Med Chem Drugs for Diabetes Mellitus DM FPG

Med. Chem: Drugs for Diabetes Mellitus (DM) FPG 12 th Ed. W & G page 666 -704 6 th Ed. Foye’s Page 855 -876 Shengquan Liu, Ph. D. 11/15/18, Th, PM 1 1 File name: DM_181115 p. Th
![Glucagon is a catabolic (energy releasing) hormone ↑ [BG] Insulin is a anabolic (energy Glucagon is a catabolic (energy releasing) hormone ↑ [BG] Insulin is a anabolic (energy](http://slidetodoc.com/presentation_image/261aa31610df2f82a7087ba7ef01cf2e/image-2.jpg)
Glucagon is a catabolic (energy releasing) hormone ↑ [BG] Insulin is a anabolic (energy storing) hormone ↓ [BG]

Drugs Used in DM & Their MOA: Outline SEs ↑ insulin supplies or actions ↓ [BG] 1. 8 Insulins: differ in onset & Do. A 1. 2. 3. Rapid, short (lispro, aspart, glulisine, regular) Intermediate (NPH, lente) Long (glargine, detemir, degludec) Parenteral ↑ insulin supplies Tx: T 1 & 2 D 2. Pramlintide (-) glucagon secretion (Tx: T 1 & 2 D) 3. Exenatide (-) GLP-1 ®; sitagliptin (-) DPP-4 (PO): 4. Treatment of T 2 D (PO) 1. 2. 3. – 1. Sulfonylureas* & glinides Thiazolidinediones (TZDs) Biguanides (metformin*) a-Glucosidase inhibitor SLUT-2 inhibitors ↓ [BG]/ ↑ wt. ↔ [BG]/Wt. (-) KATP ↑ insulin release/↓[BG]/↑wt. (+) PPAR-g ® in adipocytes ↑ insulin action (+) AMPPK in liver ↓ glucose absorption from gut ↔[BG]/Wt. (-) reabsorption of glu. in kidney 5. Hyperglycemic glucagon ↑ c. AMP ↑ [BG] 3
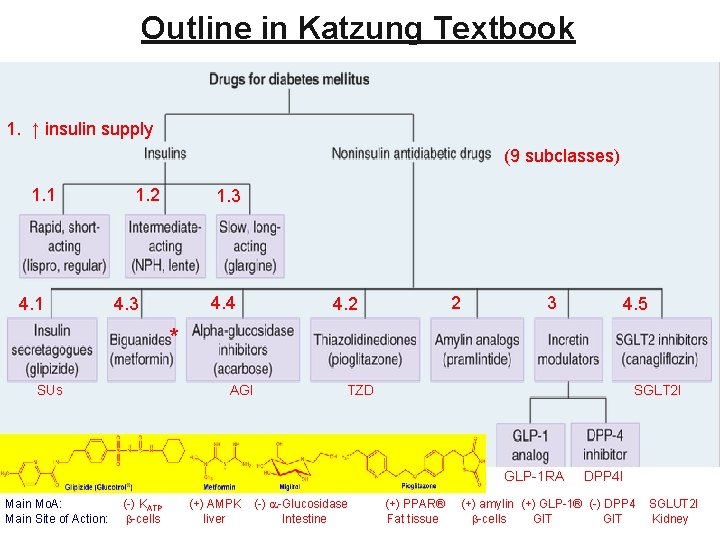
Outline in Katzung Textbook 1. ↑ insulin supply (9 subclasses) 1. 1 4. 1 1. 2 1. 3 4. 4 4. 3 2 4. 2 3 4. 5 * SUs AGI TZD SGLT 2 I GLP-1 RA Main Mo. A: Main Site of Action: (-) KATP b-cells (+) AMPK liver (-) a-Glucosidase Intestine (+) PPAR® Fat tissue DPP 4 I (+) amylin (+) GLP-1® (-) DPP 4 b-cells GIT SGLUT 2 I Kidney

Learning Objectives: To Know Chemical basis of T 1 & 2 D Route of administration (PO or SC(Q)/IM/IV) Drug classification based on structure & Mo. A Advantages and disadvantages of each class 1. Insulins, protein or peptide drugs SC(Q)/IM/IV 1. Mo. A: indications/SEs 2. SAR PD; SP (structure and physical property) R PK l 2. Structure onset/Do. A Oral antidiabetics (PD(K): ADME) l Identify drug classes by structure, chemical classification, Mo. A, Efficacy/SEs, SAR, PK (e. g. F) & onset/Do. A 1. 2. Insulin secretagogues, metformin Thiazolidinediones, a-Glucosidase inhibitors 5

← Starch Sugars Glu Glycogen (Sucrose) Glu In Plants 1 2 3 4 5 b Excess Glu is stored as glycogen in liver & muscle in Animal 6 6
![Healthy Condition: Balanced Blood Gluose [BG] • After Meal 1. ↑ [BG] ↑ [ATP] Healthy Condition: Balanced Blood Gluose [BG] • After Meal 1. ↑ [BG] ↑ [ATP]](http://slidetodoc.com/presentation_image/261aa31610df2f82a7087ba7ef01cf2e/image-7.jpg)
Healthy Condition: Balanced Blood Gluose [BG] • After Meal 1. ↑ [BG] ↑ [ATP] (-) KATP channel ↑ [Ca 2+]in ↑ Insulin secretion from b-cells 2. Insulin in blood (+) insulin® in tissues ↑ GLUT* synthesis & translocation ↑ Glu uptake 3. Glu is transported into 1. muscle cells ↑ E 2. liver cells storage 3. fat cells 4. ↑ Glu uptake in tissues 5. ↓ [BG] normal [BG] • • Glucagon ↑ [BG] How does insulin to it? Target cells in tissues 7
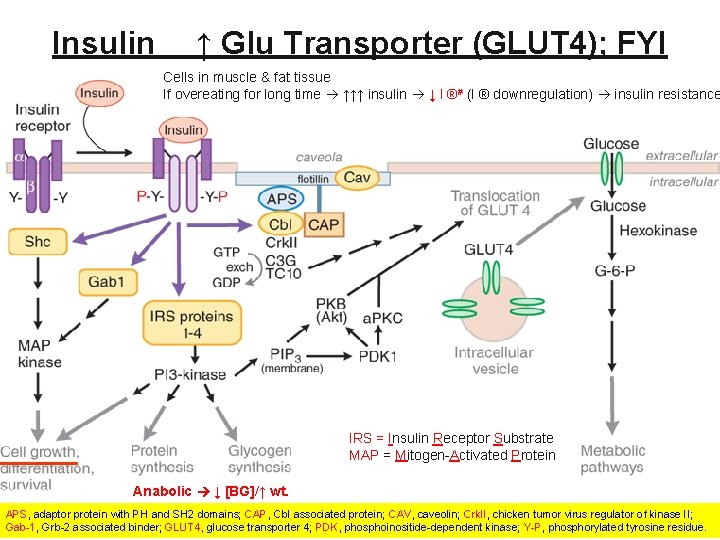
Insulin ↑ Glu Transporter (GLUT 4); FYI Cells in muscle & fat tissue If overeating for long time ↑↑↑ insulin ↓ I ®# (I ® downregulation) insulin resistance IRS = Insulin Receptor Substrate MAP = Mitogen-Activated Protein Anabolic ↓ [BG]/↑ wt. APS, adaptor protein with PH and SH 2 domains; CAP, Cbl associated protein; CAV, caveolin; Crk. II, chicken tumor virus regulator of kinase II; Gab-1, Grb-2 associated binder; GLUT 4, glucose transporter 4; PDK, phosphoinositide-dependent kinase; Y-P, phosphorylated tyrosine residue. 8

T 1 D: Glu Can Not Enter Cells Passively BG can NOT cross cell membranes passively BG is delivered to fat, muscle, liver & other cells only by Glu transporters (GLUT 1 -5) • • – GLUT-4 is the most important & relevant to insulin Inserted into the muscle & adipose cell membranes • In muscle/adipose cells: insulin ↑ GLUT 4 cells • – – – take up & use Glu provide energy to cells convert any excess to storage form (glycogen/protein/fat) • • Glu glycogen in liver and muscle Glu amino acids, lactates or proteins in liver and muscle In adipose cells: E is stored as triglycerides (TG) • – Glu TG (fat) stored there 9
![↓ Insulin ↑ [BG] Polyuria/dipsia/phagia However, Glu is not transported across cell membranes & ↓ Insulin ↑ [BG] Polyuria/dipsia/phagia However, Glu is not transported across cell membranes &](http://slidetodoc.com/presentation_image/261aa31610df2f82a7087ba7ef01cf2e/image-10.jpg)
↓ Insulin ↑ [BG] Polyuria/dipsia/phagia However, Glu is not transported across cell membranes & is unable to enter cells w/o insulin • – – These cells will undergo “starvation in midst of plenty” You eat, but polyphagia (hunger)/fatigue/↓ wt. ↑ [BG] ↑ [urine Glu] glucosuria/polyuria/↓ wt. polydipsia(thirst) ……… “ 3 Ps” T 1 D Polyuria Polyphagia Polydipsia Glucosuria 10
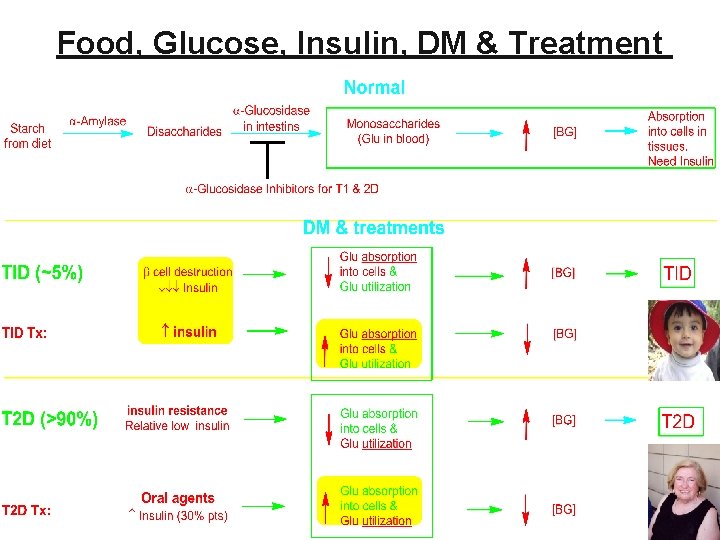
Food, Glucose, Insulin, DM & Treatment 11

