Medical Device Discovery Appraisal Program Onboarding Presentation with

Medical Device Discovery Appraisal Program Onboarding Presentation with PMO This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Agenda • The CMMI Model • This Program, the stakeholders, and you • What to expect next • • Program flow, required activities, and benefits Intake Call and Appraisal Schedule Appraisal Results After the Appraisal • Metrics Collection • How to stay informed and engaged • Legal and Accounting This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

What is CMMI®? The Capability Maturity Model Integration (CMMI®) is a capability improvement model that can be adapted to solve any performance issue at any level of the organization in any industry. The Model provides guidelines and recommendations for helping your organization diagnose problems and improve performance. Used by over 10, 000 organizations from more than 100 countries all over the world, CMMI helps you identify and achieve measurable business goals. See Case Studies. This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

What is this program? This pilot program leverages the CMMI framework as the standard maturity model by which medical device organizations may measure their capability to produce high quality devices and increase patient safety. FDA will adjust their engagement activities and submission requirements as a recognition of this independent assessment of quality maturity. The CDRH Case for Quality Voluntary Improvement Program (Cf. Q VIP) pilot was announced in the Federal Register on December 28, 2017. Reduced defects / rework Reduced costs Accelerated time to market Increased Customer Satisfaction A culture of quality - across the organization. This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Case for Quality Voluntary Improvement Program 2011: FDA Study- Dr. Jeff Shuren: CDRH Director Discusses Case for Quality 2014: MDIC joins the Case for Quality: Overview of the Case for Quality 2015: Deloitte Study: CMMI Selection 2016: Proof of Concept Pilots: Outcomes 2017: Early Adopters: Edwards Lifesciences, Boston Scientific 2018: Testimonies: Medtronic & Baxter, Celo. Nova and STERIS This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
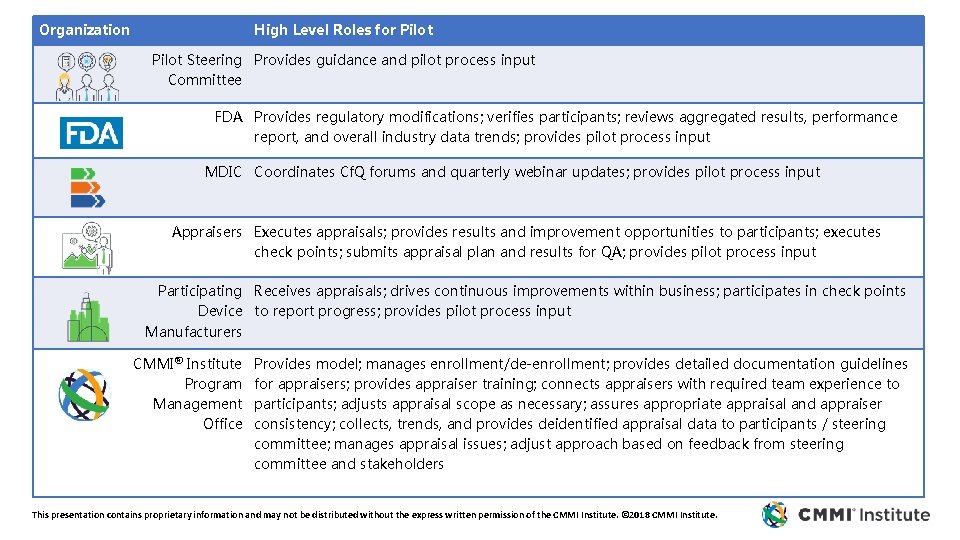
Organization High Level Roles for Pilot Steering Provides guidance and pilot process input Committee FDA Provides regulatory modifications; verifies participants; reviews aggregated results, performance report, and overall industry data trends; provides pilot process input MDIC Coordinates Cf. Q forums and quarterly webinar updates; provides pilot process input Appraisers Executes appraisals; provides results and improvement opportunities to participants; executes check points; submits appraisal plan and results for QA; provides pilot process input Participating Receives appraisals; drives continuous improvements within business; participates in check points Device to report progress; provides pilot process input Manufacturers CMMI® Institute Program Management Office Provides model; manages enrollment/de-enrollment; provides detailed