Medical Genetics Mohammed ElKhateeb Dental Postgraduate MG Lec

Medical Genetics Mohammed El-Khateeb Dental Postgraduate MG - Lec. 1 22 nd June 2014

OBJECTIVES § Understanding of Basic genetics § Be able to draw, and understand, a family tree § Have awareness of when you should be considering a genetic condition § Have a working knowledge of the most important genetic conditions § Know how & when to refer to local specialist genetics services

What’s a ___? • Genetics : Is the branch of biology that deals with heredity and variation in all living organisms • The subfields of genetics : Ø Human genetics, Ø Animal genetics, Ø Plant genetics Ø Medical genetics

What’s a Medical Genetics? • Is the science or study of biological variation as it pertains to health and disease • Any application of genetic principles to medical practice. “Genetics – study of individual genes and their effects”: Includes studies of inheritance, mapping disease, genes, diagnosis, treatment, and genetic counseling

Foundations of Heredity Science § Variable traits are inherited § Gene – trait-specific unit of heredity § Alternative versions of a gene (alleles) determine the trait § Each parent transmits an allele to the offspring

History of Medical Genetics • Early Genetics • Mendel - 1860 s • Modern Experimental Genetics - 1900 s • Maize, drosophila, mouse • Medical Genetics - 1960 s to the present

Mendel Inheritance Australian monk who formulated fundamental law of heredity in early 1860 s. § Theories of inheritances. Gregor Mendel (1822 -1884) § Reshuffling of genes from generation to generations He studied mathematics at University of Vienna

Mendel studies seven characteristics in the garden pea
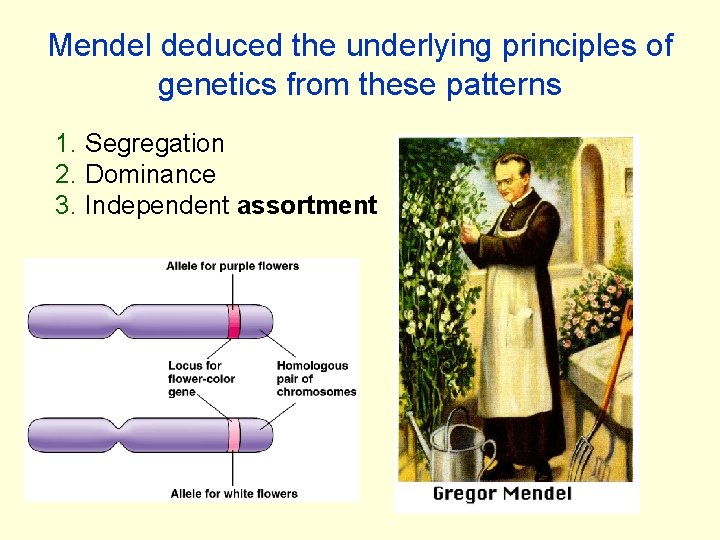
Mendel deduced the underlying principles of genetics from these patterns 1. Segregation 2. Dominance 3. Independent assortment

A Conceptual History of Medical Genetics 1900 1901 1902 1918 1931 1937 1955 1970 1976 Mendel’s Laws rediscovered Dominant inheritance of brachydactyly Inborn errors of metabolism Anticipation described Cytoplasmic inheritance of mitochondrial DNA Linkage of color blindness and hemophilia Human diploid chromosome number is 46 Amniocentesis for chromosomal disorders Tay-Sachs screening Human globin genes cloned

A Conceptual History of Medical Genetics 1985 Mendel’s Laws rediscovered PCR 1986 Duchenne muscular dystrophy gene 1986 Cystic Fibrosis gene 1987 Predictive genetic testing for Huntington Disease 1998 Decision to sequence entire human genome 1991 Medical genetics became an ABMS specialty 1991 2001 Draft sequence for the human genome Human genome sequence completed

Genetics – history and key concepts… 1860 s Mendel’s work on peas allows the conclusion that traits are inherited through discrete units passed from one generation to the next 1870 s Friedrich Miescher describes nucleic acids 1909 The word ‘gene’ coined by Danish botanist Wilhelm Johannsen 1910 Thomas Morgan’s work on fruitflies demonstrates that genes lie on chromosomes 1940 s Barbara Mc. Clintock describes mobile genetic elements in maize 1977 Phillip Sharp and Richard Roberts find that protein-coding genes are carried in segments 1944 Oswald Avery shows in bacteria that nucleic acids are the ‘transforming principle’ 1953 James Watson and Francis Crick publish the double helix model for DNA’s chemical structure 1958 Crick proposes the ‘central dogma’ for biological information flow: that DNA makes RNA makes protein 2001 initial results from the Human Genome Project published

