MEDICAL GRANDROUNDS OCTOBER 22 2009 LEDESMA HALL LEONID

MEDICAL GRANDROUNDS OCTOBER 22, 2009 LEDESMA HALL LEONID ZAMORA MD

OBJECTIVES To present a case of a young female who developed complicated pneumonia secondary to Influenza A(H 1 N 1) Virus Infection To discuss the latest updates on the Virus.

General Data AMG 23 years old Female Single Filipino a student from DLSU Lives in Paranaque Known Diabetic

Chief Complaint DYSPNEA
HISTORY OF PRESENT ILLNESS 5 DAYS PTA non-productive cough body malaise high grade fever (Tmax 40. 5 C) Consult at RITM throat swab done voluntary isolation at home

HISTORY OF PRESENT ILLNESS 2 DAYS PTA persistence of the above symptoms self-medicated with Cefuroxime 500 mg 2 x a day No consult

HISTORY OF PRESENT ILLNESS Few hours PTA increasing frequency and severity of cough Dyspnea No chest pains, orthopnea, paroxysmal nocturnal dyspnea Throat swab done in RITM POSITIVE ER consult

Review of Systems No headache No loss of consciousness No blurring of vision No nausea, vomiting No dysuria, hematuria No diarrhea/constipation No bleeding No polyuria, polydipsia, polyphagia

Past Medical History Diabetes since 2007 – on Metformin 500 mg TID and Rosiglitazone 4 mg OD Hypertensive since 2008 - no maintenance medications (HBP 150/90 – UBP 130/80) (+) PCOS x 5 months on Norethisterone (Primolut) and Medroxyprogesterone acetate (Provera) No previous hospitalizations or surgeries No known allergies.

Family History Diabetes and Hypertension both parents (+) Asthma maternal side (cousins) No Asthma No Cancer

Personal and Social History Non smoker Occasional alcoholic beverage drinker No recent Travel outside Metro Manila Student of DLSU – was recently closed due to reported cases of positive Inluenza A(H 1 N 1)

Physical Examination Conscious, coherent, in respiratory distress BP 110/70 HR 115 reg RR 30 Temp 39 C Ht 162. 5 cm Wt 109 kg BMI 41. 3 Warm moist skin. No active dermatosis. Pink palpebral conjunctivae, anicteric sclerae, moist buccal mucosa, nonhyperemic posterior pharyngeal wall, tonsils not enlarged, (+) alar flaring. Supple neck, no cervical lymphadenopathies, (+) neck vein distention, no carotid bruit.

Physical Examination Symmetrical chest expansion, no lagging, (+) tight air entry, (+) wheezes, bilateral. Adynamic precordium, AB at 5 th Left intercostal space, Mid clavicular line, tachycardic, regular rhythm, no murmurs, no gallop rhythm. Flabby abdomen, normoactive bowel sounds, nontender, no masses, no organomegaly. No abdominal bruit. Full and equal pulses. No cyanosis. No edema.

Salient Features 23 years old, Female in respiratory distress. a student from DLSU BP 110/70 HR 115 reg Diabetic RR 30 Temp 39 C (+) alar flaring (+) neck vein distention (+) tight air entry (+) wheezes, bilateral. Obese Dyspnea Increasing severity of cough Fever POSITIVE for A(H 1 N 1)

ADMITTING IMPRESSION • SEVERE SEPSIS SECONDARY TO • • COMMUNITY ACQUIRED PNEUMONIA SECONDARY TO INFLUENZA A(H 1 N 1) DIABETES MELLITUS, TYPE 2, NON-INSULIN REQUIRING OBESE CLASS III HYPERTENSION STAGE 1 POLYCYSTIC OVARIAN SYNDROME

COURSE IN THE WARD At the ER In respiratory distress VS BP 110/70 HR 115 reg RR 30 Temp 39 C O 2 sat 82% at room air NPO CBC, CXR, ECG, ABGs Hooked to Pulse Oximeter MVM 0. 5 PNSS 1 L x 100 ml/hr Rx: Fenoterol + Ipratropium (Berodual) nebulization q 4 h, Oseltamivir 75 mg tab q 12 h, Budesonide 500 mcg BID nebulization, Hydrocortisone 100 mg q 8 h Paracetamol 300 mg q 4 h
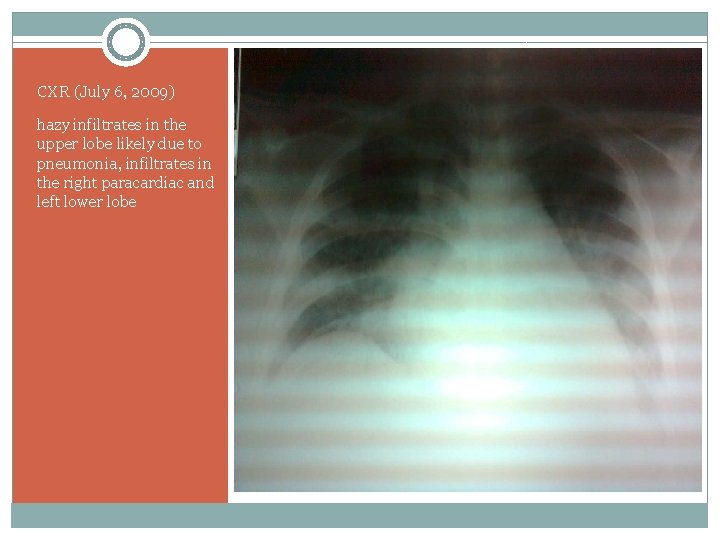
CXR (July 6, 2009) hazy infiltrates in the upper lobe likely due to pneumonia, infiltrates in the right paracardiac and left lower lobe
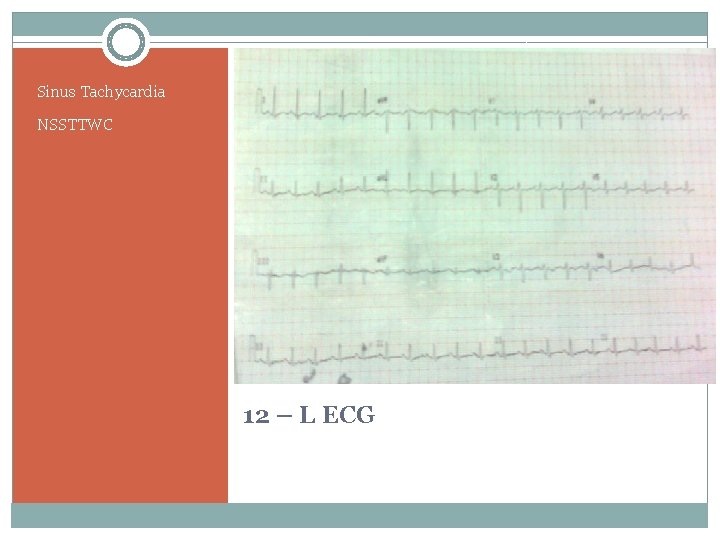
Sinus Tachycardia NSSTTWC 12 – L ECG
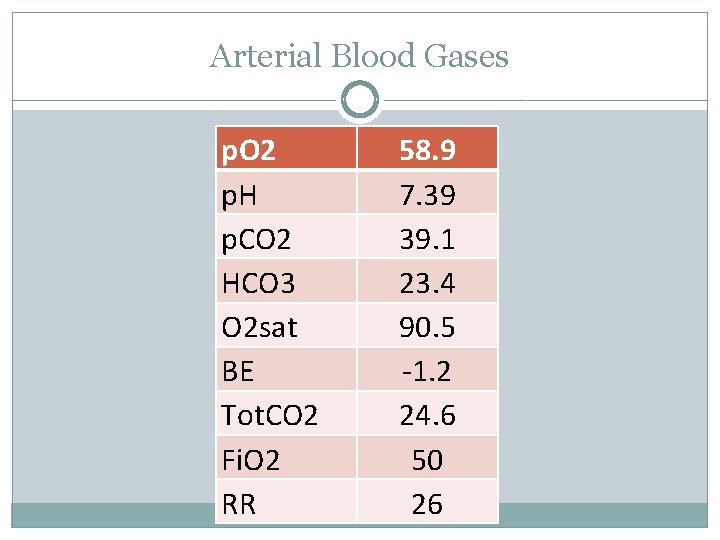
Arterial Blood Gases p. O 2 p. H p. CO 2 HCO 3 O 2 sat BE Tot. CO 2 Fi. O 2 RR 58. 9 7. 39 39. 1 23. 4 90. 5 -1. 2 24. 6 50 26
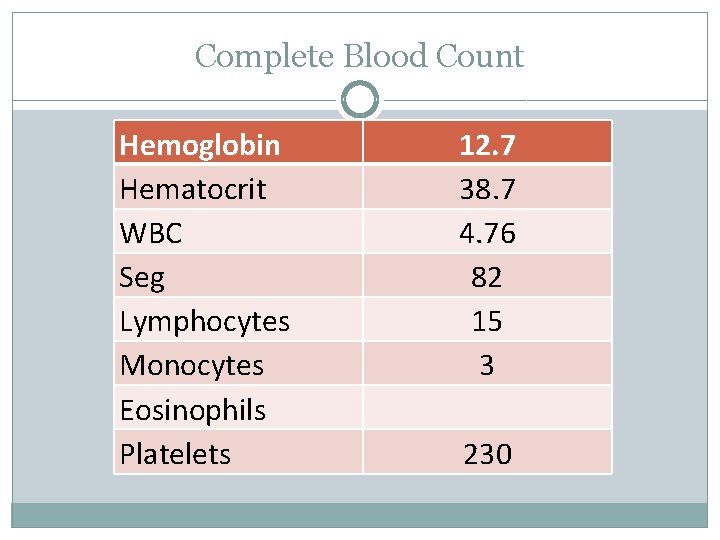
Complete Blood Count Hemoglobin Hematocrit WBC Seg Lymphocytes Monocytes Eosinophils Platelets 12. 7 38. 7 4. 76 82 15 3 230
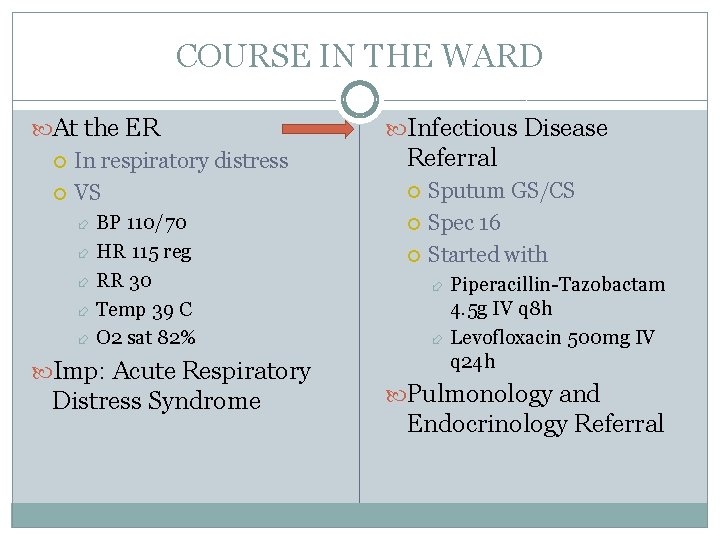
COURSE IN THE WARD At the ER In respiratory distress VS BP 110/70 HR 115 reg RR 30 Temp 39 C O 2 sat 82% Imp: Acute Respiratory Distress Syndrome Infectious Disease Referral Sputum GS/CS Spec 16 Started with Piperacillin-Tazobactam 4. 5 g IV q 8 h Levofloxacin 500 mg IV q 24 h Pulmonology and Endocrinology Referral

