Mechanistic notation The mechanism just described is an

Mechanistic notation The mechanism just described is an example of an SN 1 process. SN 1 stands for substitution-nucleophilicunimolecular. The molecularity of the rate-determining step defines the molecularity of the overall reaction.
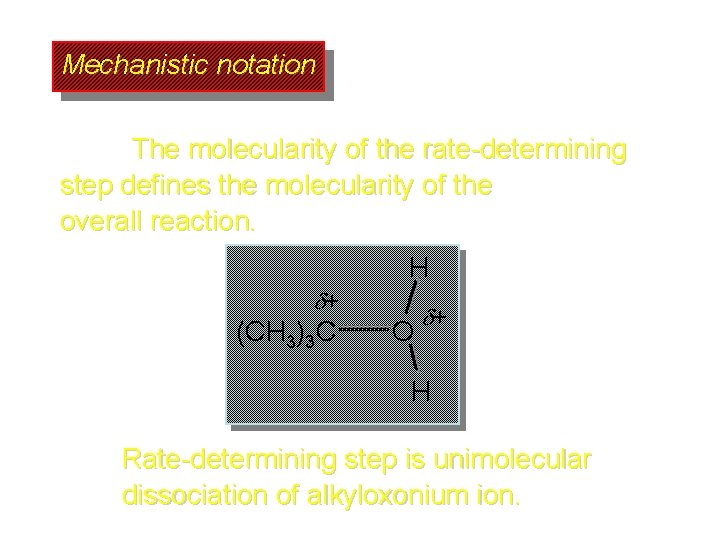
Mechanistic notation The molecularity of the rate-determining step defines the molecularity of the overall reaction. H + (CH 3)3 C O + H Rate-determining step is unimolecular dissociation of alkyloxonium ion.

Effect of Alcohol Structure on Reaction Rate

slow step is: ROH 2+ R+ + H 2 O The more stable the carbocation, the faster it is formed. Tertiary carbocations are more stable than secondary, which are more stable than primary, which are more stable than methyl. Tertiary alcohols react faster than secondary, which react faster than primary, which react faster than methanol.

Hammond's Postulate If two succeeding states (such as a transition state and an unstable intermediate) are similar in energy, they are similar in structure. Hammond's postulate permits us to infer the structure of something we can't study (transition state) from something we can study (reactive intermediate).
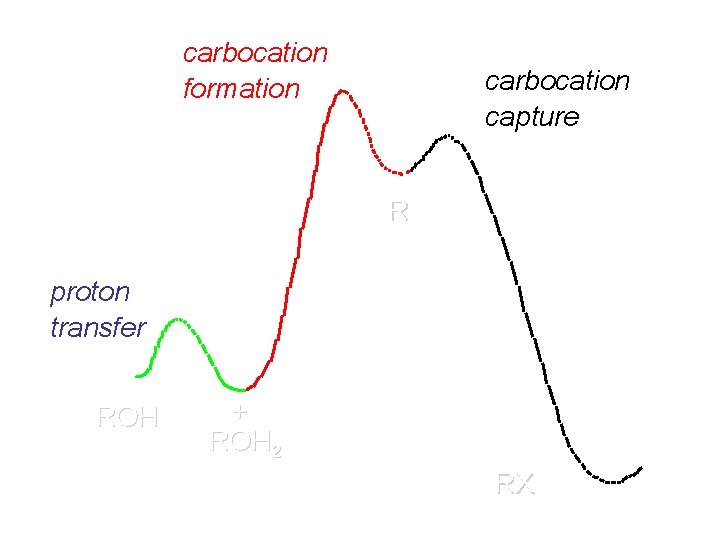
carbocation formation carbocation capture R+ proton transfer ROH + ROH 2 RX
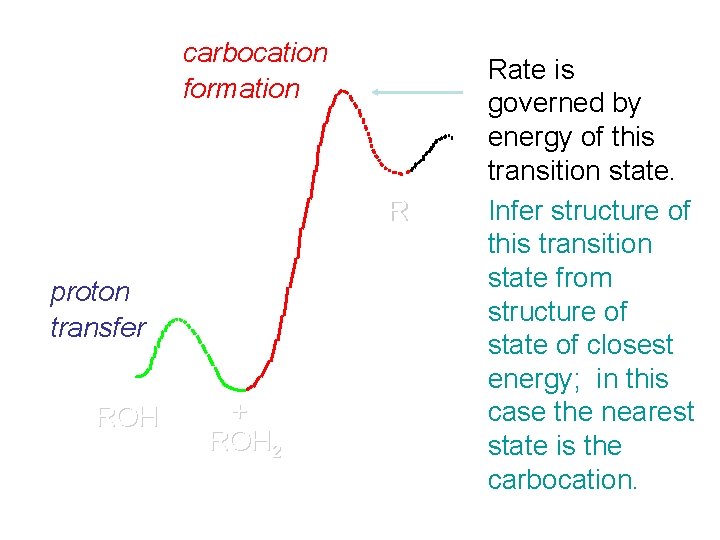
carbocation formation R+ proton transfer ROH + ROH 2 Rate is carbocation governed by capture energy of this transition state. Infer structure of this transition state from structure of state of closest energy; in this case the nearest state is the carbocation. RX