Medical Genetics: 1950 s to the present § DNA Genetics • 1953 - Watson and Crick’s Double Helix • 1992 – 2003 Human Genome Project • 2003 -> the future of medical dx & tx § Prenatal Genetics • 1970 s - Prenatal Ultrasound & Amniocentesis § Inheritance of Genetically Complex Disorders • • • Non-Mendelian Genetics Genomic Imprinting Triple Nucleotide Repeats Mitochondrial Inheritance 1990 s - Neuropsychiatric Disorders, Diabetes, Cardiovascular § Interaction of genes with environmental triggers

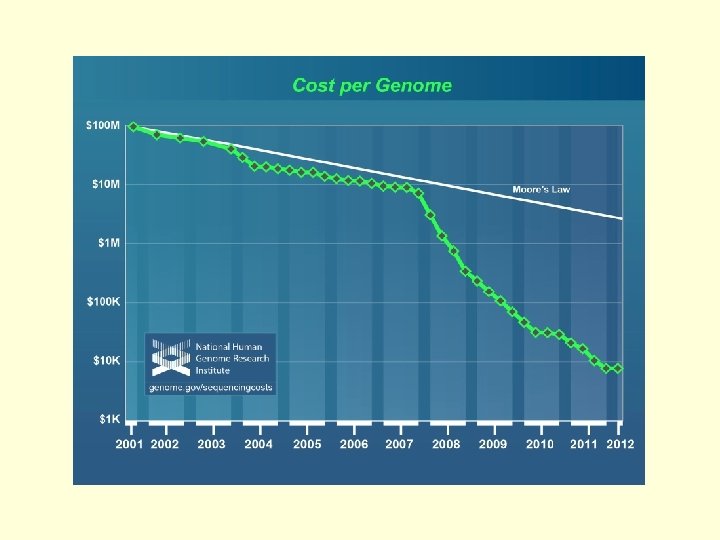
Human Genome Project § Proposed in 1985 § 1988. Initiated and § § § funded by NIH and US Dept. of Energy ($3 billion set aside) 1990. Work begins. 1998. Celera announces a 3 -year plan to complete the project years early Published in Science and Nature in February, 2001

What we’ve learned from our genome so far… § There a relatively small number of human genes, less than 30, 000, but they have a complex architecture that we are only beginning to understand appreciate. ü We know where 85% of genes are in the sequence. ü We don’t know where the other 15% are because we haven’t seen them “on” (they may only be expressed during fetal development). ü We only know what about 20% of our genes do so far. § So it is relatively easy to locate genes in the genome, but it is hard to figure out what they do.

Human genome content § 1 -2 % codes for protein products § 24% important for translation § 75% “junk” § Repetitive elements • Satellites (regular, mini-, micro-) • Transposons • Retrotransposons • Parasites

Some Facts Ø In human beings, 99. 9% bases are same Ø Remaining 0. 1% makes a person unique l Ø Different attributes / characteristics / traits § how a person looks § diseases he or she develops These variations can be: l l l Harmless (change in phenotype) Harmful (diabetes, cancer, heart disease, Huntington's disease, and hemophilia ) Latent (variations found in coding and regulatory regions, are not harmful on their own, and the change in each gene only becomes apparent under certain conditions e. g. susceptibility to heart attack)

Importance of Genetics to Medicine § 10% of the patients seen in GP have a disorder with a genetic component § 80% of MR due to genetic component § 2 -3% background population risk for a major birth defect 15% overall miscarriage risk for any pregnancy § 25 -50% first trimester miscarriage risk § 30 -50% first trimester losses due to chromosome anomalies § >30% pediatric hospital admissions due to GD § GD affect all major systems, any age, any race, male or female

Importance of Genetics to Medicine Ø Changing focus of medicine: § Primary care physicians vs specialists Prevention vs treatment § Genetic causation for both rare and common diseases § Human Genome Project Designer drugs § § Ø Problem based approach taken in medical schools Ø Genetics as the link between basic research & clinical observation