ACUTE RESPIRATORY DISTRESS SYNDROME Acute onset of Respiratory Failure Diffuse Bilateral infiltrates on Chest radiograph Absence of left atrial hypertension (PCWP < 18 mm. Hg or no clinical evidence of increased left atrial pressure) Hypoxemia, Pa. O 2/Fi. O 2 < 200
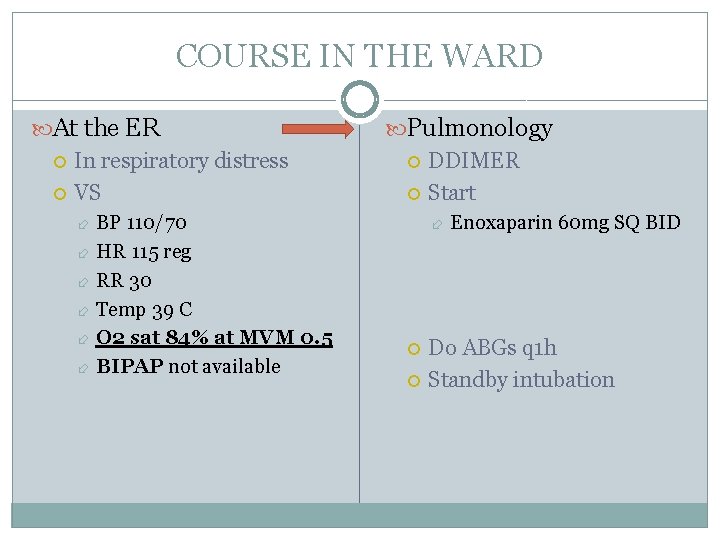
COURSE IN THE WARD At the ER In respiratory distress VS BP 110/70 HR 115 reg RR 30 Temp 39 C O 2 sat 84% at MVM 0. 5 BIPAP not available Pulmonology DDIMER Start Enoxaparin 60 mg SQ BID Do ABGs q 1 h Standby intubation
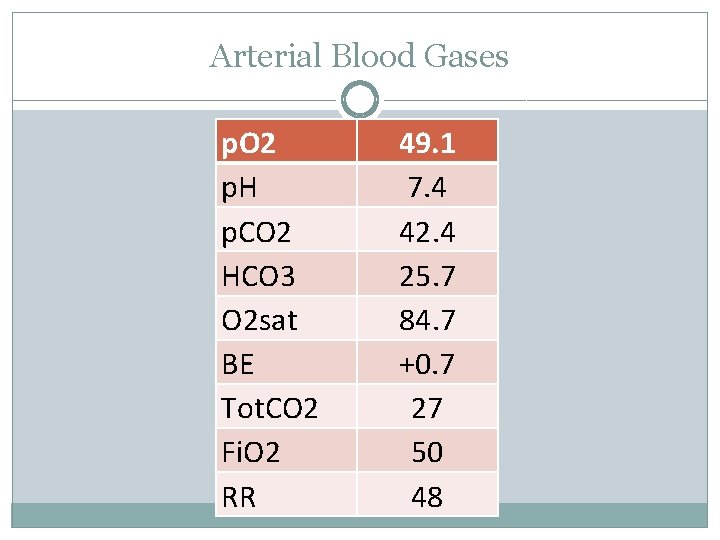
Arterial Blood Gases p. O 2 p. H p. CO 2 HCO 3 O 2 sat BE Tot. CO 2 Fi. O 2 RR 49. 1 7. 4 42. 4 25. 7 84. 7 +0. 7 27 50 48
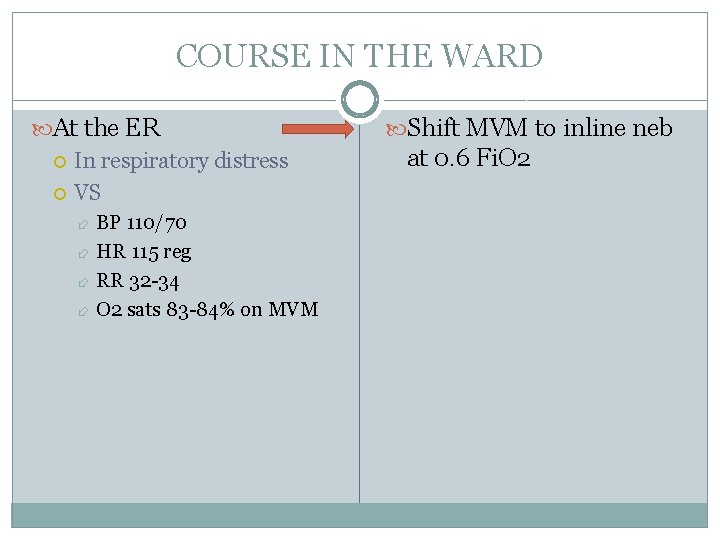
COURSE IN THE WARD At the ER In respiratory distress VS BP 110/70 HR 115 reg RR 32 -34 O 2 sats 83 -84% on MVM Shift MVM to inline neb at 0. 6 Fi. O 2
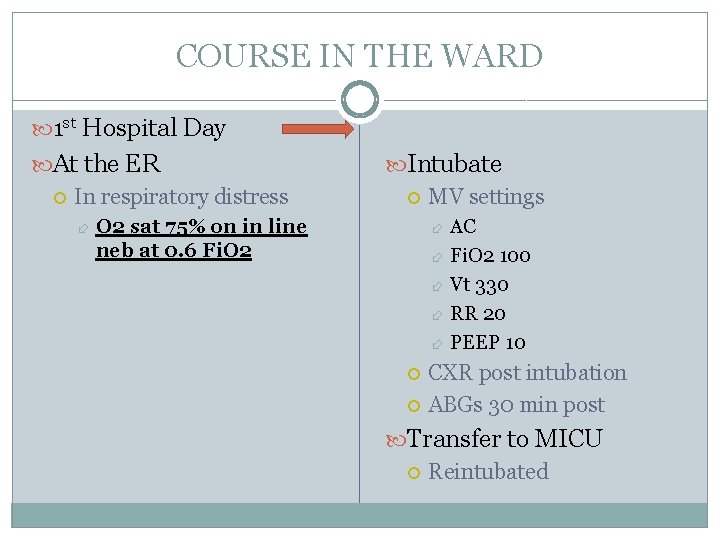
COURSE IN THE WARD 1 st Hospital Day At the ER In respiratory distress Intubate MV settings O 2 sat 75% on in line neb at 0. 6 Fi. O 2 AC Fi. O 2 100 Vt 330 RR 20 PEEP 10 CXR post intubation ABGs 30 min post Transfer to MICU Reintubated
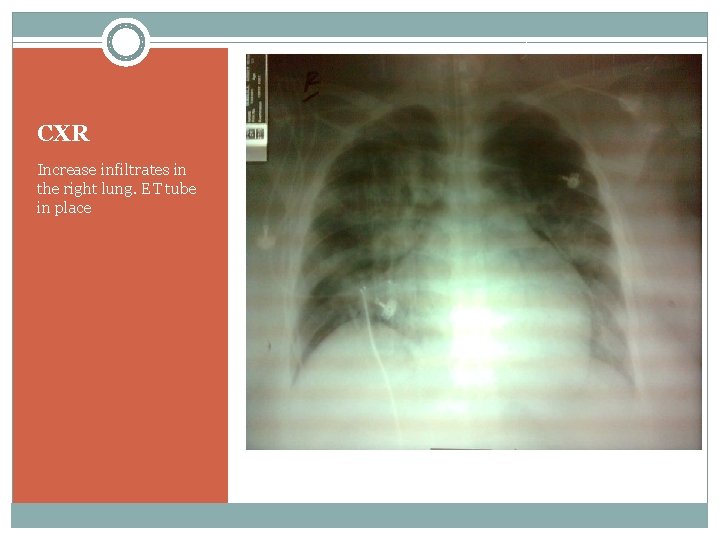
CXR Increase infiltrates in the right lung. ET tube in place
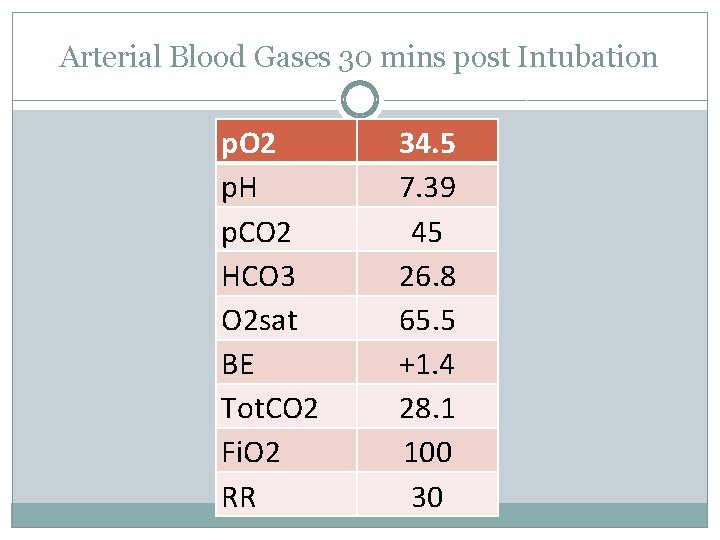
Arterial Blood Gases 30 mins post Intubation p. O 2 p. H p. CO 2 HCO 3 O 2 sat BE Tot. CO 2 Fi. O 2 RR 34. 5 7. 39 45 26. 8 65. 5 +1. 4 28. 1 100 30
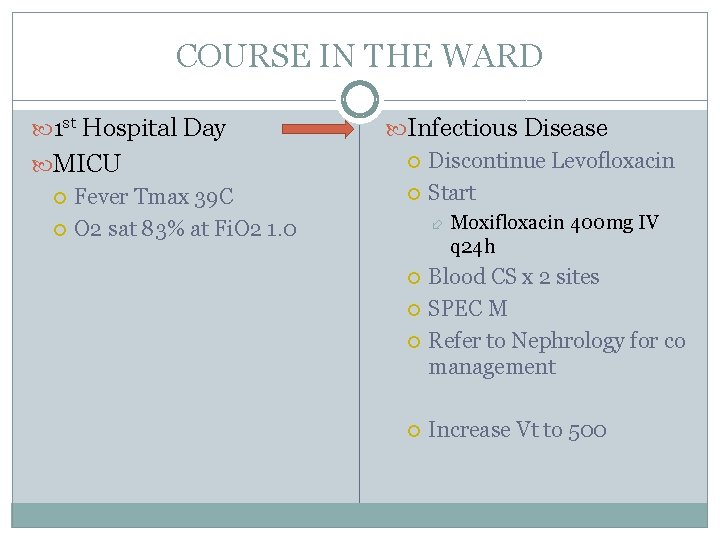
COURSE IN THE WARD 1 st Hospital Day MICU Fever Tmax 39 C O 2 sat 83% at Fi. O 2 1. 0 Infectious Disease Discontinue Levofloxacin Start Moxifloxacin 400 mg IV q 24 h Blood CS x 2 sites SPEC M Refer to Nephrology for co management Increase Vt to 500
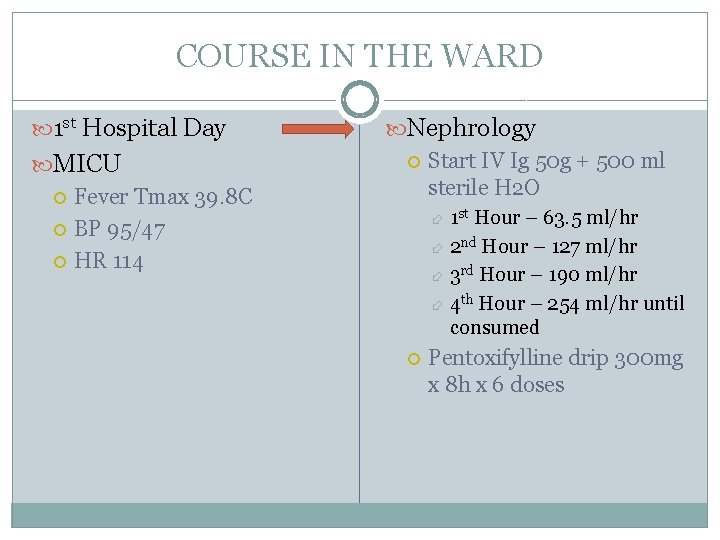
COURSE IN THE WARD 1 st Hospital Day MICU Fever Tmax 39. 8 C BP 95/47 HR 114 Nephrology Start IV Ig 50 g + 500 ml sterile H 2 O 1 st Hour – 63. 5 ml/hr 2 nd Hour – 127 ml/hr 3 rd Hour – 190 ml/hr 4 th Hour – 254 ml/hr until consumed Pentoxifylline drip 300 mg x 8 h x 6 doses