Treatment of DM: ↑ Insulin Supply +/or Action The most common pancreatic disease requiring pharmacologic therapy is DM • – ↓ insulin production +/or its action Tx: ↑ insulin production +/or its action – T 1 D: Treated with 17 formulations of insulin administered parenterally (mostly sc) – T 2 D: 7 types of oral & injectable antidiabetic agents Glucagon can be used to treat severe hypoglycemia in DM, b/c • – If ↓ [BG] ↑ glucagon release ↑ (glycogen glucose) ↑ [BG] 12

1. Insulin Preparations (Mainly For T 1 D) Human insulins are prepared by • 1. 2. Enzymatic conversion of the terminal amino acid of beef/pork insulin (Novolin) or Bacterial recombinant DNA technology (Humulin = regular insulin) A protein rapid degradation in GIT • – – t 1/2 = only a few min. in circulation orally inactive Administered parenterally (SC, IM or IV) – Typically SC injection • Many preparations are formulated w/ different onset & Do. A • 5 rates of onset & DOA 1. 2. 3. 4. 5. Rapid-acting: Short-acting: Intermediate-acting: Long-acting: Mixed insulins lispro, aspart, glulisine (sc. postprandial) regular insulin (sc, iv) Neutral Protamine Hagedorn (NPH) detemir, glargine, degludec (Do. A 20 h) mimic endogenous insulin secretion 13
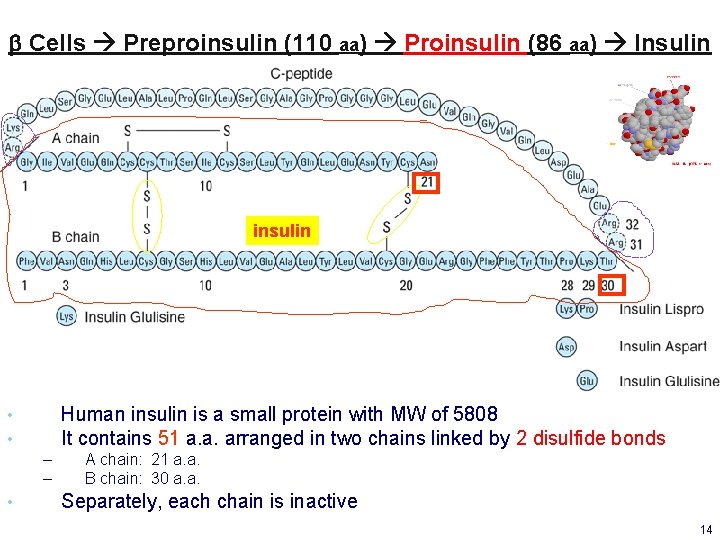
b Cells Preproinsulin (110 aa) Proinsulin (86 aa) Insulin insulin Human insulin is a small protein with MW of 5808 It contains 51 a. a. arranged in two chains linked by 2 disulfide bonds • • – – • A chain: 21 a. a. B chain: 30 a. a. Separately, each chain is inactive 14
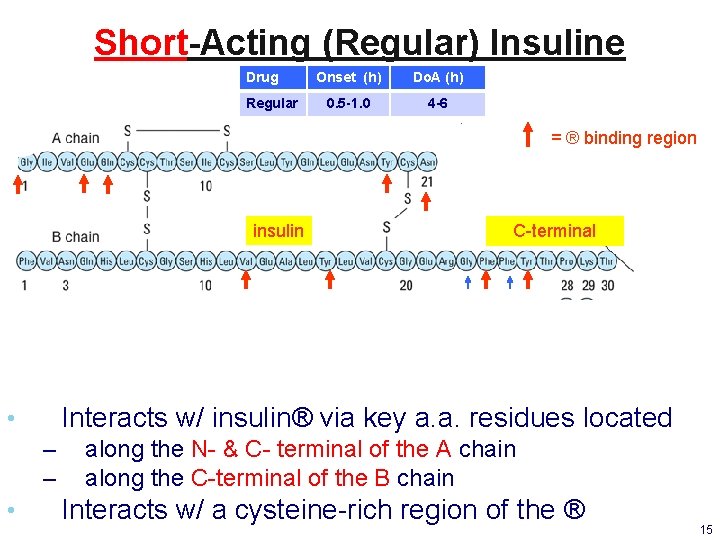
Short-Acting (Regular) Insuline Drug Regular Onset (h) Do. A (h) 0. 5 -1. 0 4 -6 = ® binding region insulin Interacts w/ insulin® via key a. a. residues located • – – • C-terminal along the N- & C- terminal of the A chain along the C-terminal of the B chain Interacts w/ a cysteine-rich region of the ® 15

Water Soluble Insulin Monomers Have Fast-Onset 1. ↑ Water solubility (vs. solid) ↑ SC absorption rate 1. 2. Faster onset of action Short Do. A Only insulin monomer is able to (+) insulin ® 1. 2. 3. Monomer is the active form t 1/2 = 5 -6 min. Zn 2+ ↓ monomer formation Regular insulin* associates as inactive hexamers – Dissociation of hexamers & dimers to monomers is the rate limitingstep for absorption rate – 4. Administration 30 -45 min. before a meal C-terminal region of the B-chain is important for dimer or hexamer formation – – ↑ dimer or hexamer formation ↓ dimer or hexamer formation slow/long-acting rapid/short-acting 16
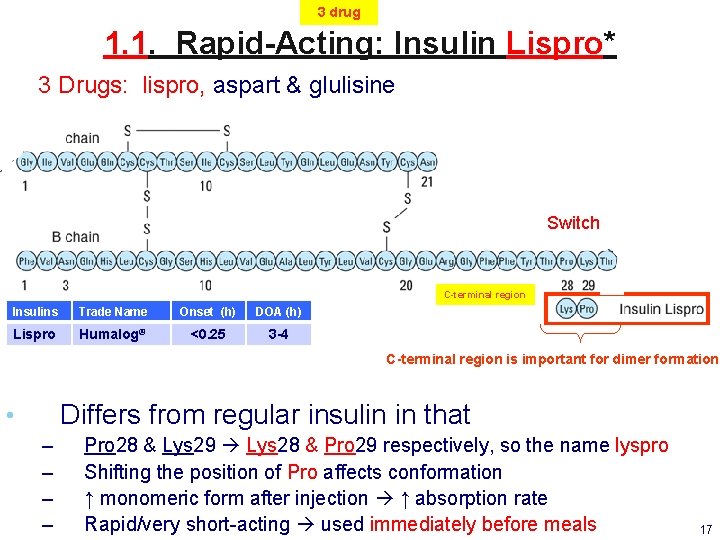
3 drug 1. 1. Rapid-Acting: Insulin Lispro* 3 Drugs: lispro, aspart & glulisine Switch C-terminal region Insulins Trade Name Onset (h) DOA (h) Lispro Humalog® <0. 25 3 -4 C-terminal region is important for dimer formation Differs from regular insulin in that • – – Pro 28 & Lys 29 Lys 28 & Pro 29 respectively, so the name lyspro Shifting the position of Pro affects conformation ↑ monomeric form after injection ↑ absorption rate Rapid/very short-acting used immediately before meals 17
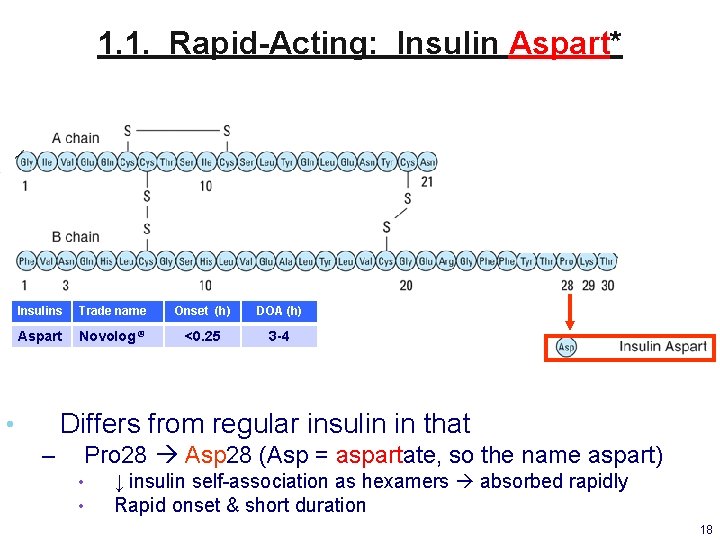
1. 1. Rapid-Acting: Insulin Aspart* Insulins Trade name Onset (h) DOA (h) Aspart Novolog ® <0. 25 3 -4 Differs from regular insulin in that • – Pro 28 Asp 28 (Asp = aspartate, so the name aspart) • • ↓ insulin self-association as hexamers absorbed rapidly Rapid onset & short duration 18
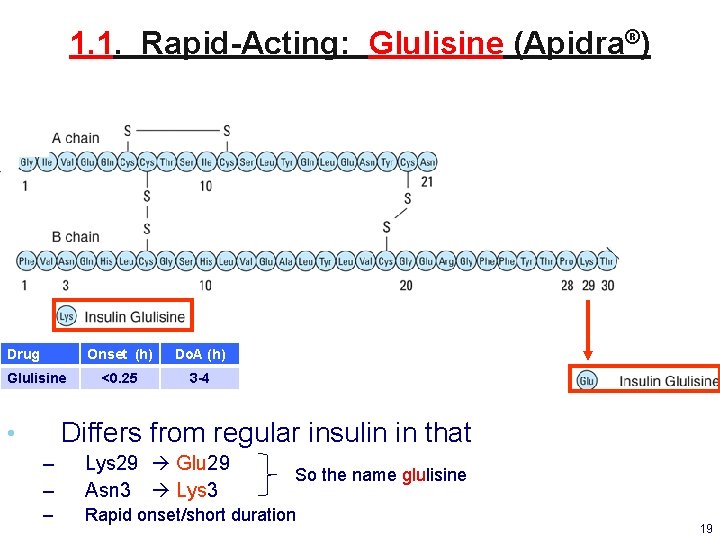
1. 1. Rapid-Acting: Glulisine (Apidra®) Drug Glulisine Onset (h) Do. A (h) <0. 25 3 -4 Differs from regular insulin in that • – – Lys 29 Glu 29 Asn 3 Lys 3 – Rapid onset/short duration So the name glulisine 19