Categories of Genetic Disease • Genetic component of multifactorial illnesses – Polygenic conditions – Interaction of genetic & environmental factors • Addition or deletion of entire chromosomes or parts of chromosomes • Single gene disorders Ø Autosomal dominant Ø Autosomal recessive Ø X-linked
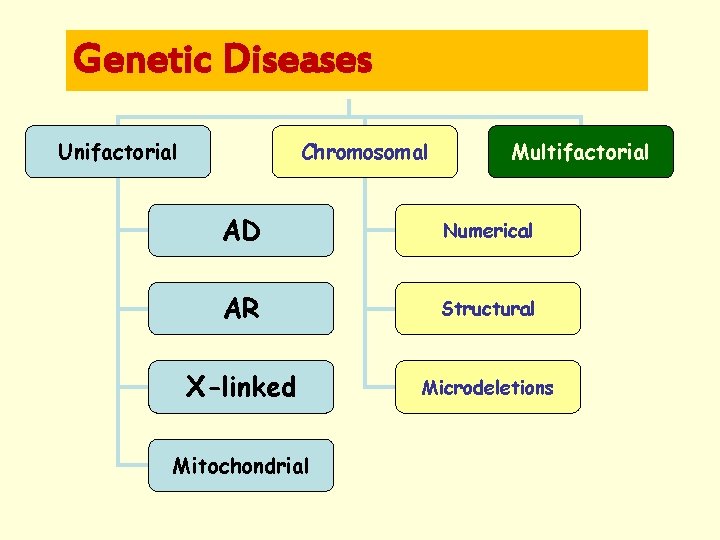
Genetic Diseases Unifactorial Chromosomal Multifactorial AD Numerical AR Structural X-linked Microdeletions Mitochondrial
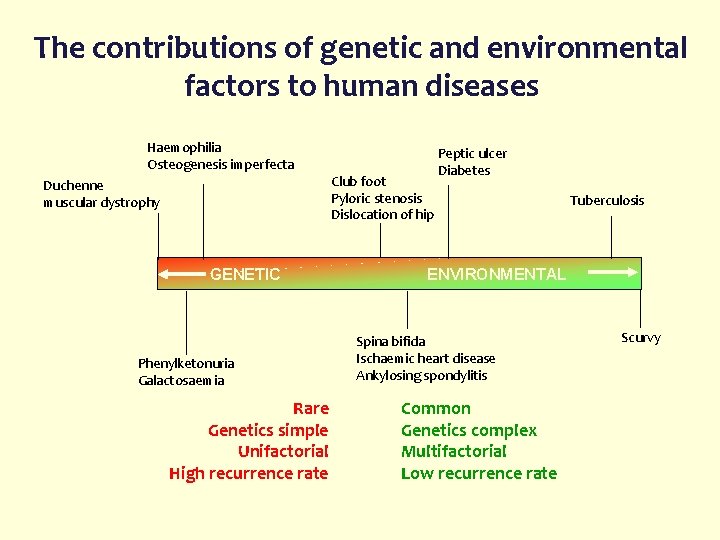
The contributions of genetic and environmental factors to human diseases Haemophilia Osteogenesis imperfecta Club foot Pyloric stenosis Dislocation of hip Duchenne muscular dystrophy GENETIC Phenylketonuria Galactosaemia Rare Genetics simple Unifactorial High recurrence rate Peptic ulcer Diabetes Tuberculosis ENVIRONMENTAL Spina bifida Ischaemic heart disease Ankylosing spondylitis Common Genetics complex Multifactorial Low recurrence rate Scurvy
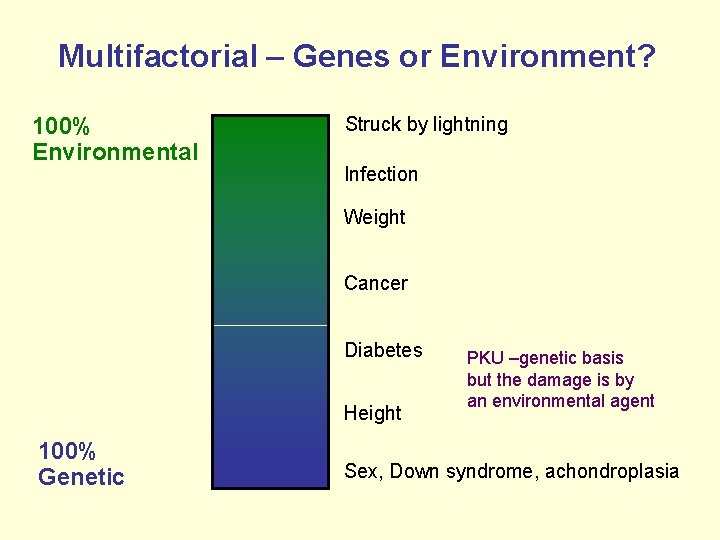
Multifactorial – Genes or Environment? 100% Environmental Struck by lightning Infection Weight Cancer Diabetes Height 100% Genetic PKU –genetic basis but the damage is by an environmental agent Sex, Down syndrome, achondroplasia