Pentoxifylline in severe sepsis: a double-blind, randomized placebocontrolled study KH STAUBACH, J SCHRÖDER, P ZABEL AND F STÜBER DEPT. OF SURGERY, MEDICAL UNIVERSITY OF LÜBECK AND KIEL, FORSCHUNGSZENTRUM BORSTEL Critical Care 1998, 2(Suppl 1): P 017 doi: 10. 1186/cc 147

51 patients MOF-score lower in POF treated patients Pa. O 2/Fio. O 2 -ratio was significantly improved in POF treated patients Pressure-adjusted heart rate (HR×CVP/MAP) was significantly improved from day 6 to day 10 (P < 0. 05)

Polyclonal Intravenous Immunoglobulin for the Treatment of Severe Sepsis and Septic Shock in Critically Ill Adults: A Systematic Review and Meta-analysis CONCLUSION: DEMONSTRATES AN OVERALL REDUCTION IN MORTALITY WITH THE USE OF IVIG FOR THE ADJUNCTIVE TREATMENT OF SEVERE SEPSIS AND SEPTIC SHOCK IN ADULTS Critical Care Medicine: Kevin B. Laupland, MD, MSc; Andrew W. Kirkpatrick, MD; Anthony Delaney, MBBS, MSc. Published: 01/14/2008