1 drug 1. 2. Intermediate-Acting Insulins (NPH*) Onset (h) Do. A (h) Peak Appearance NPH 1 -4 10 -16 Peakless Cloudy or whitish solution SC only Neutral Protamine Hagedorn (NPH; insulin Isophane) • – – – • Drug A suspension of regular insulin complexed w/ zinc & protamine in a phosphate buffer cloudy* ↓ water solubility & absorption ↓ absorption rate Insulin dissolve more gradually after s. c. slow onset Proteolytic enzymes cleave the protamine from insulin slowly to permit absorption of insulin ↑ Do. A ↑ aggregation of insulin into dimers & hexamers (insulin dimers, hexamers & its complex is not absorbed inactive) Combination products of lispro, aspart insulin & their respective protamine complexes also available 20
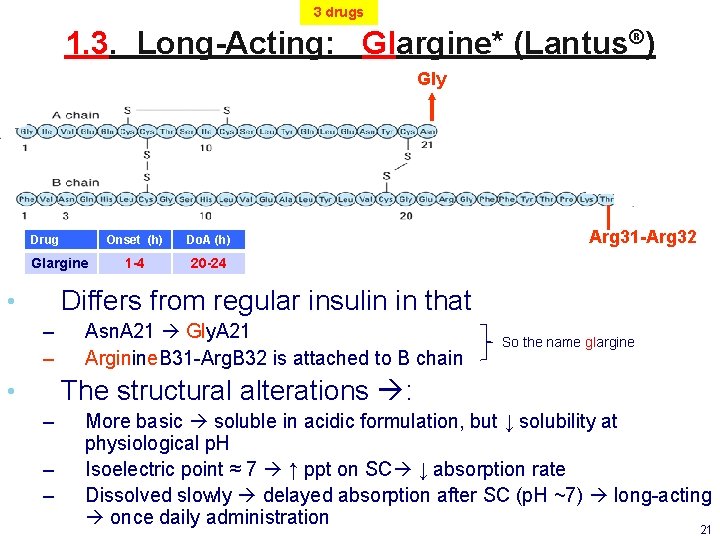
3 drugs 1. 3. Long-Acting: Glargine* (Lantus®) Gly Drug Glargine Onset (h) Do. A (h) 1 -4 20 -24 Arg 31 -Arg 32 Differs from regular insulin in that • – – Asn. A 21 Gly. A 21 Arginine. B 31 -Arg. B 32 is attached to B chain So the name glargine The structural alterations : • – – – More basic soluble in acidic formulation, but ↓ solubility at physiological p. H Isoelectric point ≈ 7 ↑ ppt on SC ↓ absorption rate Dissolved slowly delayed absorption after SC (p. H ~7) long-acting once daily administration 21
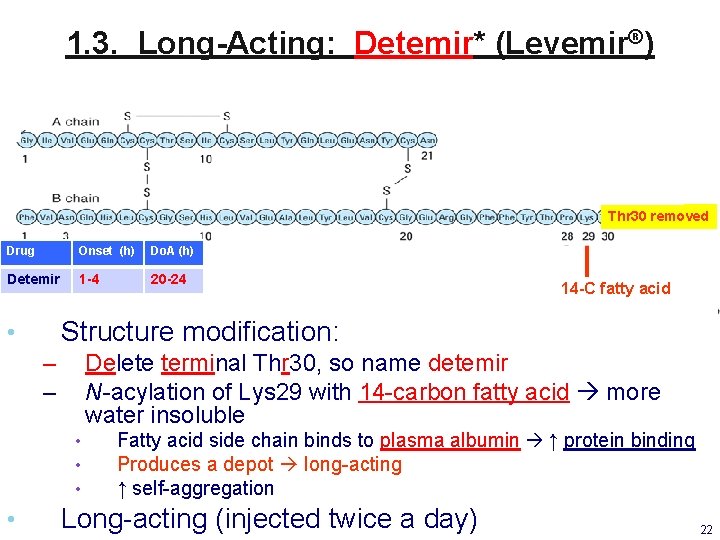
1. 3. Long-Acting: Detemir* (Levemir®) Thr 30 removed Drug Onset (h) Do. A (h) Detemir 1 -4 20 -24 Structure modification: • – – Delete terminal Thr 30, so name detemir N-acylation of Lys 29 with 14 -carbon fatty acid more water insoluble • • 14 -C fatty acid Fatty acid side chain binds to plasma albumin ↑ protein binding Produces a depot long-acting ↑ self-aggregation Long-acting (injected twice a day) 22
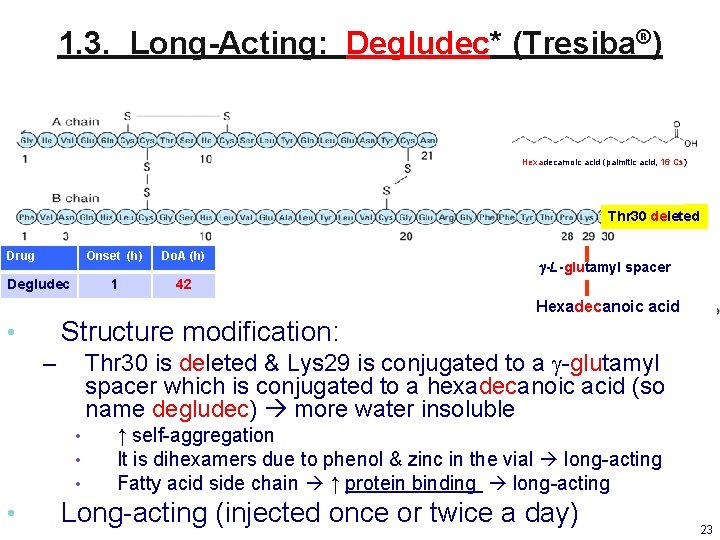
1. 3. Long-Acting: Degludec* (Tresiba®) Hexadecamoic acid (palmitic acid, 16 Cs) Thr 30 deleted Drug Degludec Onset (h) Do. A (h) 1 42 g-L-glutamyl spacer Hexadecanoic acid Structure modification: • Thr 30 is deleted & Lys 29 is conjugated to a g-glutamyl spacer which is conjugated to a hexadecanoic acid (so name degludec) more water insoluble – • • ↑ self-aggregation It is dihexamers due to phenol & zinc in the vial long-acting Fatty acid side chain ↑ protein binding long-acting Long-acting (injected once or twice a day) 23
29 (14 C acid)
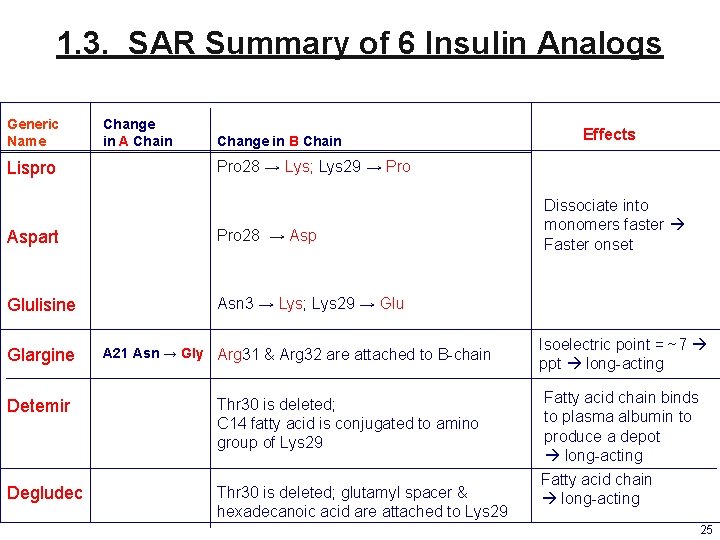
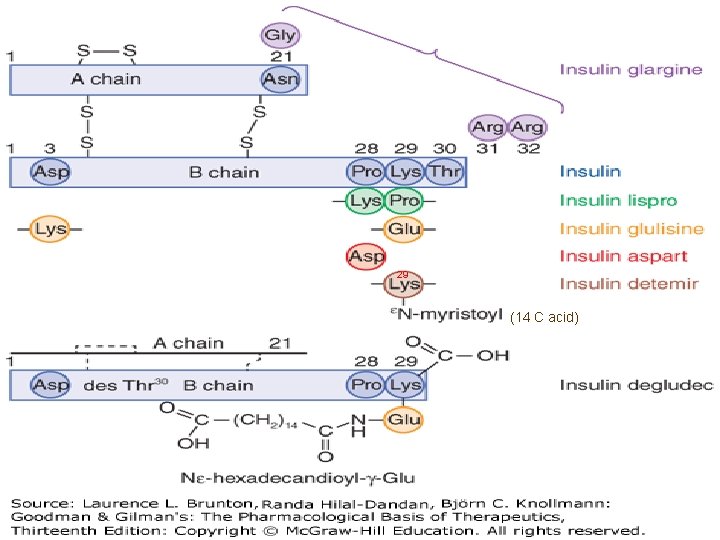
1. 3. SAR Summary of 6 Insulin Analogs Generic Name Lispro Change in A Chain Change in B Chain Pro 28 → Lys; Lys 29 → Pro Aspart Pro 28 → Asp Glulisine Asn 3 → Lys; Lys 29 → Glu Glargine Effects A 21 Asn → Gly Arg 31 & Arg 32 are attached to B-chain Detemir Thr 30 is deleted; C 14 fatty acid is conjugated to amino group of Lys 29 Degludec Thr 30 is deleted; glutamyl spacer & hexadecanoic acid are attached to Lys 29 Dissociate into monomers faster Faster onset Isoelectric point = ~7 ppt long-acting Fatty acid chain binds to plasma albumin to produce a depot long-acting Fatty acid chain long-acting 25
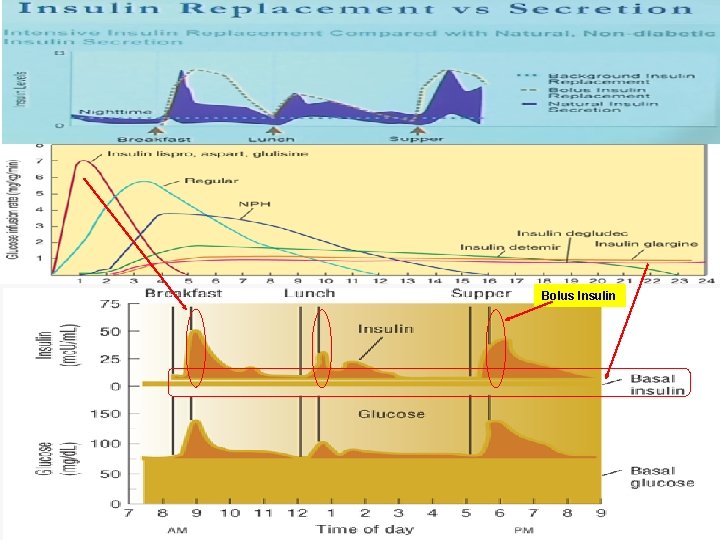
Bolus Insulin
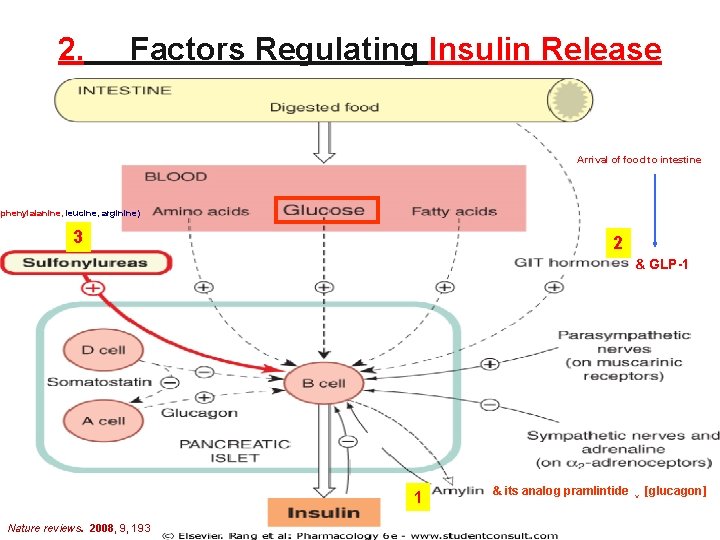
2. Factors Regulating Insulin Release Arrival of food to intestine (phenylalanine, leucine, arginine) 3 2 & GLP-1 1 Nature reviews. 2008, 9, 193 & its analog pramlintide ↓ [glucagon] 27