Polygenic diseases ØThe most common yet still the least understood of human genetic diseases ØResult from an interaction of multiple genes, each with a minor effect ØThe susceptibility alleles are common ØType I and type II diabetes, autism, osteoarthritis
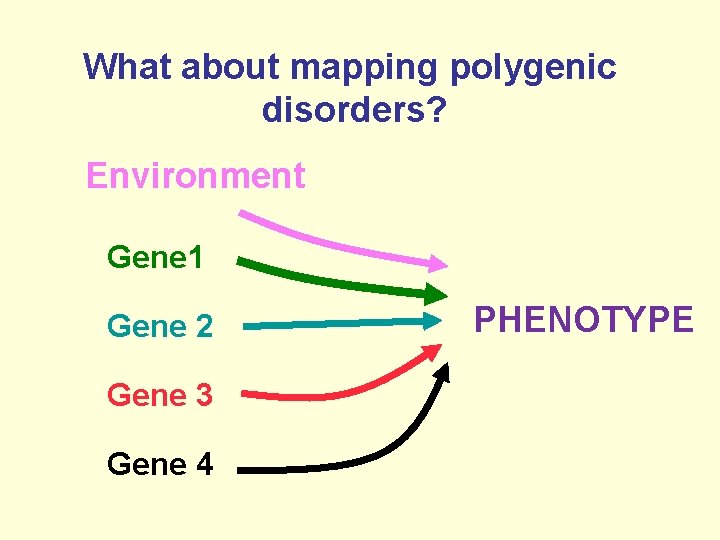
What about mapping polygenic disorders? Environment Gene 1 Gene 2 Gene 3 Gene 4 PHENOTYPE

Polygenic Diseases • The most common yet still the least understood of human genetic diseases – – Type I and type II diabetes Primary generalised osteoarthritis Hypertension Autism • Result from an interaction of multiple genes, each with a minor effect • Some inherited mutations and some environmental factors (somatic mutations)

Population Genetics § Identifies how much genetic variation exists in populations • Investigates factors, such as migration, population size, and natural selection, that change the frequency of a specific gene over time § Coupled with DNA technology, investigates evolutionary history and DNA identification techniques