COURSE IN THE WARD 1 st Hospital Day MICU Fever Tmax 39. 8 C BP 95/47 HR 114 CVP 20 Nephrology Start Dopamine 400 mg/100 ml PNSS at 3 mcg/kg/min Dobutamine 500 mg/1 ooml PNSS at 10 mcg/kg/min Voluven 6% 500 ml x 25 oml/hr Hydrocortisone 50 mg IV q 8 h Furosemide 120 mg IV now

COURSE IN THE WARD 1 st Hospital Day MICU Albumin 3 Nephrology Plasma Cortisol CBC, CXR portable , DDIMER, CRP, Spec 16, Urinalysis, ABGs, Urine CS Foley Catheter inserted Start Esomeprazole 40 mg IVOD Human Albumin 25% 100 ml x 1 hr
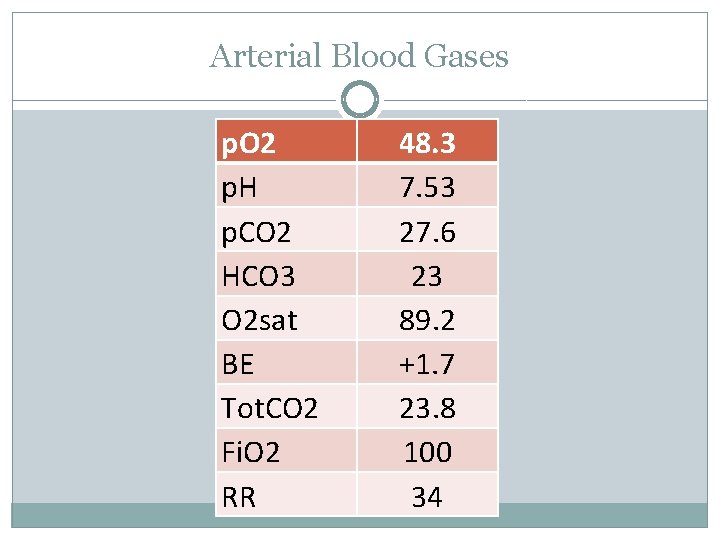
Arterial Blood Gases p. O 2 p. H p. CO 2 HCO 3 O 2 sat BE Tot. CO 2 Fi. O 2 RR 48. 3 7. 53 27. 6 23 89. 2 +1. 7 23. 8 100 34

COURSE IN THE WARD 1 st Hospital Day MICU VS O 2 sats 83% BP 123/64 Mech Vent Settings AC Fi. O 2 1. 0 Vt 400 RR 34 PEEP 20 Pulmonology Shift Berodual to Terbutaline nebulization q 8 h Acetylcysteine neb 600 mg
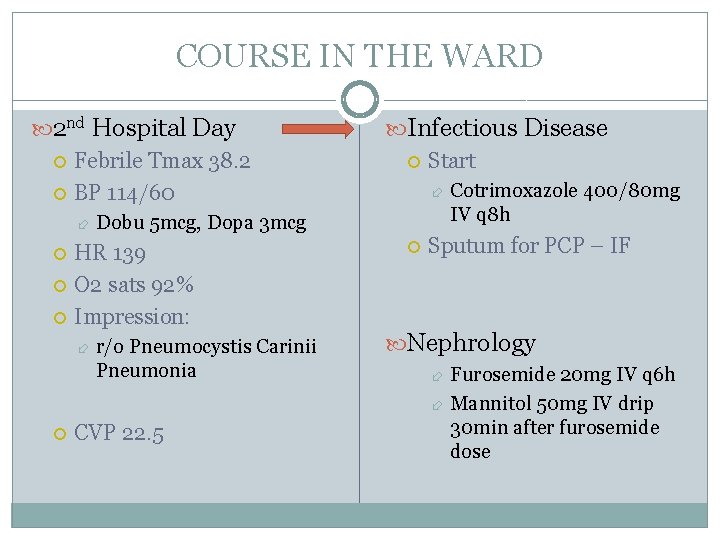
COURSE IN THE WARD 2 nd Hospital Day Febrile Tmax 38. 2 BP 114/60 Dobu 5 mcg, Dopa 3 mcg HR 139 O 2 sats 92% Impression: Infectious Disease Start r/o Pneumocystis Carinii Pneumonia CVP 22. 5 Cotrimoxazole 400/80 mg IV q 8 h Sputum for PCP – IF Nephrology Furosemide 20 mg IV q 6 h Mannitol 50 mg IV drip 30 min after furosemide dose

COURSE IN THE WARD 2 ND Hospital Day As treatment for fibroproliferative phase of ARDS Pulmonology Start Methylprednisolone 4 mg/kg q 8 h Decrease Fi. O 2 to 0. 85 0. 75

Methylprednisolone Infusion in Early Severe ARDS*Results of a Randomized Controlled Trial CONCLUSIONS: METHYLPREDNISOLONE-INDUCED DOWN-REGULATION OF SYSTEMIC INFLAMMATION WAS ASSOCIATED WITH SIGNIFICANT IMPROVEMENT IN PULMONARY AND EXTRAPULMONARY ORGAN DYSFUNCTION AND REDUCTION IN DURATION OF MECHANICAL VENTILATION AND ICU LENGTH OF AU Meduri S TGU; A YGolden E; Freire AX; Taylor E; Zaman M; Carson SJ; Gibson M; Umberger R SO Chest. 2007 Apr; 131(4): 954 -63
COURSE IN THE WARD 3 RD Hospital Day HR 120 s, RR 30 s, dyspnea O 2 sats 95% at Fi. O 2 0. 75 ECG sinus tachycardia Hold Terbutaline Increase Fi. O 2 to 1. 0 Nephrology Na 151 Shift IVF to 1/2 NSS x 40 ml/hr Dec Furosemide 20 mg q 8 h Dec Mannitol 50 mg to q 8 h Give 2 nd dose of IV Ig

COURSE IN THE WARD 4 th Hospital Day Keep decreasing Fi. O 2 below 1. 0 to keep O 2 sats above 90% 7 th Hospital Day 8 th Hospital day VS BP 120/70 Afebrile O 2 sats 96% Weaning from Mech vent started Patient was transferred out of MICU Pip-Tazo Discontinued Methylprednisolone tapering started
COURSE IN THE WARD 14 TH Hospital Day VS Patient was extubated Shifted to MVM 0. 5 BP 120/70 O 2 sats 92% On T-piece Fi. O 2 0. 35 RSB 18. 87 15 th Hospital Day ABGs p. O 2 – 75. 7 (115) At MVM 0. 5 Hooked to BIPAP 15 EPAP 5 SIDEFLOW 70

COURSE IN THE WARD 17 TH Hospital Day ABGs p. O 2 – 85. 6 on BIPAP 21 st Hospital Day CBC (07/27/09) Urine CS E. Coli WBC – 23. 65 (14. 4) Sensitive to Cefuroxime Imp: Hospital Acquired UTI BIPAP shifted to O 2 at 3 LPM Start Cefuroxime 500 mg BID Foley cath removed

COURSE IN THE WARD 28 TH Hospital Day HRCT done 31 st Hospital Day MGH

Clinical Course of ARDS Exudative Phase (Day 1 -7) Proliferative Phase (Day 7 -21) Fibrotic Phase

FINAL DIAGNOSIS • Severe Complicated Pneumonia H 1 N 1 infection with • • Acute Respiratory Distress Syndrome Diabetes Mellitus, Type 2, Non insulin Requiring Obese Class III Hypertension Stage I Polycystic Ovarian Syndrome
Influenza A (H 1 N 1)
Influenza is usually a respiratory infection Transmission Regular person-to-person transmission Primarily through contact with respiratory droplets Transmission from objects (fomites) possible National Center for Disease Prevention and Control, DOH

Key Characteristics Communicability Viral shedding can begin 1 day before symptom onset Peak shedding first 3 days of illness Correlates with temperature Subsides usually by 5 -7 th day in adults Infants, children and the immuno-compromised may shed the virus longer National Center for Disease Prevention and Control, DOH

Incubation period Time from exposure to onset of symptoms 1 to 4 days (average = 2 days) Seasonality In temperate zones, sharp peaks in winter months In tropical zones, circulates year-round with seasonal increases. National Center for Disease Prevention and Control, DOH

Individuals at Increased Risk for Hospitalizations and Death �Elderly > 65 years �Children less than two years �Certain chronic diseases Heart or lung disease, including asthma Metabolic disease, including diabetes HIV/AIDs, other immuno-suppression Conditions that can compromise respiratory function or the handling of respiratory secretions �Pregnant women National Center for Disease Prevention and Control, DOH

INTENSIVE-CARE PATIENTS WITH SEVERE NOVEL INFLUENZA A (H 1 N 1) VIRUS INFECTION --- MICHIGAN, JUNE 2009 10 patients wiith Influenza A(H 1 N 1) and ARDS admitted at ICU. Of the 10 patients 9 were obese (BMI>30) including 7 who are extremely obese (BMI>40). Five had pulmonary emboli; Nine had MODS. 3 patients died. Clinicians should be aware of the potential for severe complications of H 1 N 1 virus infection in extremely obese patients.