2. Pramlintide* (An Amylin Agonist, FYI) 37 a. a. Injectable (rarely used) Short Do. A GLP-1 ® agonist is better than pramlintide for T 2 D Native amylin: produced in pancreatic b cell & secreted with insulin (+) amylin® in the brain ↓ satiety • (+) amylin® in a cells ↓ glucagon secretion ↓ [BG] Delays gastric emptying/GIT Glu absorption & ↓ appetite/wt. Unsuitable as a drug b/c – – – • – Aggregates (pancreatic amyloid) & insoluble in aqueous solution ↓abs. Modification of amylin • • • Ala 25 Ser 28 Ser 29 Pro = pramlintide (soluble) ↑ SC bioavailability Pramlintide: proline amylin peptide for T 1 & T 2 D (rarely used) • – – ↓ self-aggregation & ↑ water solubility Adjunct w/ insulin, SC before meals, t 1/2 50 min. renal elimination 28

Nature reviews. 2008, 9, 193 3. GLP-1 Agonists ↑ Insulin Secretion Glucagon-like peptide-1 (GLP-1): a mammalian intestine incretin hormone which secretes insulin after meals • – – – Consists of 2 peptides (30 & 31 a. a. ) Secreted by L cells in small intestine in response to food intake (+) GLP-1®s in b cells 1. 2. 3. 4. – Unsuitable for drugs b/c • • • ↑ 1 st-phase insulin release ↓ [BG] ↓ glucagon release ↓ gastric emptying ↑ satiety Dipeptidyl peptidase-4 (DPP-4) inactivates it by removing 2 a. a. t 1/2 = 1. 5 -2 min. DPP 4 inhibitors ↑ [GLP-1] 29
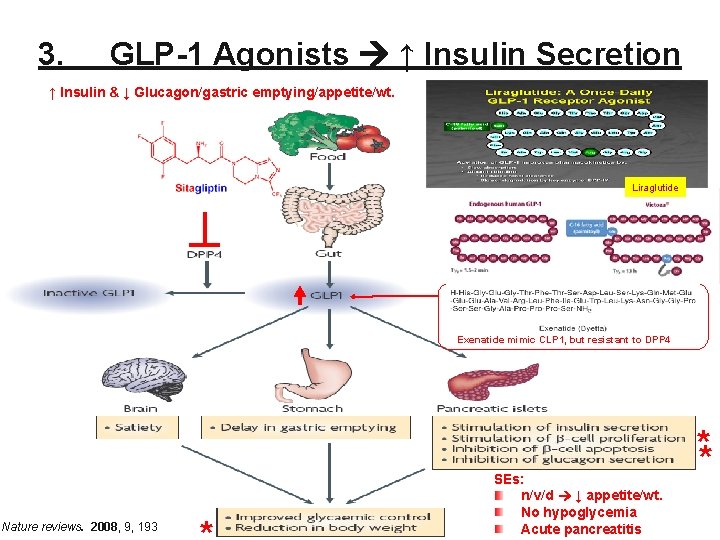
3. GLP-1 Agonists ↑ Insulin Secretion ↑ Insulin & ↓ Glucagon/gastric emptying/appetite/wt. Liraglutide Exenatide mimic CLP 1, but resistant to DPP 4 Nature reviews. 2008, 9, 193 SEs: n/v/d ↓ appetite/wt. No hypoglycemia Acute pancreatitis **

DPP-4 Inhibitors: 2 Molecular Mo. A No in US F = 85% T 1/2 = 12 h 31

3. • DPP 4 Inhibitor: Saxagliptin & Linagliptin Depeptidyl peptidase (DPP 4) is a serine protease – Primary protease responsible for degrading GLP-1 • • • Removes 2 a. a. from GLP-1 DPP-4 Inhibitors: sitagliptin (Januvia®), saxagliptin, linagliptin, alogliptin and vildagliptin (-) DPP 4 ↑ endogenous GLP-1 activity ↑ insulin secretion New class of oral agents Linagliptin (Tradjenta ®) 32

3. • – – – GLP-1 ® Agonists: Exenatide* (2005) Exendin-4 (DPP-4 - t 1/2 = 2 -3 h) 39 a. a. peptide from saliva of Gila monsters ↓ [BG] Mimics action of GLP-1 (+) GLP-1® in b cells Synthetic version of exendin-4 is: Exenatide (IM/IV) • – – Resistant to DPP-4 metabolism t 1/2 = 2 -4 h following SC (+) GLP-1 ® • • • – – • ↑ Insulin secretion in b cells & ↓ glucagon release in a cells ↓ Stomach emptying slow the rate of Glu entry into circulation ↑ New b cells production & ↓ b cells loss Tx: T 2 D that is not adequately controlled on metformin and/or a SU Peptide s. c. , no hepatic metabolism & excreted primary by kidney Similar drugs: liraglutide, albiglutide, dulaglutide 33

4. 1. 5 Groups of Oral Antidiabetic Drugs Insulin secretagogues (-) KATP (+) insulin secretion 1. 2. 1 st & 2 nd generations: sulfonylureas (SU, similar & for T 2 D) Non. SUs: meglitinides (“-glinides”) Thiazolidinediones (TZD, “-glitazone”) 1. 2. 3. Rosiglitazone Pioglitazone* Biguanides 1. Metformin** (widely used for T 2 D) The a-glucosidase inhibitors (adjunctive for T 2 D) 4. – 5. ↓ Glu absorption from intestine SGLUT 2 inhibitors (newer) – ↑ Glu reabsorption from kidney 34
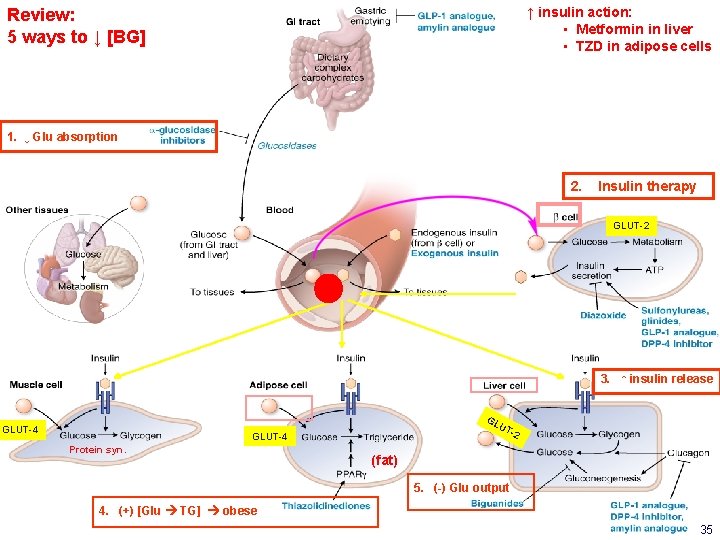
↑ insulin action: • Metformin in liver • TZD in adipose cells Review: 5 ways to ↓ [BG] 1. ↓ Glu absorption 2. Insulin therapy GLUT-2 3. ↑ insulin release GL GLUT-4 UT -2 GLUT-4 Protein syn. (fat) 5. (-) Glu output 4. (+) [Glu TG] obese 35
![4. 1. SFUs (-) KATP ↑ Insulin Release ↓ [BG] 1. After a meal 4. 1. SFUs (-) KATP ↑ Insulin Release ↓ [BG] 1. After a meal](http://slidetodoc.com/presentation_image/261aa31610df2f82a7087ba7ef01cf2e/image-36.jpg)
4. 1. SFUs (-) KATP ↑ Insulin Release ↓ [BG] 1. After a meal ATP 2 3 2 ↑ [Ca 2+]in 4 b agonists 5 b 2 -antagonists EPI/b 2 -agonists (+) M 3 ® ↑ Ca 2+ or ↑ c. AMP ↑ insulin release, EPAC 2 = Exchange protein activated by c. AMP, PKA = Protein kinase A ↑ Insulin release G&G 13 e