Chromosomal Disorders Most mutations happen in the parent’s egg/ sperm - “one off” with no established family history Duplications (whole or part) § § Autosome trisomies (Down, Edwards, Patau) XY duplications (Klinefelter XXY, Triple X) Deletions § § Autosome deletions (Cri du chat, di George’s) XY deletions (Turner XO) Translocations § § Leukaemias (Philedelphia Chromosome) Sarcomas (Ewings) Inversions Rings…. .
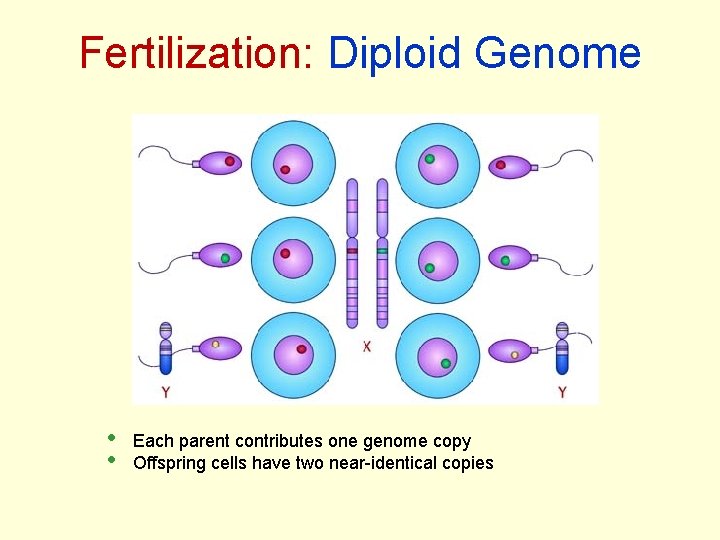
Fertilization: Diploid Genome • • Each parent contributes one genome copy Offspring cells have two near-identical copies
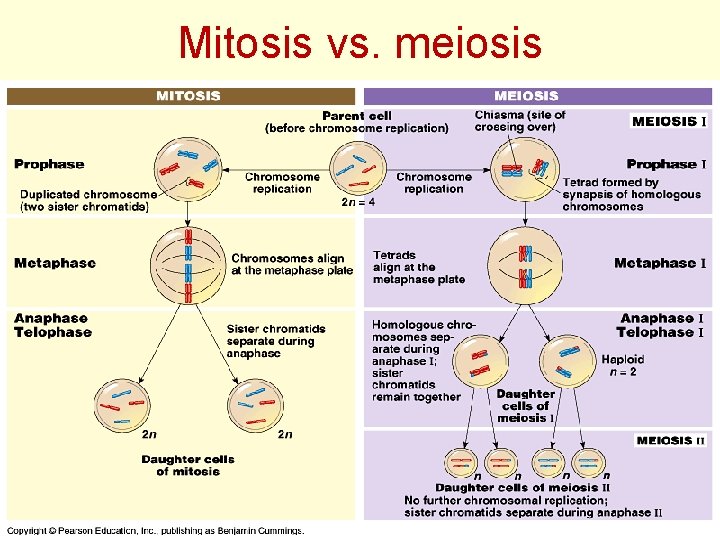
Mitosis vs. meiosis Meiosis KM 30
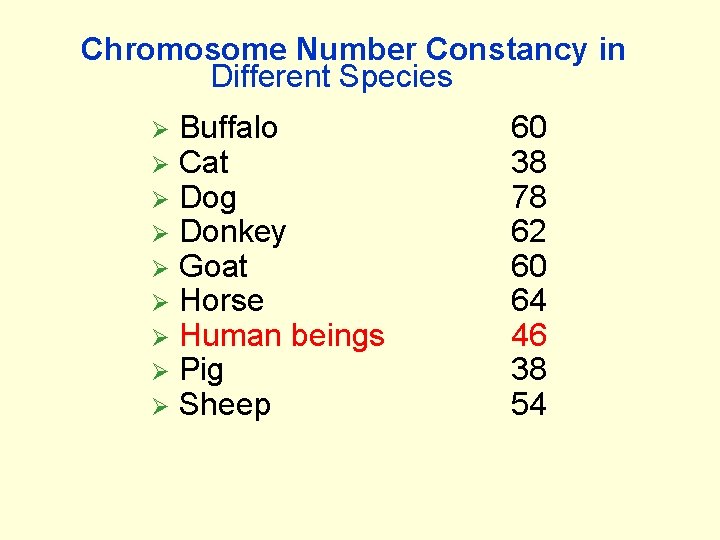
Chromosome Number Constancy in Different Species Ø Ø Ø Ø Ø Buffalo Cat Dog Donkey Goat Horse Human beings Pig Sheep 60 38 78 62 60 64 46 38 54

