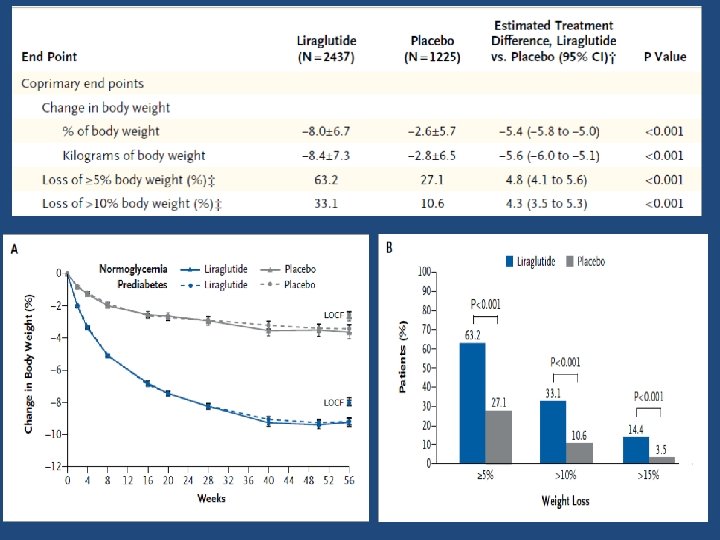
Results • A total of 1789 patients(71. 9%) in the liraglutide group, as compared with 801 patients (64. 4%) in the placebo group, completed 56 weeks of treatment • A larger percentage of patients in the liraglutide group than in the placebo group withdrew from the trial owing to adverse events (9. 9% [246 of 2487 patients] vs. 3. 8% [47 of 1244])
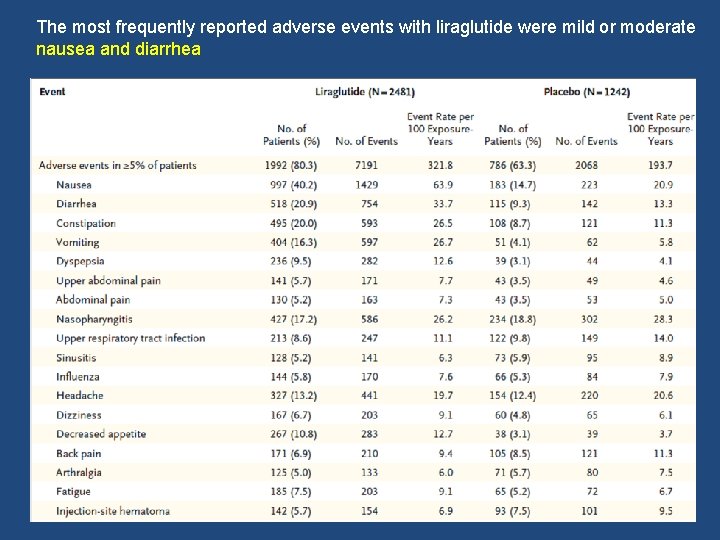
The most frequently reported adverse events with liraglutide were mild or moderate nausea and diarrhea
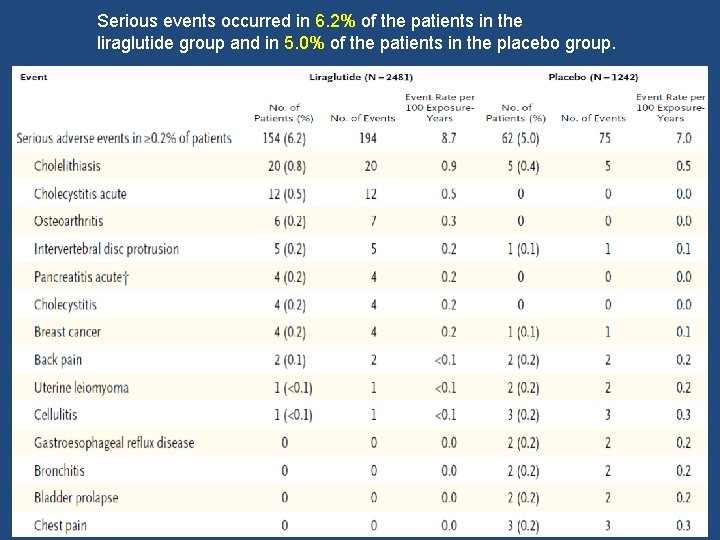