4. 1. Mo. A: SUs (-) KATP (+) Insulin Release (+) insulin release from b cells ↑ insulin level enough to overcome insulin resistance • 1. 2. 3. 4. ↑ [BG] ↑ [ATP] (-) KATP channel (+) insulin release SUs (-) sulfonylurea® (SUR 1) associated with KATP channel Closes KATP channels in b cell membrane (-) K+ efflux Depolarizes cell membrane 1. 2. (+) insulin release from functioning b cells of intact pancreas 5. ↓ hepatic clearance of insulin ↑ plasma insulin ↓ serum glucagon levels ↓ [BG] 1 st gen. SUs (rarely used) • • Tolbutamide, chlorpropamide, tolazamide 2 nd gen. SUs (mainstay for T 2 DM) • – More potent, less SEs more commonly used • • Opens voltage-dependent calcium channels Influx of calcium ↑ [Ca 2+]in Glyburide, Glipizide & Glimepiride Non. SUs (“glinides”): nateglinides & repaglinide 37
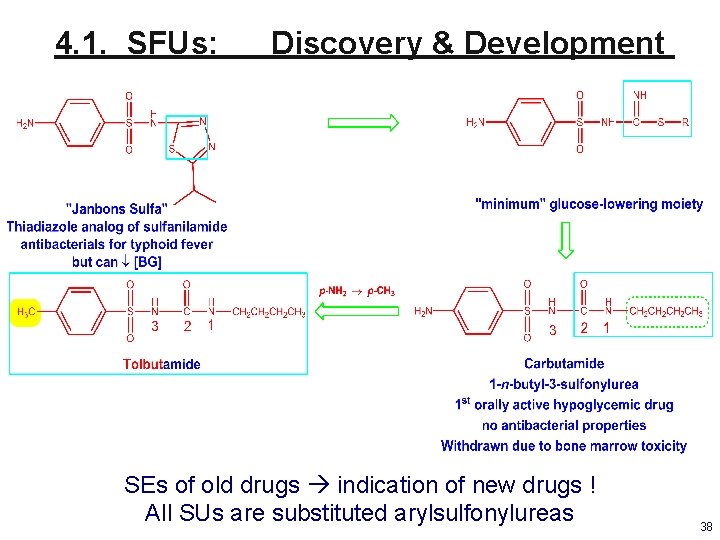
4. 1. SFUs: Discovery & Development SEs of old drugs indication of new drugs ! All SUs are substituted arylsulfonylureas 38
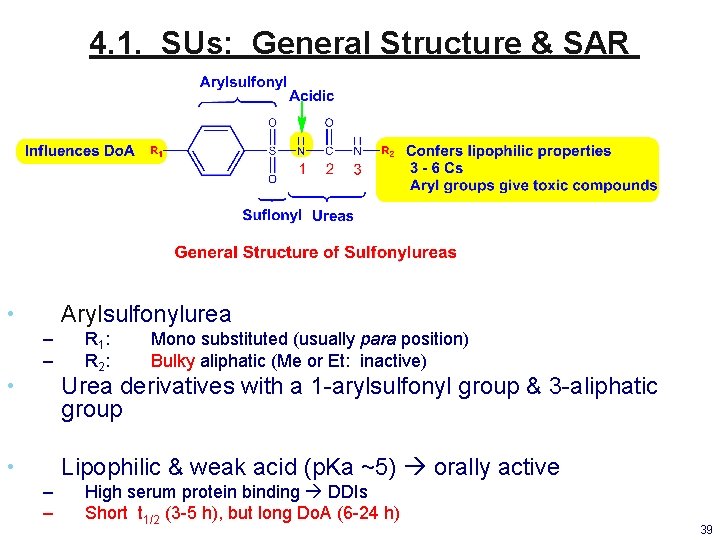
4. 1. SUs: General Structure & SAR Arylsulfonylurea • – – R 1 : R 2 : Mono substituted (usually para position) Bulky aliphatic (Me or Et: inactive) • Urea derivatives with a 1 -arylsulfonyl group & 3 -aliphatic group • Lipophilic & weak acid (p. Ka ~5) orally active – – High serum protein binding DDIs Short t 1/2 (3 -5 h), but long Do. A (6 -24 h) 39

4. 1. Chlorpropamide Very Long Do. A 40
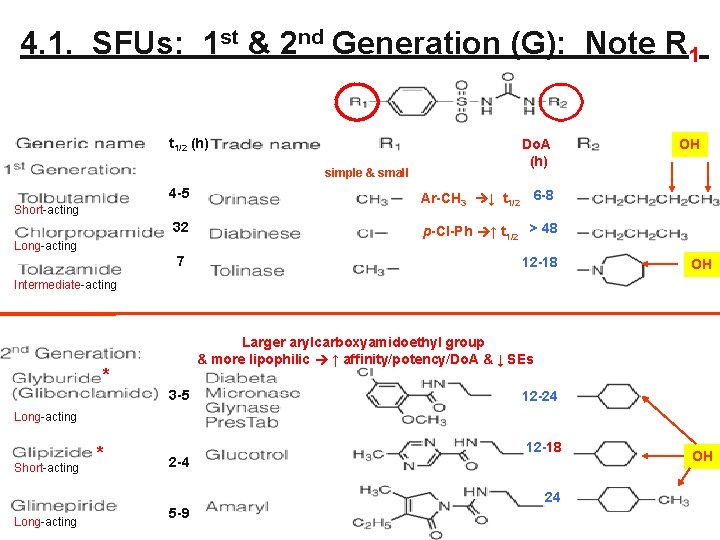
4. 1. SFUs: 1 st & 2 nd Generation (G): Note R 1 t 1/2 (h) simple & small Do. A (h) 4 -5 Ar-CH 3 ↓ t 1/2 6 -8 32 p-Cl-Ph ↑ t 1/2 > 48 7 12 -18 Short-acting Long-acting OH OH Intermediate-acting No in US Larger arylcarboxyamidoethyl group & more lipophilic ↑ affinity/potency/Do. A & ↓ SEs * 3 -5 12 -24 Long-acting Short-acting * Long-acting (Some difference) 2 -4 5 -9 12 -18 OH 24 41

4. 1. SUs: 2 nd Gen*: Larger R 1 ↑ Potency • • Large R 1 group attached to aromatic ring ↑ binding affinity to SUR 1 of KATP channel ↑ potency/Do. A ↓ dose Fewer SEs/DDIs than 1 st generation SUs C/I: pregnancy, lactation More frequently prescribed for mild T 2 D 42

Glyburide is Metabolized to Long-Acting, Active Metabolites • • Glyburide: lipophilic (Log P/D 7. 4 = 3. 93/2. 28) 99% protein binding & large Vd long Do. A (24 h) Metabolism of glipizide is similar, but inactive metabolites. C/I of glyburide: hepatic or renal impairment (CLcr <50) or ≥ 43 60 yo (glipizide or glimepiride are safer & preferred SUs)