DNA DIFFERENCES TWO BASIC FACTORS and the same number of chromosomes and genes
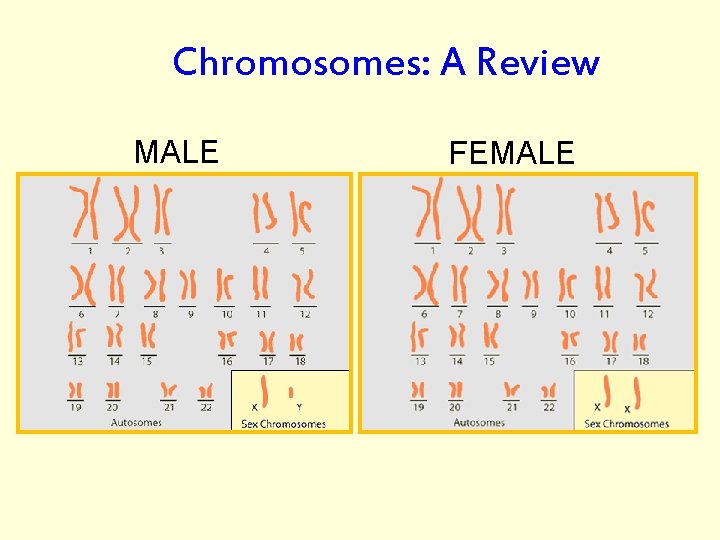
Chromosomes: A Review MALE FEMALE
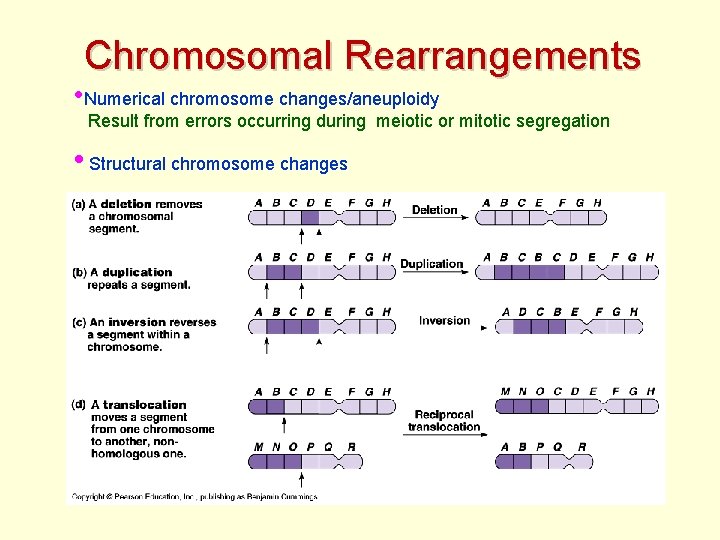
Chromosomal Rearrangements • Numerical chromosome changes/aneuploidy Result from errors occurring during meiotic or mitotic segregation • Structural chromosome changes

Single Gene Disorders FAutosomal recessive FAutosomal dominant FX-linked recessive FX-linked dominant
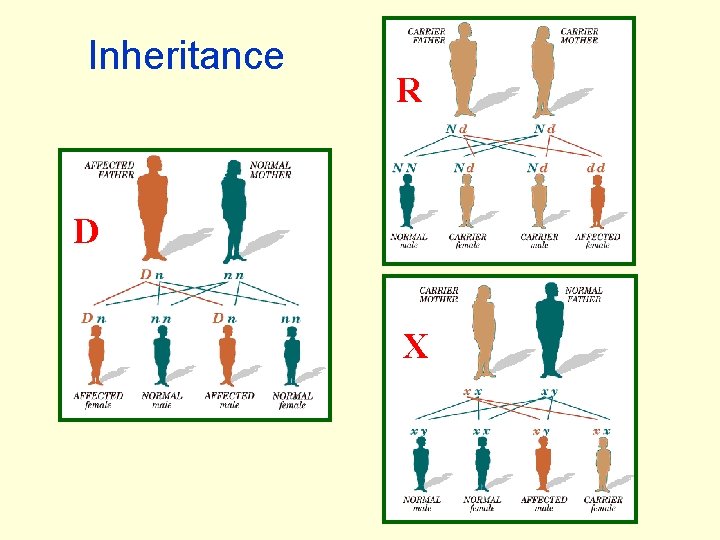
Inheritance R D X
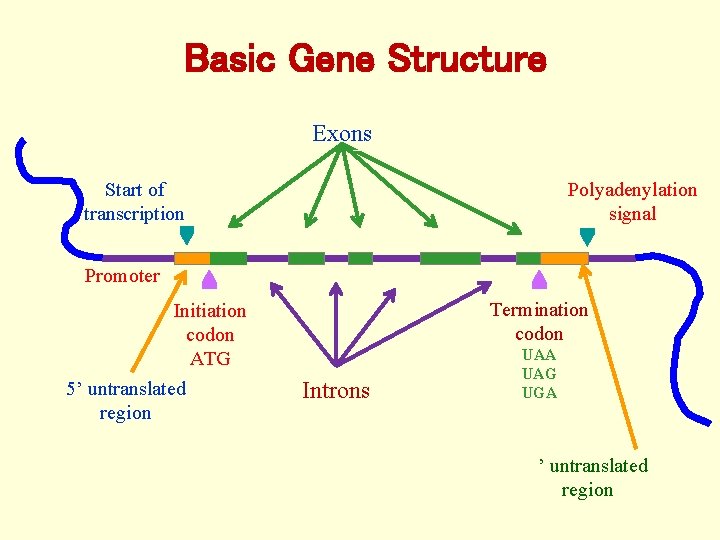
Basic Gene Structure Exons Polyadenylation signal Start of transcription Promoter Termination codon Initiation codon ATG 5’ untranslated region Introns UAA UAG UGA 3’ untranslated region