Conclusion Predominance of males High prevalence of obesity Frequency of clinically significant pulmonary emboli and MODS

Influenza A Viruses Influenza A viruses categorized by subtype • Classified according to two surface proteins Hemagglutinin (H) – 16 known Neuraminidase (N) – 9 known N H National Center for Disease Prevention and Control, DOH

Nomenclature Virus type Strain number Virus subtype A / Sydney / 05 / 97 (H 3 N 2) Place virus isolated Year isolated National Center for Disease Prevention and Control, DOH

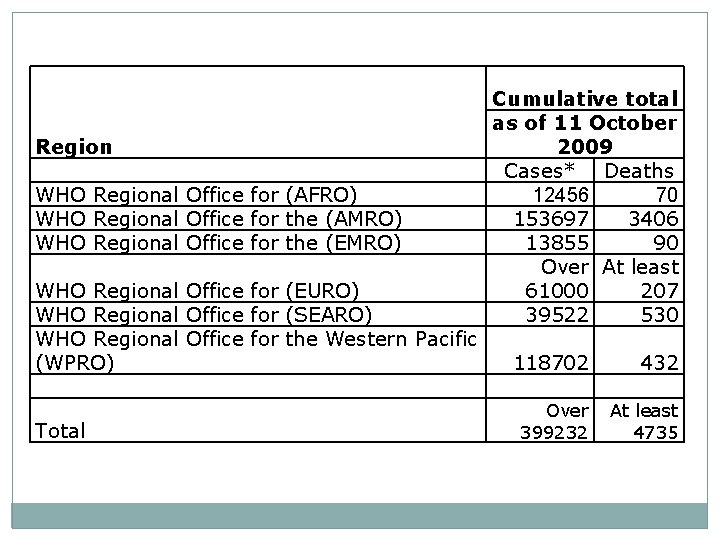
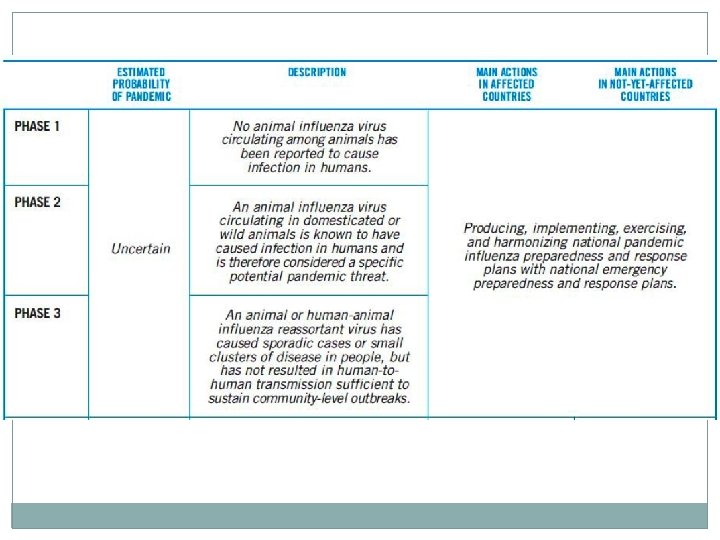
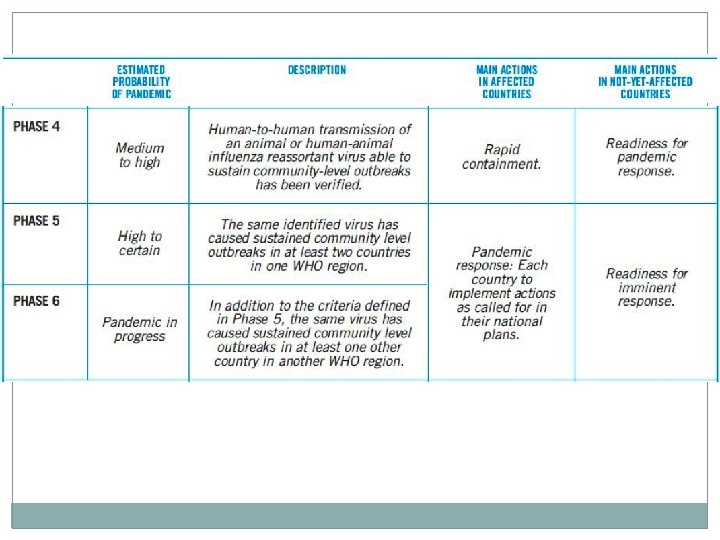
Region WHO Regional Office for (AFRO) WHO Regional Office for the (AMRO) WHO Regional Office for the (EMRO) WHO Regional Office for (EURO) WHO Regional Office for (SEARO) WHO Regional Office for the Western Pacific (WPRO) Total Cumulative total as of 11 October 2009 Cases* Deaths 12456 70 153697 3406 13855 90 Over At least 61000 207 39522 530 118702 432 Over At least 399232 4735

Influenza A (H 1 N 1) is a novel virus �Unusual combination of genetic material from pigs, birds & humans which have re-assorted �Affects all age groups �Vaccines for human seasonal flu can not protect humans against the novel virus National Center for Disease Prevention and Control, DOH

Swine Influenza Viruses �RNA viruses �Pigs can be infected by avian influenza and human influenza viruses as well as swine influenza viruses. �Re-assort and new viruses that are a mix of swine, human and avian influenza viruses can EMERGE National Center for Disease Prevention and Control, DOH
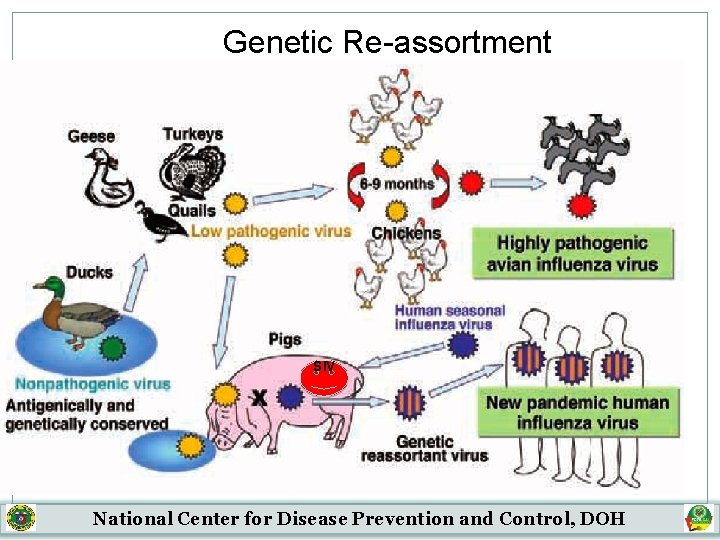
Genetic Re-assortment SIV National Center for Disease Prevention and Control, DOH

Signs & Symptoms of Influenza A (H 1 N 1) �Fever �Lethargy �Lack of appetite �Coughing �Runny Nose �Sore throat �Nausea / Vomiting �Diarrhea National Center for Disease Prevention and Control, DOH

Swine H 1 N 1 vs. Human H 1 N 1 �swine H 1 N 1 flu virus NOT the same as human H 1 N 1 virus �antigenically very different from human H 1 N 1 viruses �vaccines for human seasonal flu can not protect humans from swine H 1 N 1 National Center for Disease Prevention and Control, DOH

Transmission: Food-Borne? �NO �Influenza A (H 1 N 1) viruses are not transmitted through food �Safe to eat properly handled and cooked pork and pork products �Cook pork at an internal temperature of 70°C (160°F) National Center for Disease Prevention and Control, DOH

Diagnosis and Laboratory Confirmation �Clinically diagnosed �Respiratory Specimen • • first 4 to 5 days of illness can shed for 10 days or longer �Specimens sent to US CDC • ONLY laboratory that can isolate and identify swine influenza type A virus National Center for Disease Prevention and Control, DOH

Specimens �Upper respiratory tract specimens as recommended are the most appropriate. �taken from the deep nostrils (nasal swab), nasopharynx (nasopharyngeal swab), Nasopharyngeal aspirate, throat or bronchial aspirate.