4. 1. 2 nd Gen. SUs: Glimepiride*: Metabolism Similar to glipizide, the most potent (daily dose 1 mg) Heterocyclic rings is pyrrolidine • • – • • Log P = 2. 94 Completely metabolized in liver to inactive metabolites Some SUs cause sulfonamide allergy remove S ? 44
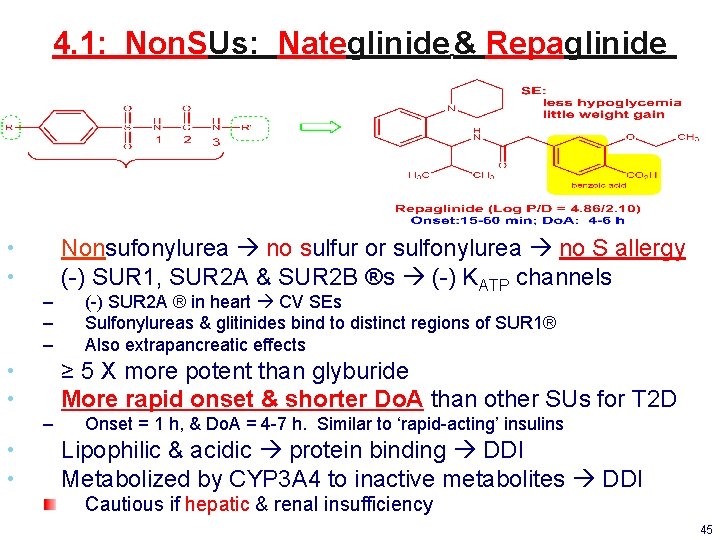
4. 1: Non. SUs: Nateglinide & Repaglinide • • – – – (-) SUR 2 A ® in heart CV SEs Sulfonylureas & glitinides bind to distinct regions of SUR 1® Also extrapancreatic effects ≥ 5 X more potent than glyburide More rapid onset & shorter Do. A than other SUs for T 2 D • • – • • Nonsufonylurea no sulfur or sulfonylurea no S allergy (-) SUR 1, SUR 2 A & SUR 2 B ®s (-) KATP channels Onset = 1 h, & Do. A = 4 -7 h. Similar to ‘rapid-acting’ insulins Lipophilic & acidic protein binding DDI Metabolized by CYP 3 A 4 to inactive metabolites DDI Cautious if hepatic & renal insufficiency 45
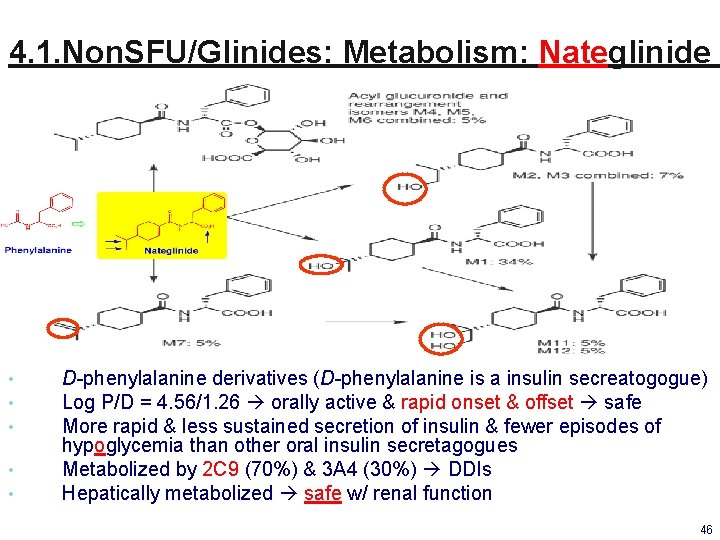
4. 1. Non. SFU/Glinides: Metabolism: Nateglinide • • • D-phenylalanine derivatives (D-phenylalanine is a insulin secreatogogue) Log P/D = 4. 56/1. 26 orally active & rapid onset & offset safe More rapid & less sustained secretion of insulin & fewer episodes of hypoglycemia than other oral insulin secretagogues Metabolized by 2 C 9 (70%) & 3 A 4 (30%) DDIs Hepatically metabolized safe w/ renal function 46
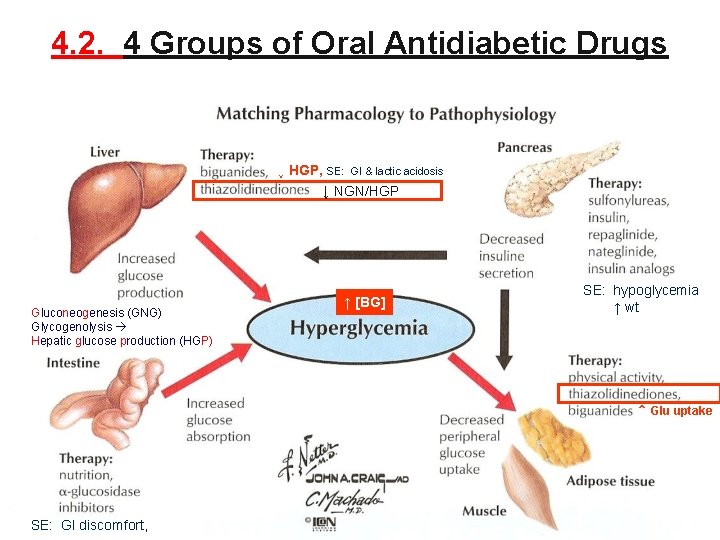
4. 2. 4 Groups of Oral Antidiabetic Drugs ↓ HGP, SE: GI & lactic acidosis ↓ NGN/HGP Gluconeogenesis (GNG) Glycogenolysis Hepatic glucose production (HGP) ↑ [BG] SE: hypoglycemia ↑ Insulin release & supplies ↑ wt ↑ Glu uptake 1. Insulin secretagogues SE: GI discomfort, ↓ Glu absorption 47
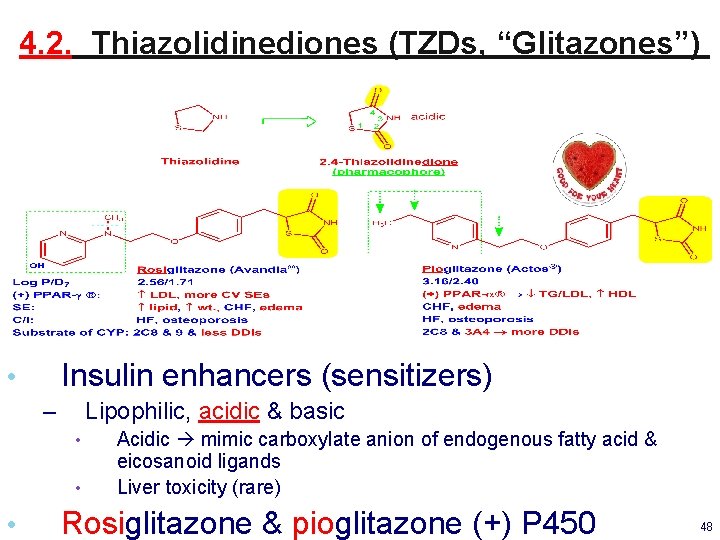
4. 2. Thiazolidinediones (TZDs, “Glitazones”) Insulin enhancers (sensitizers) • – Lipophilic, acidic & basic • • • Acidic mimic carboxylate anion of endogenous fatty acid & eicosanoid ligands Liver toxicity (rare) Rosiglitazone & pioglitazone (+) P 450 48
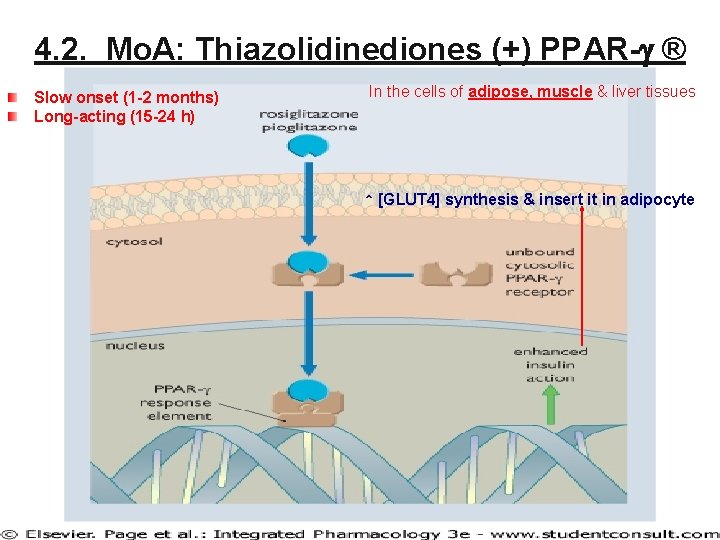
4. 2. Mo. A: Thiazolidinediones (+) PPAR-g ® Slow onset (1 -2 months) Long-acting (15 -24 h) In the cells of adipose, muscle & liver tissues ↑ [GLUT 4] synthesis & insert it in adipocyte 49
![4. 2. TZDs: Rosiglitazone* & Pioglitazone* Proposed Mo. A (↓ insulin resistance ↓ [BG]) 4. 2. TZDs: Rosiglitazone* & Pioglitazone* Proposed Mo. A (↓ insulin resistance ↓ [BG])](http://slidetodoc.com/presentation_image/261aa31610df2f82a7087ba7ef01cf2e/image-50.jpg)
4. 2. TZDs: Rosiglitazone* & Pioglitazone* Proposed Mo. A (↓ insulin resistance ↓ [BG]) • – (+) nuclear peroxisome proliferator-activated-gamma ® (PPAR-g ®) • Regulates transcription of genes encoding proteins involved in carbohydrate & lipid metabolism – – – ↑ synthesis & translocation of Glu transporter proteins (GLUT 1 & 4) ↑ Glu uptake in adipose, muscle & liver tissue ↓ [BG] ↑ [Glu TG] in adipocytes* ↓ [BG], but ↑ wt. (-) gluconeogenesis (GNG) in liver ↓ [HGP] ↓ [BG] ↑ target tissues (adipose, skeletal muscle & liver) sensitivity to insulin ↓ insulin resistance ↓ [BG] ↓ both fasting & postprandial hyperglycemia Slow onset & offset of activity 50
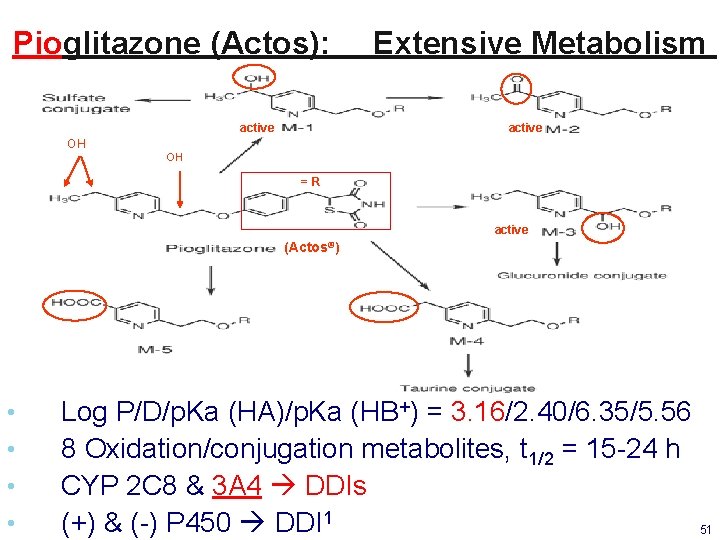
Pioglitazone (Actos): active Extensive Metabolism active OH OH =R active (Actos®) • • Log P/D/p. Ka (HA)/p. Ka (HB+) = 3. 16/2. 40/6. 35/5. 56 8 Oxidation/conjugation metabolites, t 1/2 = 15 -24 h CYP 2 C 8 & 3 A 4 DDIs (+) & (-) P 450 DDI 1 51
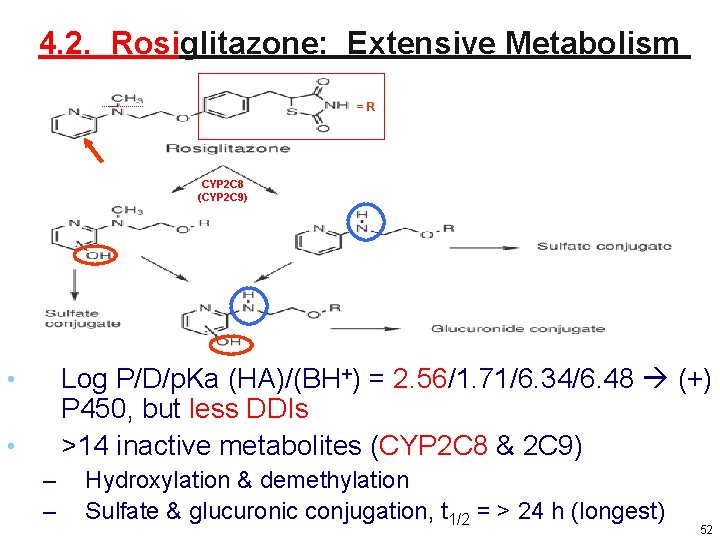
4. 2. Rosiglitazone: Extensive Metabolism =R CYP 2 C 8 (CYP 2 C 9) Log P/D/p. Ka (HA)/(BH+) = 2. 56/1. 71/6. 34/6. 48 (+) P 450, but less DDIs >14 inactive metabolites (CYP 2 C 8 & 2 C 9) • • – – Hydroxylation & demethylation Sulfate & glucuronic conjugation, t 1/2 = > 24 h (longest) 52
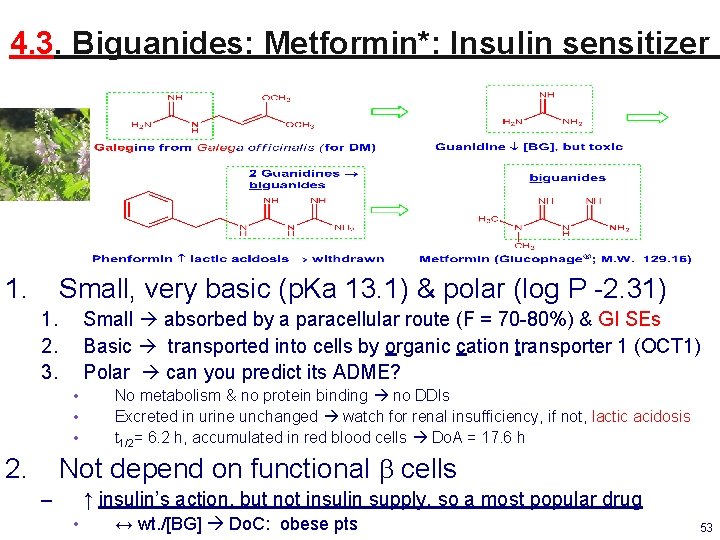
4. 3. Biguanides: Metformin*: Insulin sensitizer 1. Small, very basic (p. Ka 13. 1) & polar (log P -2. 31) 1. 2. 3. Small absorbed by a paracellular route (F = 70 -80%) & GI SEs Basic transported into cells by organic cation transporter 1 (OCT 1) Polar can you predict its ADME? • • • No metabolism & no protein binding no DDIs Excreted in urine unchanged watch for renal insufficiency, if not, lactic acidosis t 1/2= 6. 2 h, accumulated in red blood cells Do. A = 17. 6 h. Clin Pharmacokinet. 2011; 50(2): 81 Not depend on functional b cells 2. – ↑ insulin’s action, but not insulin supply, so a most popular drug • ↔ wt. /[BG] Do. C: obese pts 53