4. 13 Reaction of Primary Alcohols with Hydrogen Halides. The SN 2 Mechanism
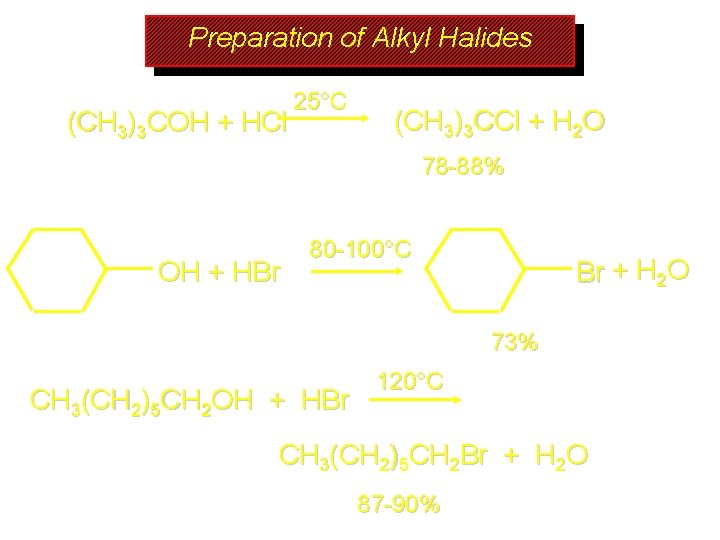
Preparation of Alkyl Halides (CH 3)3 COH + HCl 25°C (CH 3)3 CCl + H 2 O 78 -88% OH + HBr 80 -100°C Br + H 2 O 73% CH 3(CH 2)5 CH 2 OH + HBr 120°C CH 3(CH 2)5 CH 2 Br + H 2 O 87 -90%

Preparation of Alkyl Halides Primary carbocations are too high in energy to allow SN 1 mechanism. Yet, primary alcohols are converted to alkyl halides. Primary alcohols react by a mechanism called SN 2 (substitution-nucleophilic-bimolecular). CH 3(CH 2)5 CH 2 OH + HBr 120°C CH 3(CH 2)5 CH 2 Br + H 2 O 87 -90%