Laboratory Diagnosis Rapid Influenza Diagnostic Test � Antigen detection test that detect influenza viral nucleoprotein antigen � Can provide results within 30 minutes � Sensitivity 40 -69%
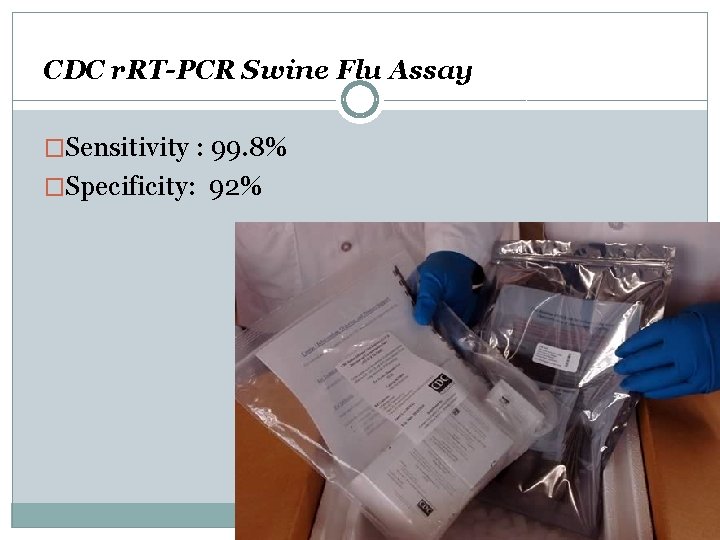
CDC r. RT-PCR Swine Flu Assay �Sensitivity : 99. 8% �Specificity: 92%
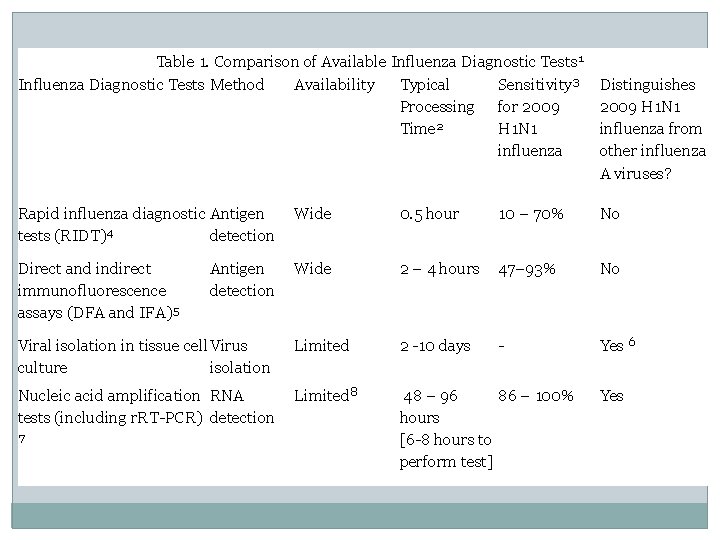
Table 1. Comparison of Available Influenza Diagnostic Tests 1 Influenza Diagnostic Tests Method Availability Typical Sensitivity 3 Processing for 2009 Time 2 H 1 N 1 influenza Distinguishes 2009 H 1 N 1 influenza from other influenza A viruses? Rapid influenza diagnostic Antigen tests (RIDT)4 detection Wide 0. 5 hour 10 – 70% No Direct and indirect immunofluorescence assays (DFA and IFA)5 Wide 2 – 4 hours 47– 93% No Viral isolation in tissue cell Virus culture isolation Limited 2 -10 days - Yes 6 Nucleic acid amplification RNA tests (including r. RT-PCR) detection 7 Limited 8 48 – 96 86 – 100% hours [6 -8 hours to perform test] Antigen detection Yes

Treatment �Influenza A (H 1 N 1) is sensitive to: Oseltamivir (tamiflu) Zanamivir �Self medication is discouraged, may induce drug resistance �Chemoprophylaxis Oseltamivir National Center for Disease Prevention and Control, DOH
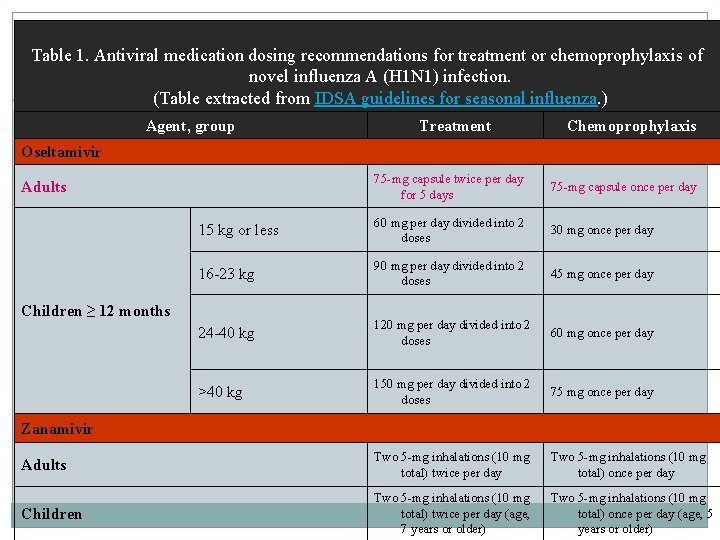
Table 1. Antiviral medication dosing recommendations for treatment or chemoprophylaxis of novel influenza A (H 1 N 1) infection. (Table extracted from IDSA guidelines for seasonal influenza. ) Agent, group Treatment Chemoprophylaxis Oseltamivir 75 -mg capsule twice per day for 5 days 75 -mg capsule once per day 15 kg or less 60 mg per day divided into 2 doses 30 mg once per day 16 -23 kg 90 mg per day divided into 2 doses 45 mg once per day 24 -40 kg 120 mg per day divided into 2 doses 60 mg once per day >40 kg 150 mg per day divided into 2 doses 75 mg once per day Adults Two 5 -mg inhalations (10 mg total) twice per day Two 5 -mg inhalations (10 mg total) once per day Children Two 5 -mg inhalations (10 mg total) twice per day (age, 7 years or older) Two 5 -mg inhalations (10 mg total) once per day (age, 5 years or older) Adults Children ≥ 12 months Zanamivir

CDC Health Advisory � Distributed via Health Alert Network July 09, 2009 Three Reports of Oseltamivir Resistant Novel Influenza A (H 1 N 1) Viruses

ANTI VIRAL CHEMOPROPHYLAXIS �Post-exposure prophylaxis for health care workers, first responders and workers who did not have adequate PPE when in contact. �Anti-viral prophylaxis is not recommended to close contacts except when they belong to high risk group.