4. 3. Metformin (Englycemic): Proposed Mo. A 1 If ↓ cellular energy (ATP) ↑ AMPactivated protein kinase (AMPK, a glu sensor) ↑ (anabolic catabolic activity) Metformin (+) AMPK ↓ GNG/HGP/HGO & ↑ cellular Glu utilization ↓ insulin resistance 2. 1. 2. 3. ↓ hepatic & renal gluconeogenesis (lactate glu) ↓ excessive glu production & ↑ [lactate] SE: lactic acidosis (rare) ↑ muscle uptake of glu & tissue sensitivity to insulin ↑ glu utilization Minor (AMPK-independent) Mo. A 1. 2. 3. 2. 4. 3. 5. ↓ glu absorption from GIT GI SEs: nausea, flatulent (gas), diarrhea ↓ plasma glucagon & (glu lactate) by enterocytes (+) AMPK ↑ glu removal from blood/uptake in tissues 4. (+) glycolysis in tissues glu utilization Less hypoglycemia SEs & no ↑ wt. ↓ postprandial & fasting [BG] in pts with T 2 D 54

4. 4. a-Glucosidase Inhibitors: Mo. A To be absorbed from GIT into blood • – Complex carbohydrates must first be hydrolyzed to monosaccharides a-Glucosidase • – Converts complex starches, oligosaccharides & disaccharides to monosaccharides that can be transported out of intestinal lumen & into blood 55

4. 4. a-Glucosidase Inhibitors: Carbohydrate analogs (“starch blockers”) Competitively (-) a-glucosidase within intestine (-) disaccharide hydrolysis delay monosaccharide production ↓ Glu absorption rate in intestine ↓ [BG] (-) Carbohydrate digestion in small intestine • • • – • • • Mo. A ↑ time required for glu absorption No risk of hypoglycemia or weight gain ↑ GLP-1 release into circulation ↓ [BG] ↓ Postprandial [BG] rise & taken before a meal Diet might be better medication? 56
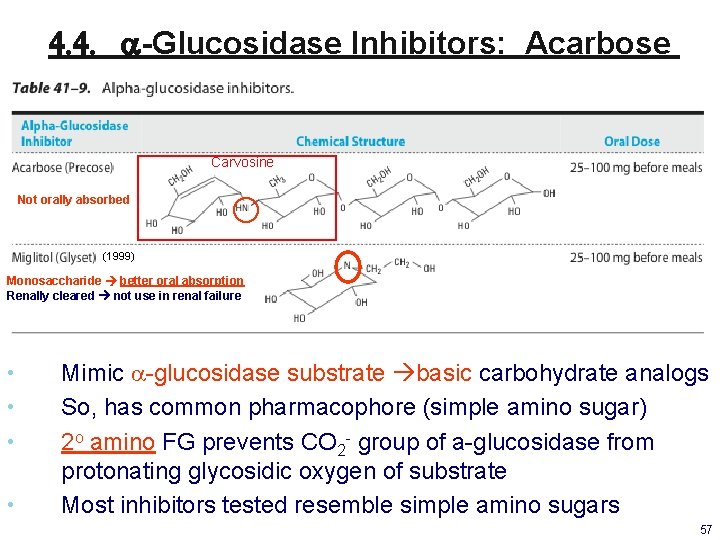
4. 4. a-Glucosidase Inhibitors: Acarbose Carvosine Not orally absorbed (1999) Monosaccharide better oral absorption Renally cleared not use in renal failure • • Mimic a-glucosidase substrate basic carbohydrate analogs So, has common pharmacophore (simple amino sugar) 2 o amino FG prevents CO 2 - group of a-glucosidase from protonating glycosidic oxygen of substrate Most inhibitors tested resemble simple amino sugars 57