Serious events occurred in 6. 2% of the patients in the liraglutide group and in 5. 0% of the patients in the placebo group.
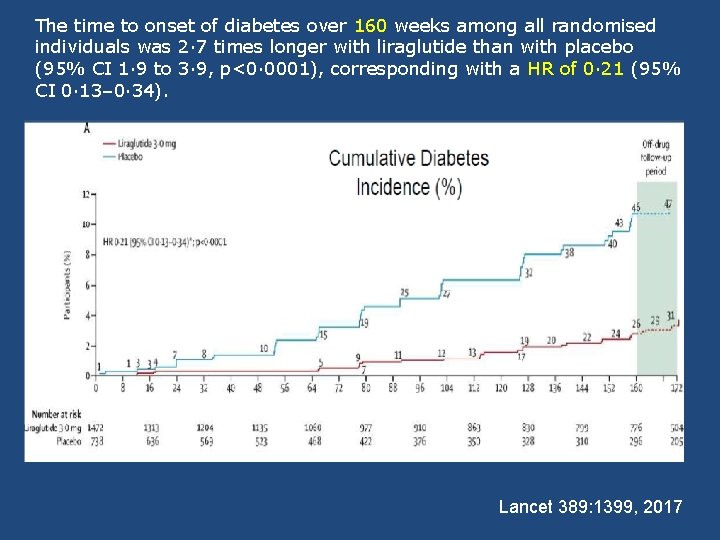
The time to onset of diabetes over 160 weeks among all randomised individuals was 2· 7 times longer with liraglutide than with placebo (95% CI 1· 9 to 3· 9, p<0· 0001), corresponding with a HR of 0· 21 (95% CI 0· 13– 0· 34). Lancet 389: 1399, 2017
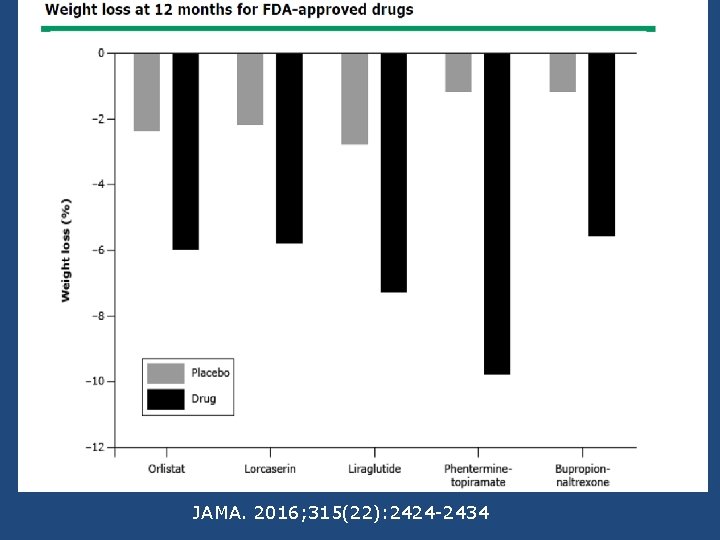
JAMA. 2016; 315(22): 2424 -2434
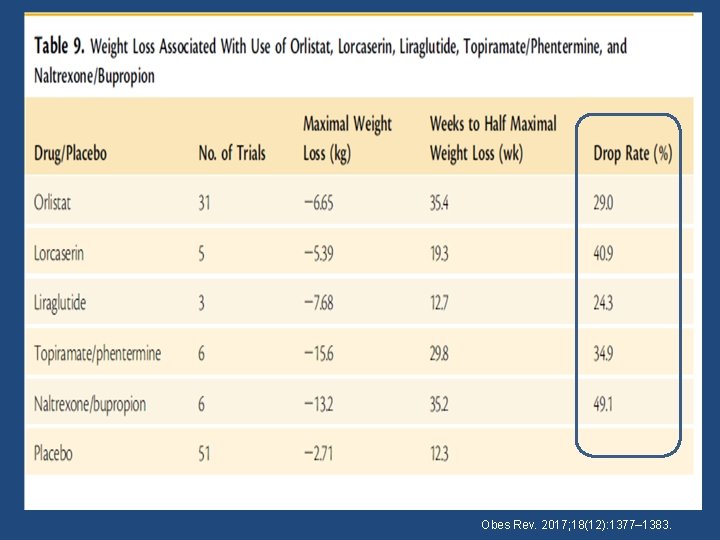
Obes Rev. 2017; 18(12): 1377– 1383.
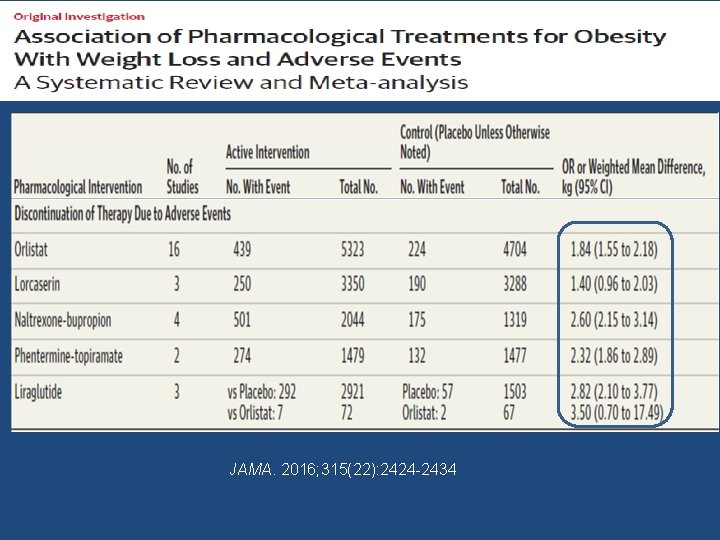
JAMA. 2016; 315(22): 2424 -2434