Vaccine �Process of production is underway, but may take 5 – 6 months �Seasonal influenza vaccine provides protection against the seasonal human influenza strains only National Center for Disease Prevention and Control, DOH
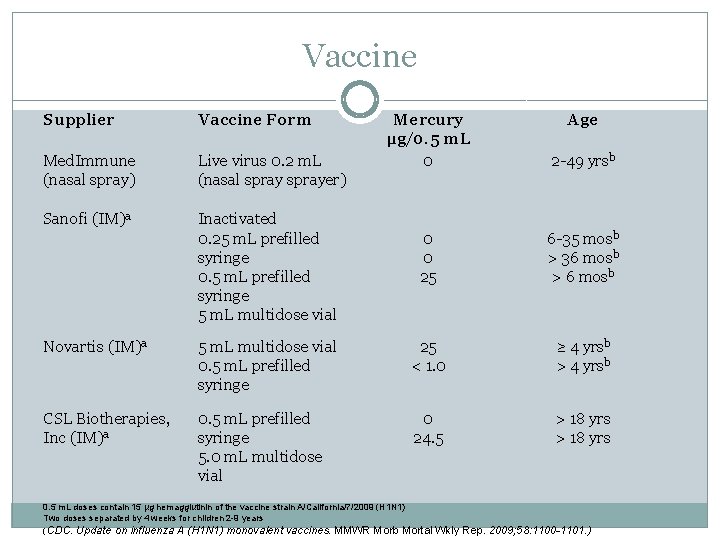
Vaccine Supplier Vaccine Form Med. Immune (nasal spray) Live virus 0. 2 m. L (nasal sprayer) Sanofi (IM)a Inactivated 0. 25 m. L prefilled syringe 0. 5 m. L prefilled syringe 5 m. L multidose vial Mercury µg/0. 5 m. L 0 Age 2 -49 yrsb 0 0 25 6 -35 mosb > 36 mosb > 6 mosb Novartis (IM)a 5 m. L multidose vial 0. 5 m. L prefilled syringe 25 < 1. 0 ≥ 4 yrsb > 4 yrsb CSL Biotherapies, Inc (IM)a 0. 5 m. L prefilled syringe 5. 0 m. L multidose vial 0 24. 5 > 18 yrs 0. 5 m. L doses contain 15 µg hemagglutinin of the vaccine strain A/California/7/2009 (H 1 N 1) Two doses separated by 4 weeks for children 2 -9 years (CDC. Update on influenza A (H 1 N 1) monovalent vaccines. MMWR Morb Mortal Wkly Rep. 2009; 58: 1100 -1101. )
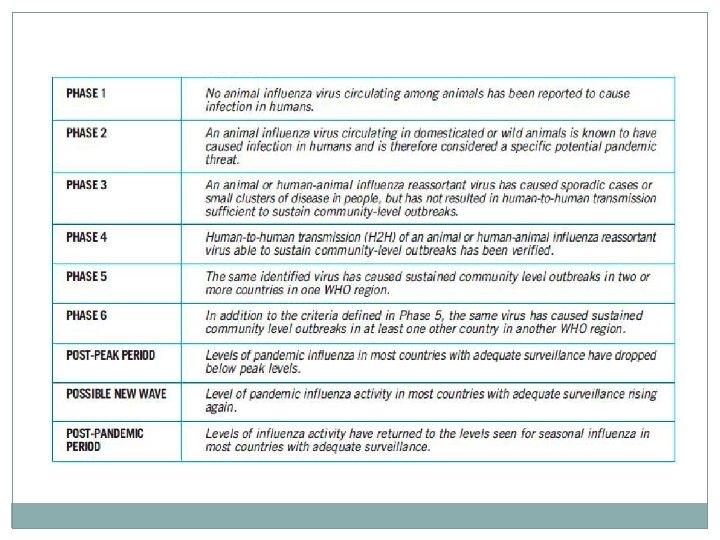
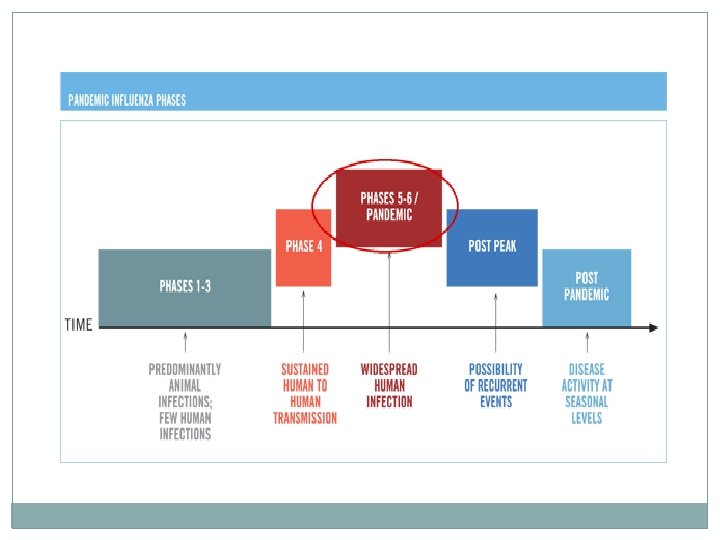

INTERIM GUIDELINES NO. 2: INFECTION CONTROL AND USE OF PERSONAL PROTECTIVE EQUIPMENT IN INFLUENZA A HEALTH CARE PERSONNEL �Standard and droplet precautions when working in direct contact �If there is risk of splashes: particulate respirator; eye protection; clean, non-sterile, long sleeved gown; sterile gloves �Frequent and proper handwashing

�Isolate patient in a single room. �Reinforce standard precautions with droplet and contact precaution �Use appropriate Personal Protective Equipment for all those entering patients rooms. �Restrict number of visitors. �Healthcare workers on direct contact should monitor their own temperature 2 X a day and report any febrile event.

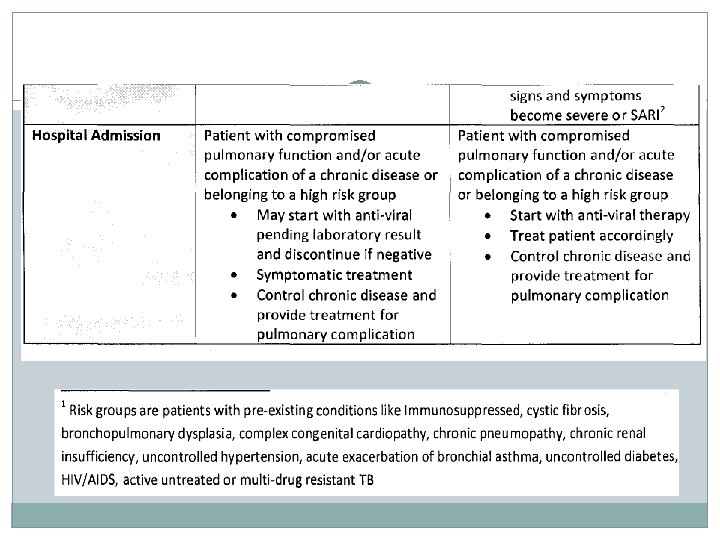
INTERIM GUIDELINES NO. 17: REVISED CLINICAL CASE MANAGEMENT OF INFLUENZA A(H 1 N 1) VIRUS INFECTION



Vaccination Influenza vaccine is the best prevention for seasonal influenza. Inactivated viruses in the vaccine developed from three circulating strains (generally 2 Type A and 1 Type B strain) Therefore, seasonal “flu shot” only works for 3 influenza subtypes and will not work on pandemic strains. Live, intranasal spray vaccine for healthy non- pregnant persons 5 -49 years Inactivated, injectable vaccine for persons 6 months and older National Center for Disease Prevention and Control, DOH

Influenza Viruses Classified into types A, B, and C • • • Only Types A and B cause significant disease Types B and C limited to humans Type A viruses More virulent Affect many species C Goldsmith, CDC National Center for Disease Prevention and Control, DOH