Single-Gene “Mendelian” Disorders • Structural proteins • Enzymes and inhibitors § § Osteogenesis imperfecta and Ehlers-Danlos (collagens); Marfan syndrome (fibrillin); Duchenne and Becker muscular dystrophies (dystrophin) Lysosomal storage diseases; SCID (adenosine deaminase); PKU (phenylalanine hydroxylase); Alpha-1 antitrypsin deficiency • Receptors § Familial hypercholesterolemia (LDL receptor) • Cell growth regulation § • § Neurofibromatosis type I (neurofibromin); Hereditary retinoblastoma (Rb) Transporters Cystic fibrosis (CFTR); Sickle cell disease (Hb); Thalassemias

What Can The Genes Tell Us? Ø Give us a better understanding of the underlying biology of the trait in question Ø Serve as direct targets for better treatments § Pharmacogenetics § Interventions Ø Give us better predictions of who might develop disease Ø Give us better predictions of the course of the disease Ø Lead to knowledge that can help find a cure or prevention
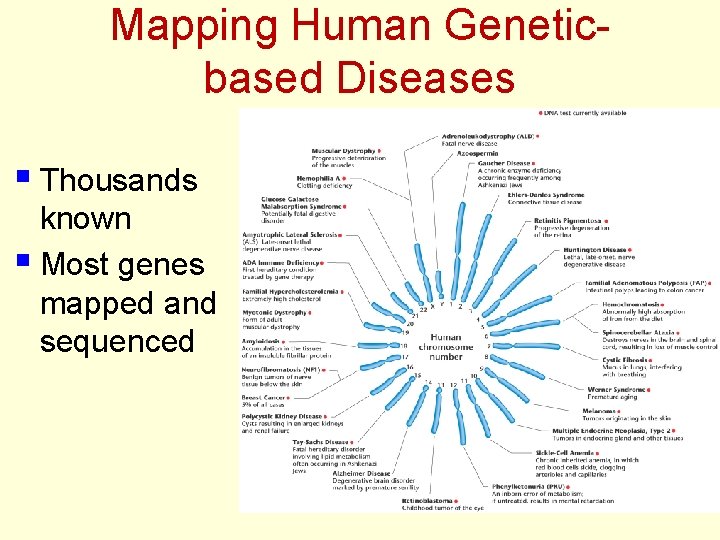
Mapping Human Geneticbased Diseases § Thousands known § Most genes mapped and sequenced
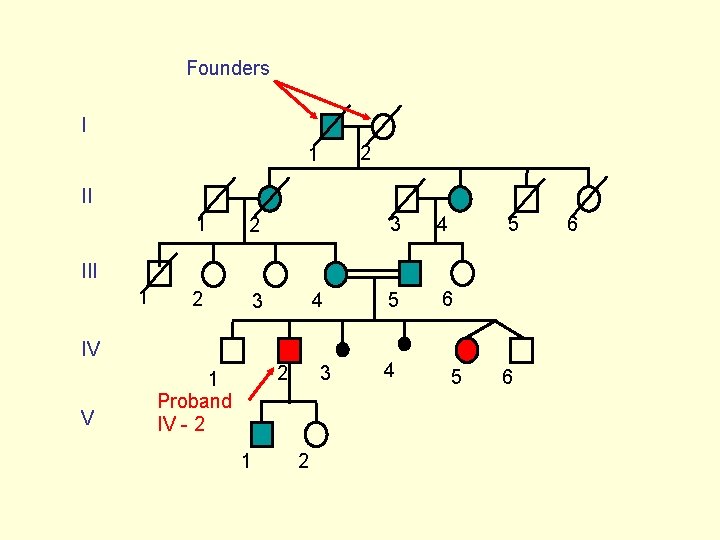
Founders I 2 1 II 1 2 2 3 3 4 5 5 6 III 1 4 IV V 2 1 Proband IV - 2 1 3 2 4 5 6 6
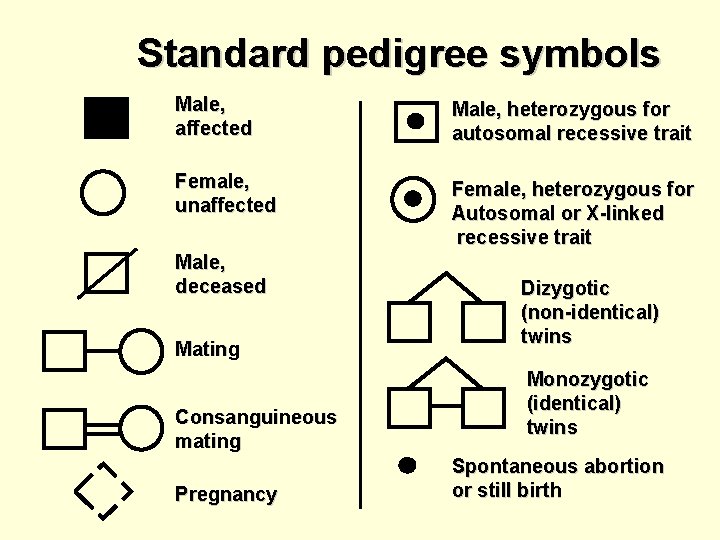
Standard pedigree symbols Male, affected Male, heterozygous for autosomal recessive trait Female, unaffected Female, heterozygous for Autosomal or X-linked recessive trait Male, deceased Mating Consanguineous mating Pregnancy Dizygotic (non-identical) twins Monozygotic (identical) twins Spontaneous abortion or still birth
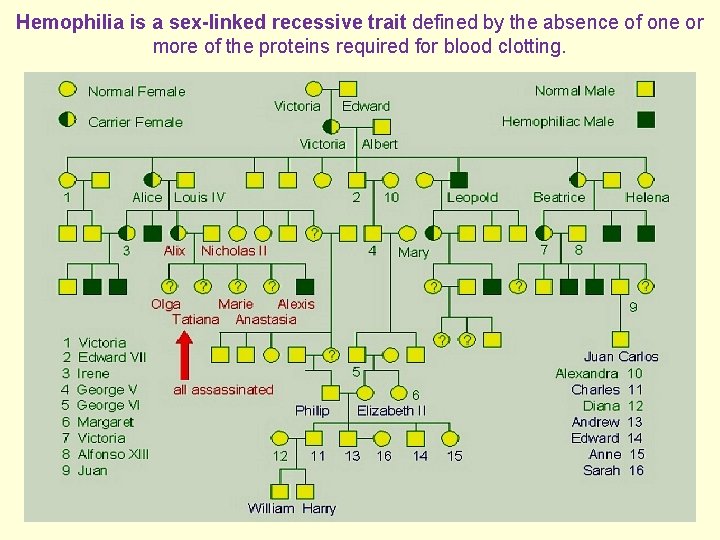
Hemophilia is a sex-linked recessive trait defined by the absence of one or more of the proteins required for blood clotting.