documentation guidelines for appraisers; provides appraiser training; connects appraisers with required team experience to participants; adjusts appraisal scope as necessary; assures appropriate appraisal and appraiser consistency; collects, trends, and provides deidentified appraisal data to participants / steering committee; manages appraisal issues; adjust approach based on feedback from steering committee and stakeholders This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
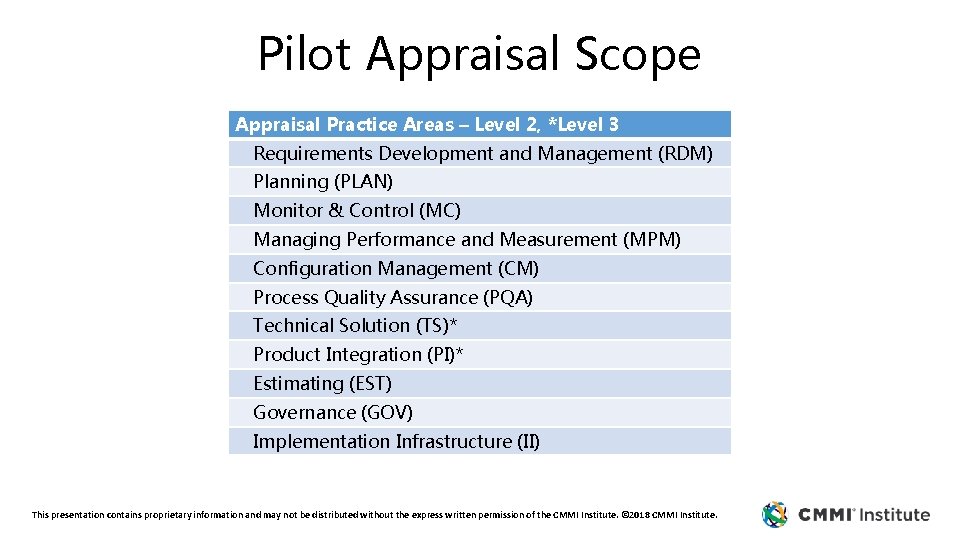
Pilot Appraisal Scope Appraisal Practice Areas – Level 2, *Level 3 Requirements Development and Management (RDM) Planning (PLAN) Monitor & Control (MC) Managing Performance and Measurement (MPM) Configuration Management (CM) Process Quality Assurance (PQA) Technical Solution (TS)* Product Integration (PI)* Estimating (EST) Governance (GOV) Implementation Infrastructure (II) This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
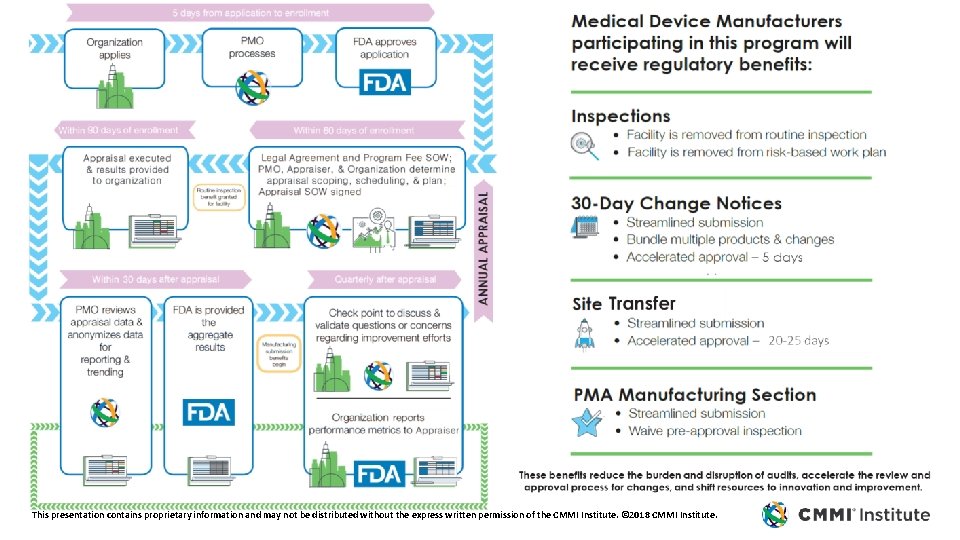
This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
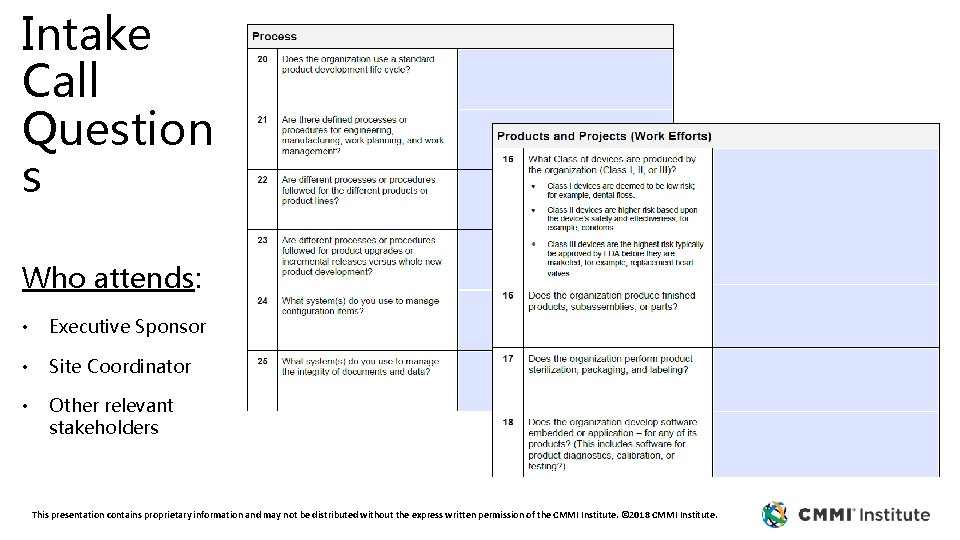
Intake Call Question s Who attends: • Executive Sponsor • Site Coordinator • Other relevant stakeholders This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Example Schedule 9: 30 – 10: 30 Facility Tour This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
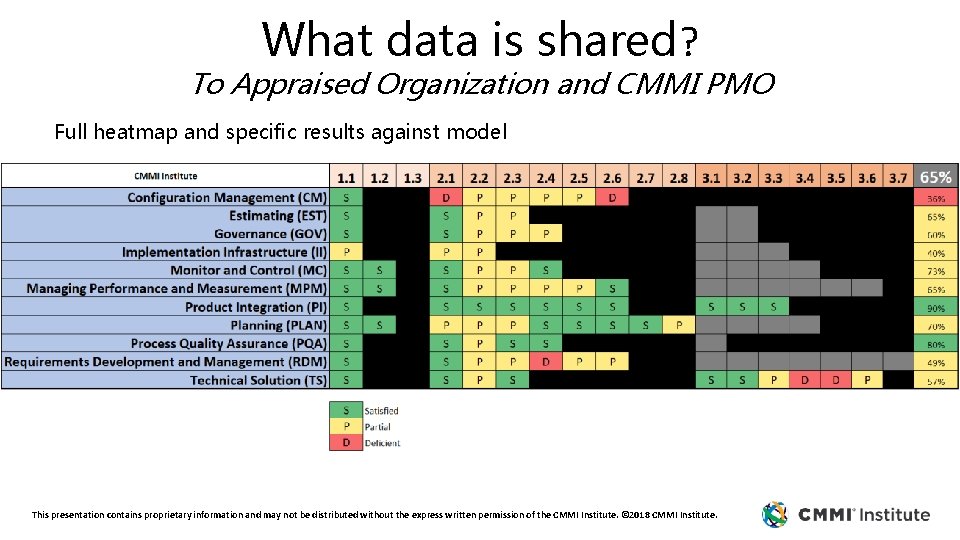
What data is shared? To Appraised Organization and CMMI PMO Full heatmap and specific results against model This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