The SN 2 Mechanism Two-step mechanism for conversion of alcohols to alkyl halides: (1) proton transfer to alcohol to form alkyloxonium ion (2) bimolecular displacement of water from alkyloxonium ion by halide
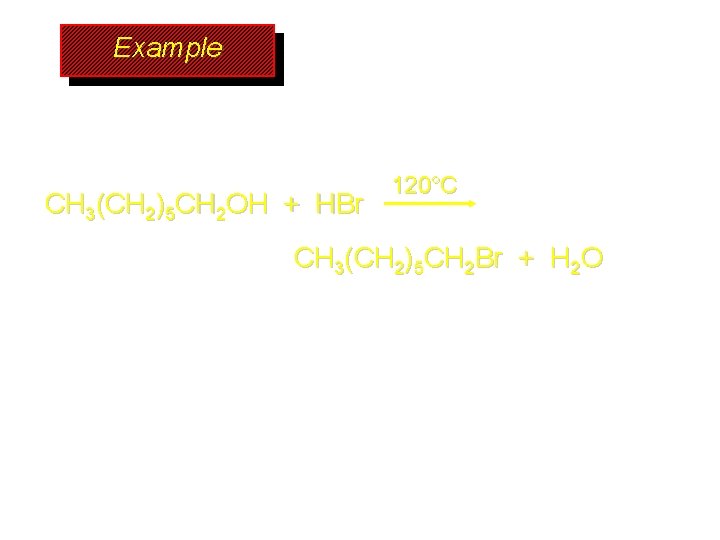
Example CH 3(CH 2)5 CH 2 OH + HBr 120°C CH 3(CH 2)5 CH 2 Br + H 2 O
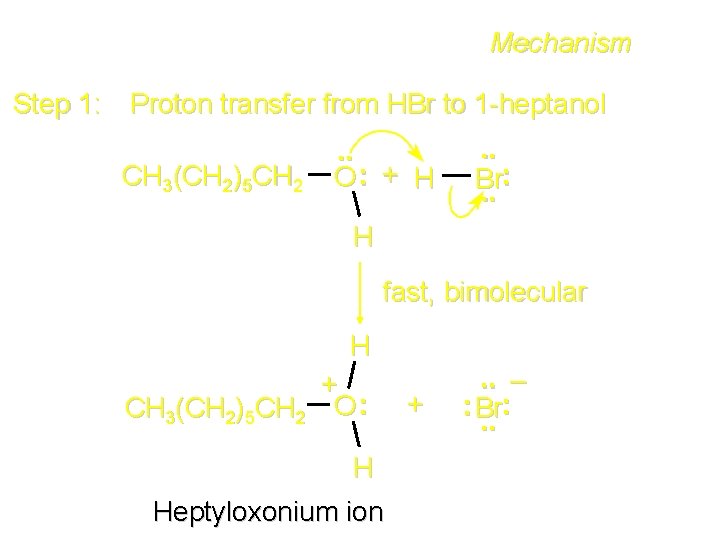
Mechanism Step 1: Proton transfer from HBr to 1 -heptanol CH 3(CH 2)5 CH 2 . . O: + H . . : Br. . H fast, bimolecular H + CH 3(CH 2)5 CH 2 O : H Heptyloxonium ion + . . – : Br: . .
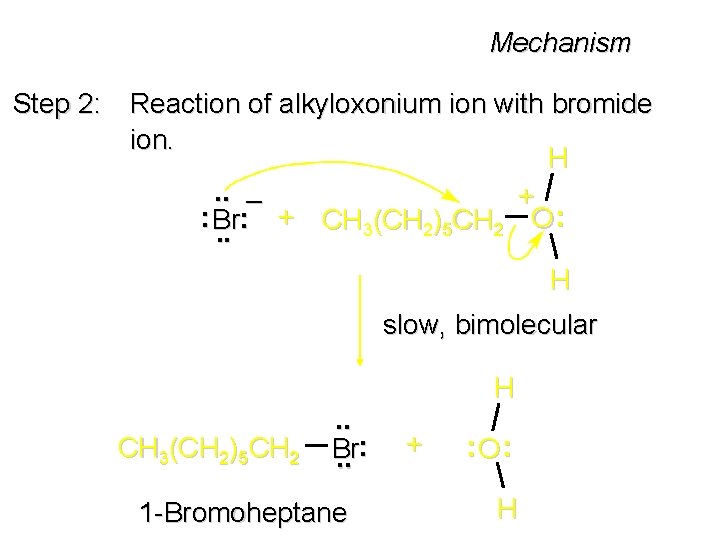
Mechanism Step 2: Reaction of alkyloxonium ion with bromide ion. H. . – + : Br: + CH 3(CH 2)5 CH 2 O : . . H slow, bimolecular CH 3(CH 2)5 CH 2 . . Br. . : 1 -Bromoheptane H + : O: H
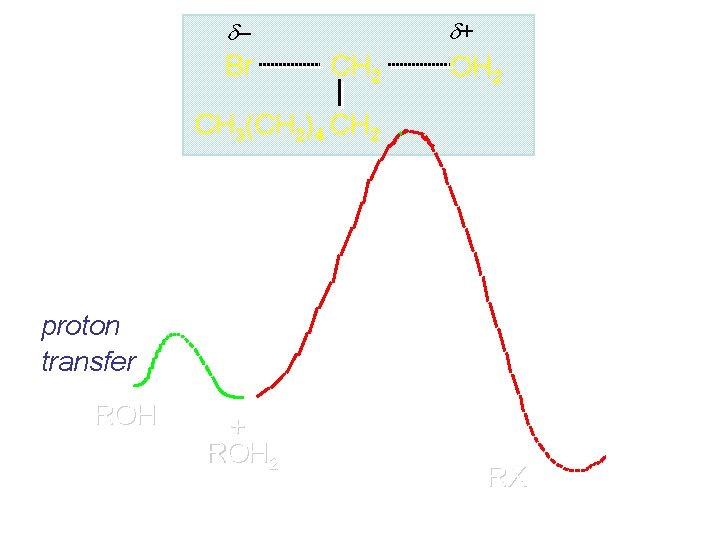
+ – Br CH 2 OH 2 CH 3(CH 2)4 CH 2 proton transfer ROH + ROH 2 RX