Non-Traditional Inheritence Ø Mitochondrial genes Ø Trinucleotide repeats Ø Genetic imprinting

Mitochondrial Inheritance • Matrilineal mode of inheritance: only mother • • passes mitochondrial DNA to offspring Higher spontaneous mutations than nuclear DNA affects both males and females , but transmitted only through females range of phenotypic severity due to heteroplasmy Example: diabetes mellitus with sensorineuronal deafness

Mitochondrial inheritance § § § This type of inheritance applies to genes in mitochondrial DNA. Mitochondrial disorders can appear in every generation of a family and can affect both males and females, but fathers do not pass mitochondrial traits to their children. E. g. Leber's hereditary optic neuropathy (LHON)

What are Genetic Variations? • Variations are simply differences in genetic • sequence Variation can be seen at every genetic level: § § § In the DNA In the genes In the chromosomes In the proteins In the function of proteins

Technology Advancement

Technological Advances ENIAC i. Pad 1946 2012

Genome Sequencing Technology Applied Biosystems 3730 DNA Analyzers Oxford Nanopore Min. ION 2002 2012

New Technologies

REFERNCES 1. MEDICAL GENETICS Jorde, Carey, Bamshad, White Published by: Mosby 2. ELEMENTS OF MEDICAL GENETICS Robert Muller and Ian Young Published by: Churchill Livingstone 3. ESSENTIAL MEDICAL GENETICS Connor, Ferguson-Smith Published: Blackwell Science

Journals • Nature Genetics – http: //www. nature. com/ng/index. html • Nature Reviews Genetics – http: //www. nature. com/nrg/index. html • Trends in Genetics – http: //www. trends. com/tig/default. htm
- Slides: 56