• Individualization of Pharmacotherapy in the Treatment of Obesity ENDOCRINE PRACTICE Vol 22 (Suppl 3) July 2016
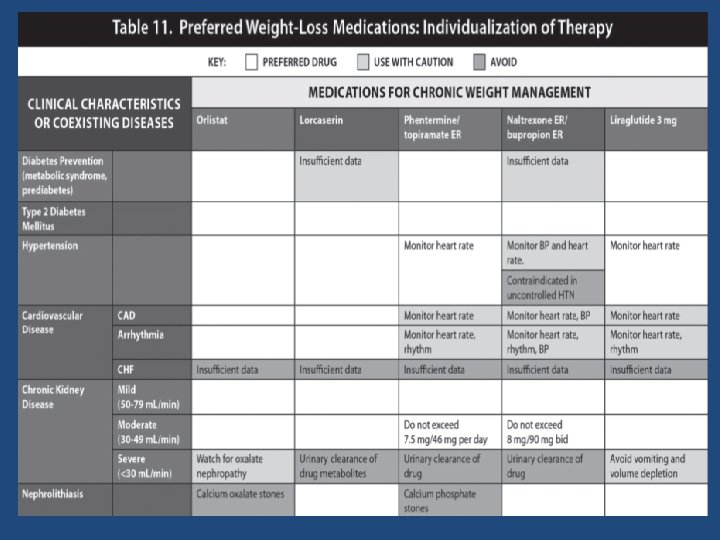
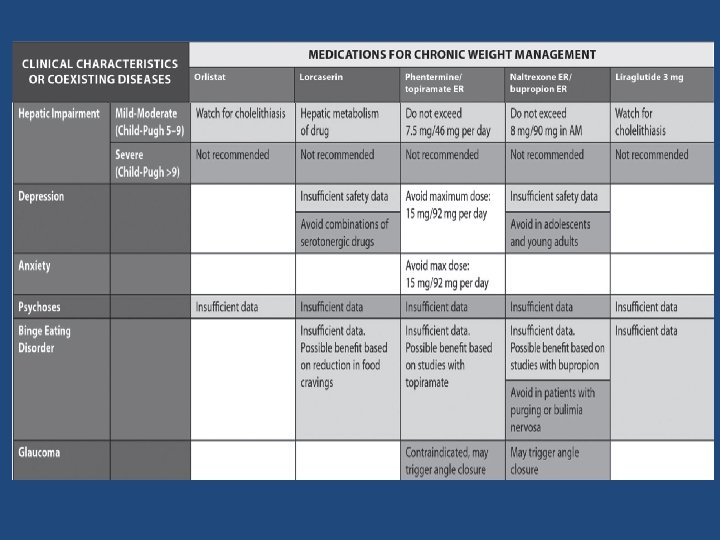