American Journal of Kidney Diseases PREHYPERTENSION, OBESITY, AND RISK OF KIDNEY DISEASE: 20 YEAR FOLLOW UP THE HUNT 1 STUDY IN NORWAY (JUNE 11, 2009) Participants with prehypertension are not at increased risk of serious kidney outcomes if BMI is less than 30 kg/m 2. However, the risk of Kidney disease increases substantially if prehypertension is present in obese participants.
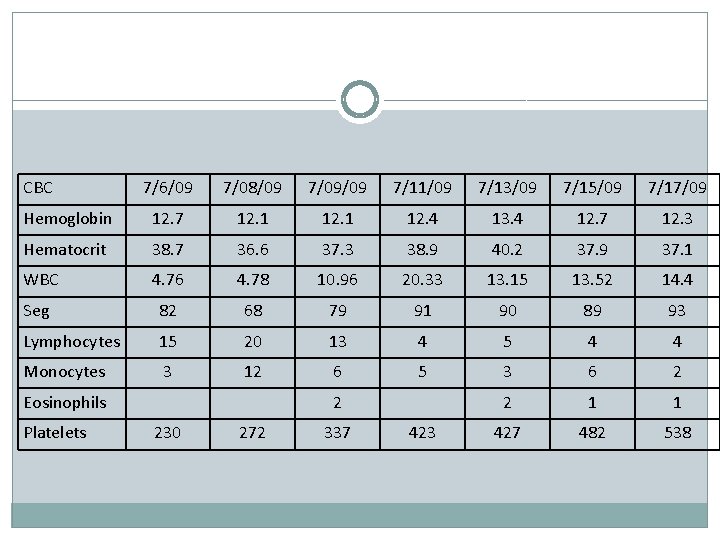
CBC 7/6/09 7/08/09 7/09/09 7/11/09 7/13/09 7/15/09 7/17/09 Hemoglobin 12. 7 12. 1 12. 4 13. 4 12. 7 12. 3 Hematocrit 38. 7 36. 6 37. 3 38. 9 40. 2 37. 9 37. 1 WBC 4. 76 4. 78 10. 96 20. 33 13. 15 13. 52 14. 4 Seg 82 68 79 91 90 89 93 Lymphocytes 15 20 13 4 5 4 4 Monocytes 3 12 6 5 3 6 2 2 1 1 427 482 538 Eosinophils Platelets 2 230 272 337 423
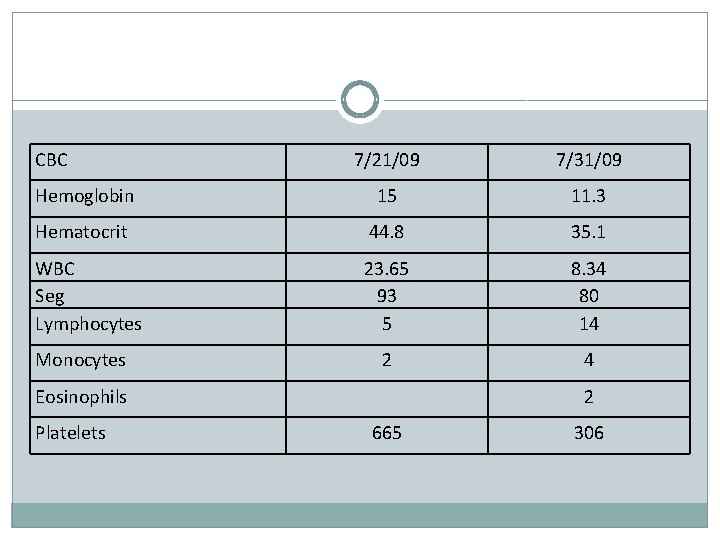
CBC 7/21/09 7/31/09 Hemoglobin 15 11. 3 Hematocrit 44. 8 35. 1 WBC Seg Lymphocytes 23. 65 93 5 8. 34 80 14 2 4 Monocytes Eosinophils Platelets 2 665 306
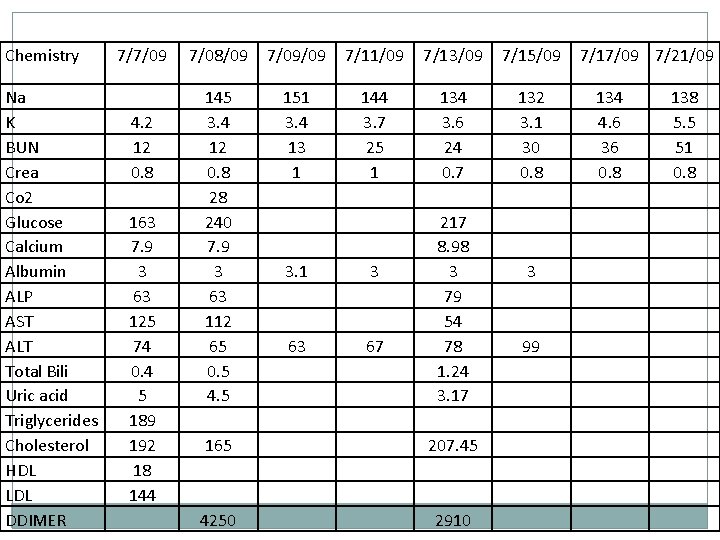
Chemistry Na K BUN Crea Co 2 Glucose Calcium Albumin ALP AST ALT Total Bili Uric acid Triglycerides Cholesterol HDL LDL DDIMER 7/7/09 4. 2 12 0. 8 163 7. 9 3 63 125 74 0. 4 5 189 192 18 144 7/08/09 7/09/09 7/11/09 7/13/09 7/15/09 145 3. 4 12 0. 8 28 240 7. 9 3 63 112 65 0. 5 4. 5 151 3. 4 13 1 144 3. 7 25 1 134 3. 6 24 0. 7 132 3. 1 30 0. 8 3. 1 3 63 67 217 8. 98 3 79 54 78 1. 24 3. 17 165 207. 45 4250 2910 3 99 7/17/09 7/21/09 134 4. 6 36 0. 8 138 5. 5 51 0. 8

CORTISOL 1659. 731 nmol/L 7/13/09 ESR 70 mm/hr Hb. A 1 c – 8. 1%

HRCT: Predominantly interstitial infiltrates in both lungs with associated ground glass opacities in both upper lobes. The interstitial infiltrates may represent interstitial fibrosis with superimposed acute alveolar inflammatory or infectious process. Minimal pneumomediastinum and subcutaneous emphysema.

CXR: Jul 6 – Hazy infiltrates in the right upper lobe likely due to pneumonia. There also infiltrates in the right paracardiac and left lower lobe. Heart is magnified. Bones and soft tissues are normal. Jul 7 – Increase infiltrates in the right lung. ET tube in place. Jul 8 – right suprahilar and paracardiac infiltrates appear confluent, however there is partial clearing of the infiltrates seen in the peripheral chest. Infiltrates in the left are unchanged. Jul 13 – Complete resorption of the right side pneumothorax. Slight clearing of Pulmonary congestion. ET tube in place. Jul 17 – No significant change in the opacities in both lungs. Minimal subcutaneous emphysema. Jul 19 – Moderate resorption of subcutaneous emphysema. No significant change in infiltrates.
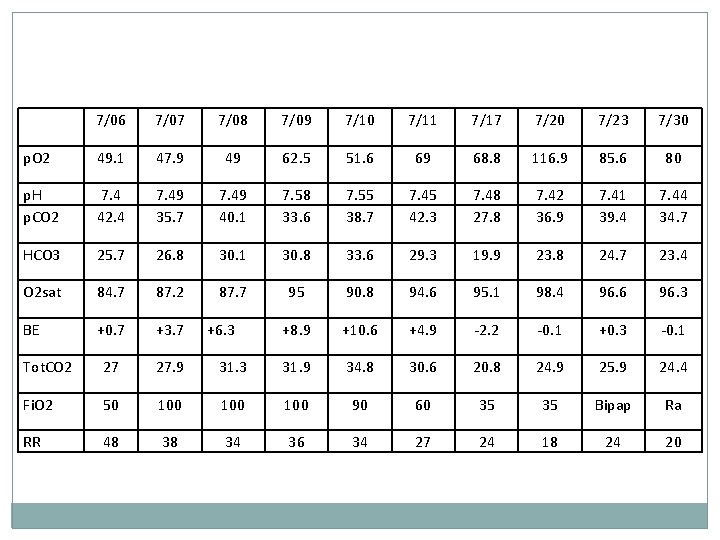
7/06 7/07 7/08 7/09 7/10 7/11 7/17 7/20 7/23 7/30 p. O 2 49. 1 47. 9 49 62. 5 51. 6 69 68. 8 116. 9 85. 6 80 p. H p. CO 2 7. 4 42. 4 7. 49 35. 7 7. 49 40. 1 7. 58 33. 6 7. 55 38. 7 7. 45 42. 3 7. 48 27. 8 7. 42 36. 9 7. 41 39. 4 7. 44 34. 7 HCO 3 25. 7 26. 8 30. 1 30. 8 33. 6 29. 3 19. 9 23. 8 24. 7 23. 4 O 2 sat 84. 7 87. 2 87. 7 95 90. 8 94. 6 95. 1 98. 4 96. 6 96. 3 BE +0. 7 +3. 7 +8. 9 +10. 6 +4. 9 -2. 2 -0. 1 +0. 3 -0. 1 Tot. CO 2 27 27. 9 31. 3 31. 9 34. 8 30. 6 20. 8 24. 9 25. 9 24. 4 Fi. O 2 50 100 100 90 60 35 35 Bipap Ra RR 48 38 34 36 34 27 24 18 24 20 +6. 3

The FDA has approved 4 vaccine preparations. The following data highlight relevant issues: All influenza vaccine preparations in the United States for the 2009 -2010 season contain residual egg protein and none contain adjuvant; Children 6 months to 9 years of age who are given influenza A (H 1 N 1) monovalent vaccine should receive 2 doses separated by about 4 weeks; persons ≥ 10 years of age should receive 1 dose; The influenza A (H 1 N 1) monovalent vaccines were made according to standards used for seasonal and influenza vaccines and have the same age group indications, precautions, and contraindications as vaccines that are FDA-approved for seasonal flu; preliminary data indicate that the safety and efficacy of the 2009 Influenza A (H 1 N 1) monoclonal vaccine is the same as winter; There is minimal evidence of significant antigenic change since the first characterization of the virus in April 2009, indicating that the virus continues to be well matched with the vaccine strain; and The vaccines of the 4 suppliers have some differences that are important to recognize: for seasonal flu vaccines; Side effects, including local pain at the injection site, were reported in 46% of recipients, and systemic reactions (headache, malaise or myalgias) were reported in 45%; the safety profile is consistent with the experience with seasonal flu vaccine; Influenza activity due to influenza A (H 1 N 1) increased in September 2009 and is expected to continue through fall and

Children 6 months to 9 years of age should receive 2 doses separated by 3 weeks. Children 10 years and older and adults should receive 1 dose. The following groups should receive the vaccine as soon as it becomes available: • Pregnant women; • People who live with or care for infants younger than 6 months of age; • Healthcare workers (HCWs) and emergency medical personnel; • Persons 6 months to 24 years of age; • Persons 25 -64 years of age who have chronic diseases (including immunodeficiency states) that pose risk for influenza. When more vaccine becomes available, the following persons should be vaccinated: • Healthy persons ages 25 -64 years; and • Adults 65 years of age and older.
- Slides: 97