Acarbose: Polar Drug PK Profile (ADME) Polar (log P -3. 03) & basic 1. Minimally absorbed (0. 5 -1. 7%) into blood designed to acts on GIT only & ↓ systemic toxicity • But, poorly tolerated not used often b/c 2. Annoying GI SEs in ~60% of pts, b/c • • ↑ fermentation of unabsorbed carbohydrate in colon/short-chain fatty acid flatulence (gas)/bloating C/I: chronic intestinal disease, IBD, cirrhosis 3. Amylases & bacterial degrade acarbose in intestine 4. Some by-products are systemically absorbed & eliminated in urine not recommended in pts with significant renal dysfunction 5. Tx: less efficacious adjunct therapy or for elderly pts 58
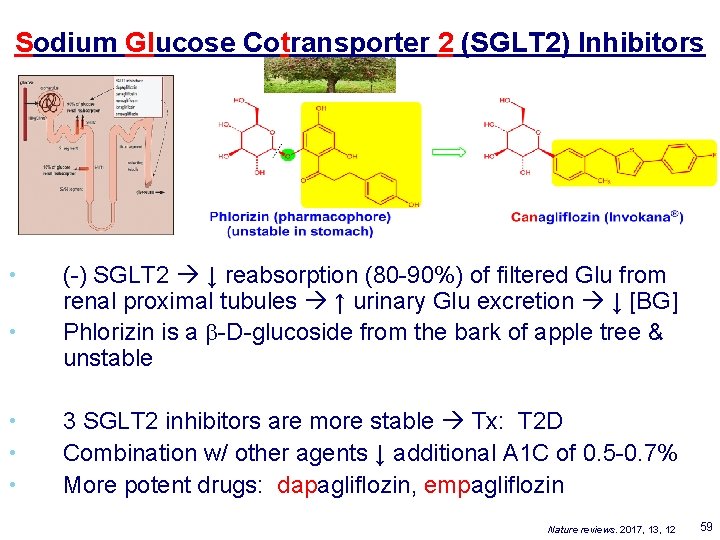
Sodium Glucose Cotransporter 2 (SGLT 2) Inhibitors • • • (-) SGLT 2 ↓ reabsorption (80 -90%) of filtered Glu from renal proximal tubules ↑ urinary Glu excretion ↓ [BG] Phlorizin is a b-D-glucoside from the bark of apple tree & unstable 3 SGLT 2 inhibitors are more stable Tx: T 2 D Combination w/ other agents ↓ additional A 1 C of 0. 5 -0. 7% More potent drugs: dapagliflozin, empagliflozin Nature reviews. 2017, 13, 12 59
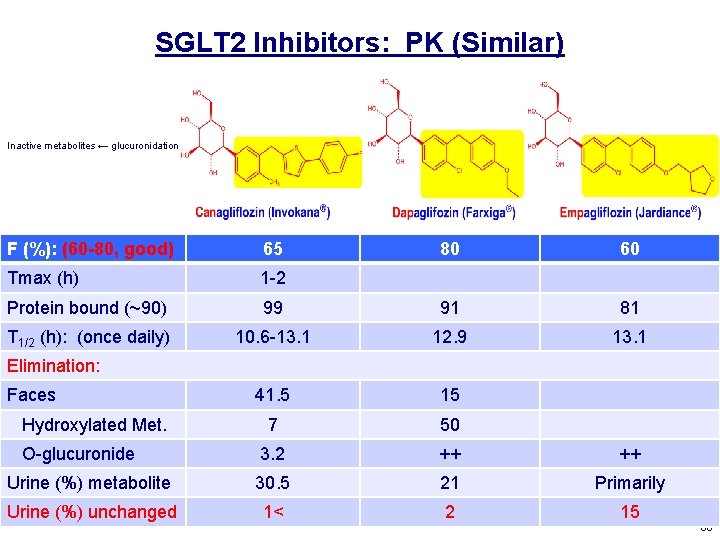
SGLT 2 Inhibitors: PK (Similar) Inactive metabolites ← glucuronidation F (%): (60 -80, good) 65 80 60 Tmax (h) 1 -2 Protein bound (~90) 99 91 81 T 1/2 (h): (once daily) 10. 6 -13. 1 12. 9 13. 1 41. 5 15 7 50 3. 2 ++ ++ Urine (%) metabolite 30. 5 21 Primarily Urine (%) unchanged 1< 2 15 Elimination: Faces Hydroxylated Met. O-glucuronide 60
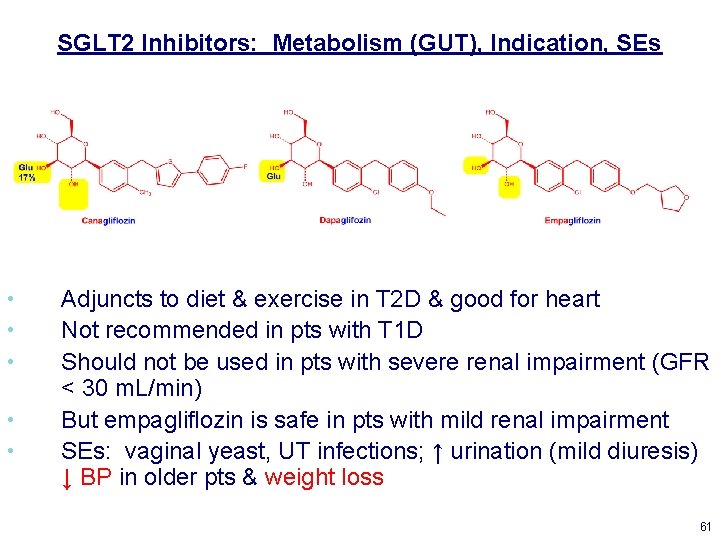
SGLT 2 Inhibitors: Metabolism (GUT), Indication, SEs • • • Adjuncts to diet & exercise in T 2 D & good for heart Not recommended in pts with T 1 D Should not be used in pts with severe renal impairment (GFR < 30 m. L/min) But empagliflozin is safe in pts with mild renal impairment SEs: vaginal yeast, UT infections; ↑ urination (mild diuresis) ↓ BP in older pts & weight loss 61
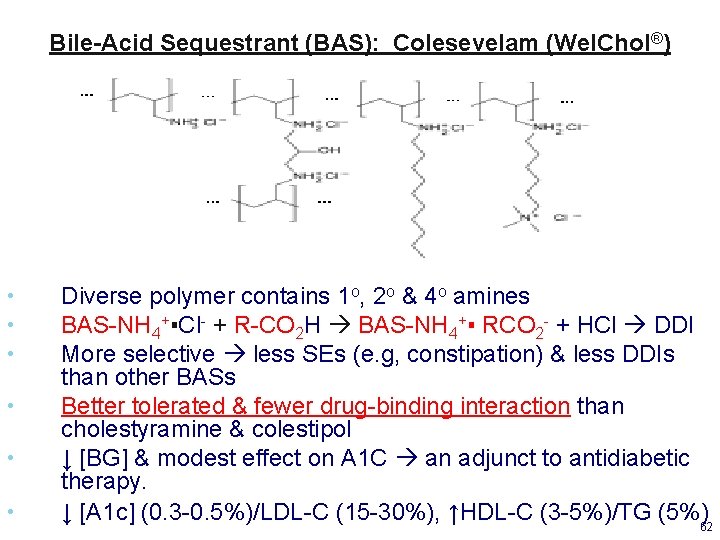
Bile-Acid Sequestrant (BAS): Colesevelam (Wel. Chol®) • • • Diverse polymer contains 1 o, 2 o & 4 o amines BAS-NH 4+▪Cl- + R-CO 2 H BAS-NH 4+▪ RCO 2 - + HCl DDI More selective less SEs (e. g, constipation) & less DDIs than other BASs Better tolerated & fewer drug-binding interaction than cholestyramine & colestipol ↓ [BG] & modest effect on A 1 C an adjunct to antidiabetic therapy. ↓ [A 1 c] (0. 3 -0. 5%)/LDL-C (15 -30%), ↑HDL-C (3 -5%)/TG (5%)62
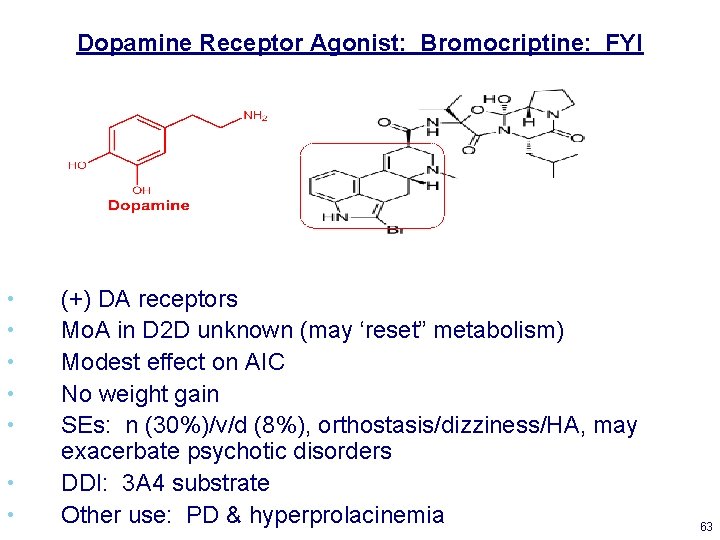
Dopamine Receptor Agonist: Bromocriptine: FYI • • (+) DA receptors Mo. A in D 2 D unknown (may ‘reset” metabolism) Modest effect on AIC No weight gain SEs: n (30%)/v/d (8%), orthostasis/dizziness/HA, may exacerbate psychotic disorders DDI: 3 A 4 substrate Other use: PD & hyperprolacinemia 63
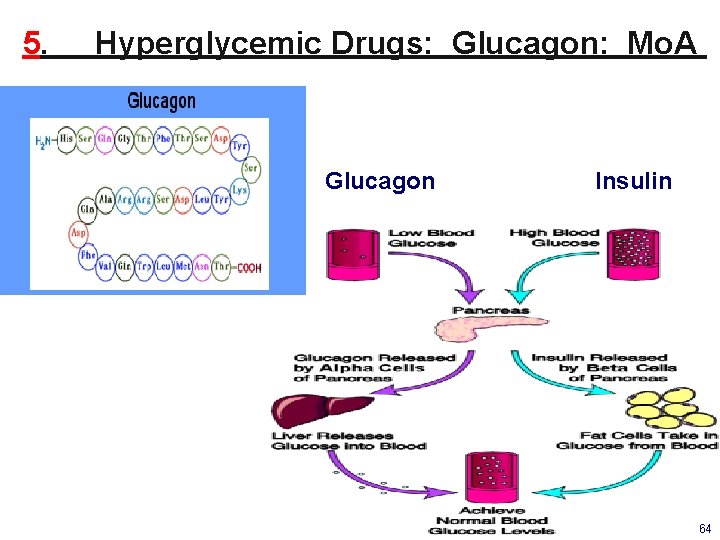
5. Hyperglycemic Drugs: Glucagon: Mo. A Glucagon Insulin 64

5. Hyperglycemic Drug: Glucagon 29 a. a. peptide of a cells of endocrine pancreas (+) GPCR glucagon ®s (↑ AC ↑ c. AMP) located in • • – – Heart ↑ heart rate & force of contraction Tx: severe b-blocker overdose (↓ c. AMP) • • – (+) depressed heart ↑ cardiac c. AMP w/o requiring access to b ®s Liver ↑ glycogenolysis/gluconeogenesis (GNG) ↑ hepatic Glu production (HGP) • • ↑ (glycogen glucose) ↑ [GB] ↑ (lactate, pyruvate, glycerol & a. a. glucose) ↑ [BG] • Tx: severe hypoglycemia in diabetics (IM or IV) If Glu administration is impossible Its action requires intact hepatic glycogen stores • – Smooth muscle • Relax smooth muscle particularly in the gut 65
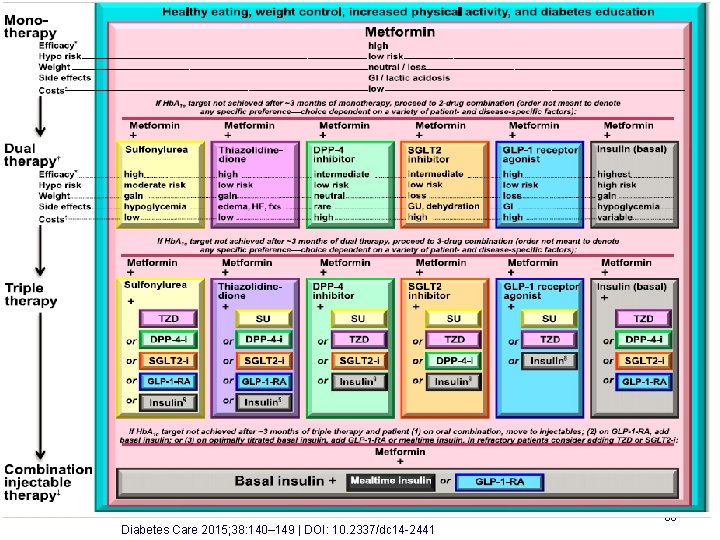
Diabetes Care 2015; 38: 140– 149 | DOI: 10. 2337/dc 14 -2441 66

THANK YOU FOR YOUR ATTENTION, QUESTIONS?
- Slides: 67