What data is shared? To FDA Practice area results and overall score for an organization This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

The Check Point • The goals of the check point process are to: 1. encourage continuous process improvement, and 2. encourage engagement in the program. • Check points with the CMMI Lead Appraiser occur every quarter after appraisal is completed. • Check points occur on the phone/digitally, approximately 1 -2 hour conversation. This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Performance Metrics a check point activity • The goals of the Performance Report: 1. Understand how organizations measure product quality so FDA and industry can work together to create solutions 2. Determine if our hypothesis is correct, that improvements to your system will lead to improvements to your products 3. Long-Term: Measure your performance against industry • Aligned with Analytics Outcomes Cf. Q initiative: product safety, effectiveness, reliability, availability • Begin discussion in 1 st check point, draft metrics in 2 nd check point This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
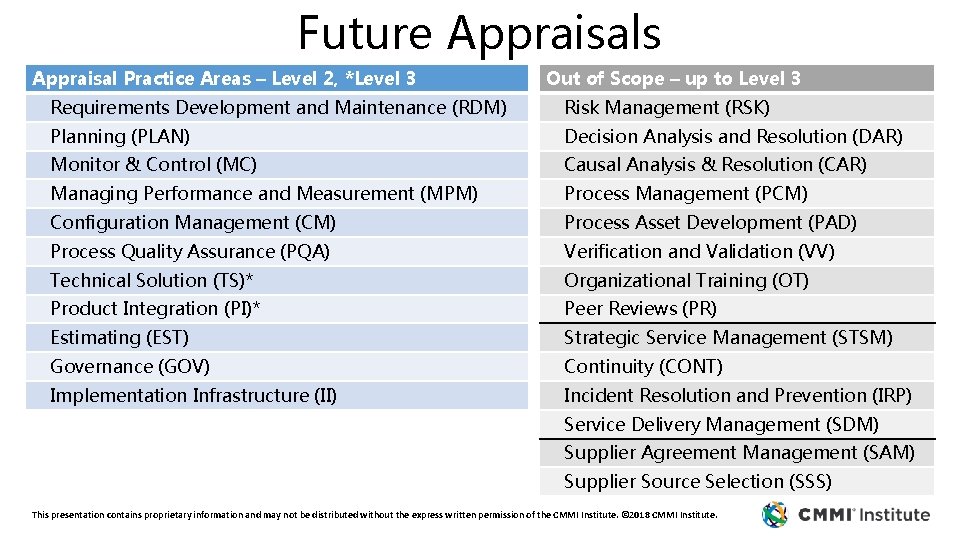
Future Appraisals Appraisal Practice Areas – Level 2, *Level 3 Out of Scope – up to Level 3 Requirements Development and Maintenance (RDM) Risk Management (RSK) Planning (PLAN) Decision Analysis and Resolution (DAR) Monitor & Control (MC) Causal Analysis & Resolution (CAR) Managing Performance and Measurement (MPM) Process Management (PCM) Configuration Management (CM) Process Asset Development (PAD) Process Quality Assurance (PQA) Verification and Validation (VV) Technical Solution (TS)* Organizational Training (OT) Product Integration (PI)* Peer Reviews (PR) Estimating (EST) Strategic Service Management (STSM) Governance (GOV) Continuity (CONT) Implementation Infrastructure (II) Incident Resolution and Prevention (IRP) Service Delivery Management (SDM) Supplier Agreement Management (SAM) Supplier Source Selection (SSS) This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

How this is different than an FDA inspection MDDAP • Focus is on actual capabilities and activities of value add of the organization • Interviews / data collection from people who perform work which provides an atmosphere for actual inspection to improve from • Drives a conversation of how to actually improve in a way that makes sense to the business The Difference • Looks beyond the CFR, and not just strict compliance • Interviews / data collection are not from just those in the “front room” or those who manage audits • Does not focus on just a corrective action list to “get into compliance” This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Stay Informed and Engaged! MDIC Forums and Webinars Who: Medical device organizations and healthcare groups interested in Case for Quality and VIP, and FDA, hosted by MDIC When: Quarterly, 1 day forums, 1 hour webinars What: FDA and CMMI Institute share program stats, lessons learned, new pursuits or features, and an industry participant to share their program experience This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Stay Informed and Engaged! Participant Updates Meeting Who: All organizations that are enrolled in the program, FDA, and MDIC, hosted by CMMI Institute When: Monthly, 1 hour What: Discuss state of program, what is next, specific data only for participating orgs (e. g. specific trends), collect feedback, make community connections This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Stay Informed and Engaged! Working Groups Additional Regulatory Benefits Objective: To identify, develop, test, and finalize any additional regulatory benefits in consideration for participants of the Program. Performance Measures Objective: To improve data transparency by identifying additional information needs and outcomes, considering improvement opportunities to the methodology, and discussing potential synergies for continuous monitoring, in order to reduce reappraisal scope and/or increase the length of time to reappraisal. Reappraisals Objective: To define and develop the standards and exceptions for conducting reappraisals. Multi-Site Appraisals Objective: To define and develop the standards and exceptions for conducting multi-site appraisals. Program Features Objective: To identify, develop, test, and finalize new desired features of the Program, as well as identify, analyze, and resolve any undesirable features of the Program. Medical Device Context Objective: To define, build, and formally develop the additional CMMI model context to support the intended tailoring for the medical device industry. EU Agencies Objective: To explore and identify approaches to share the value and success of the Program with EU Agencies, EU medical device groups and associations, and EU device makers. This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Legal and Accounting • Agreement: Required Terms • Program Fee: What this supports, $5, 000* • Appraisal Fee: Cost expectations, $50, 000* • Invoicing: POs & accounting@cmmiinstitute. com *Pricing varies depending on scope. Reduced pricing may be available to small organizations. This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.

Questions? This presentation contains proprietary information and may not be distributed without the express written permission of the CMMI Institute. © 2018 CMMI Institute.
- Slides: 21