Candidates for drug therapy • In order to promote long-term weight maintenance, we suggest the use of approved weight loss medication (over no pharmacological therapy) to ameliorate comorbidities and amplify adherence to behavior changes, which may improve physical functioning and allow for greater physical activity in individuals with a BMI 30 kg/m 2 or in individuals with a BMI of 27 kg/m 2 and at least one associated comorbid medical condition such as hypertension, dyslipidemia, type 2 diabetes (T 2 DM), and obstructive sleep apnea. J Clin Endocrinol Metab 100: 342– 362, 2015

Future Therapies • Semaglutide • The PP-fold anorectic gut hormones : peptide YY, pancreatic polypeptide • Dual agonism of GLP-1 and glucagon • Oxyntomodulin • GIP/GLP-1 co-agonist • Triple agonist combinations GLP- 1, GIP and glucagon • Triple agonism with GLP-1/OXM/PYY—the pathway to the “medical bypass”? • Selective agonists for the ADRβ 3 (Mirabegron and solabegron)


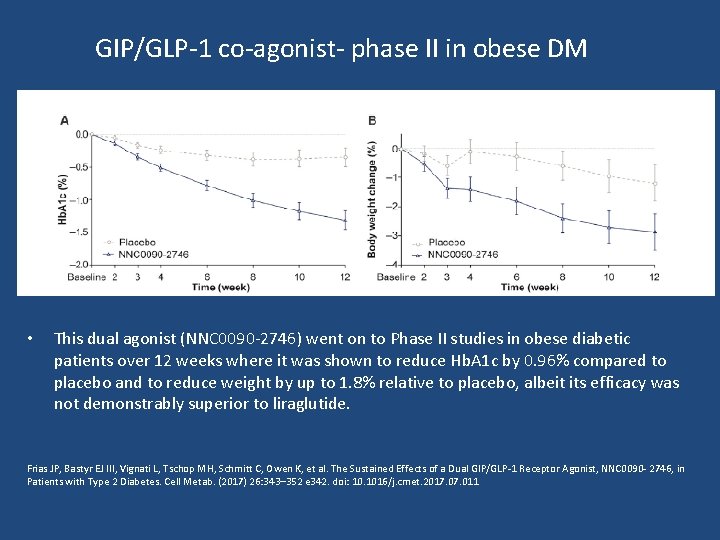
GIP/GLP-1 co-agonist- phase II in obese DM • This dual agonist (NNC 0090 -2746) went on to Phase II studies in obese diabetic patients over 12 weeks where it was shown to reduce Hb. A 1 c by 0. 96% compared to placebo and to reduce weight by up to 1. 8% relative to placebo, albeit its efficacy was not demonstrably superior to liraglutide. Frias JP, Bastyr EJ III, Vignati L, Tschop MH, Schmitt C, Owen K, et al. The Sustained Effects of a Dual GIP/GLP-1 Receptor Agonist, NNC 0090 - 2746, in Patients with Type 2 Diabetes. Cell Metab. (2017) 26: 343– 352 e 342. doi: 10. 1016/j. cmet. 2017. 011


Take home messages • Weight loss attributable to these medications are modest but still may be clinically significant • The maximal duration of published treatment results is four years for orlistat • When drug therapy is discontinued , weight is regained • Near maximal weight loss is achieved by six months in most trials • individual responses vary widely • The efficacy for weight loss of phentermine extended release topiramate appears to be greater than either orlistat or lorcaserin, but with more side effects

Take home messages • Pharmacologic therapy can be offered to obese patients who have failed to achieve their weight loss goals through diet and exercise alone. • However, there needs to be a doctor–patient discussion of the drugs’ side effects, the lack of long-term safety data, and the temporary nature of the weight loss achieved with medications before initiating therapy. • “Individualization” is the most important element of decision making for medical treatment of obesity

Thanks for your kind attention
- Slides: 87