4. 14 Other Methods for Converting Alcohols to Alkyl Halides

Reagents for ROH to RX Thionyl chloride SOCl 2 + ROH RCl + HCl + SO 2 Phosphorus tribromide PBr 3 + 3 ROH 3 RBr + H 3 PO 3
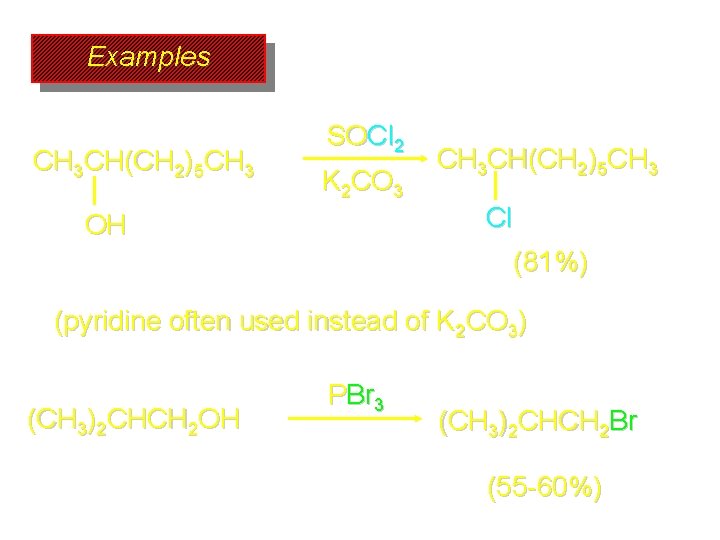
Examples CH 3 CH(CH 2)5 CH 3 SOCl 2 K 2 CO 3 OH CH 3 CH(CH 2)5 CH 3 Cl (81%) (pyridine often used instead of K 2 CO 3) (CH 3)2 CHCH 2 OH PBr 3 (CH 3)2 CHCH 2 Br (55 -60%)
- Slides: 18