Mechanisms Review A reaction occurs by the following

Mechanisms Review: A reaction occurs by the following mechanism. step 1: Ce 4+ + Mn 2+ Ce 3+ + Mn 3+ step 2: Ce 4+ + Mn 3+ Ce 3+ + Mn 4+ step 3: Tl+ + Mn 4+ Tl 3+ + Mn 2+ a. Write the net reaction b. Identify each as a reactant, product, intermediate or catalyst: Mn 2+ = Mn 4+ = Mn 3+ = Tl+ = Ce 4+ = Tl 3+ = Ce 3+ =

Mechanisms Review: A reaction occurs by the following mechanism. step 1: Ce 4+ + Mn 2+ Ce 3+ + Mn 3+ step 2: Ce 4+ + Mn 3+ Ce 3+ + Mn 4+ step 3: Tl+ + Mn 4+ Tl 3+ + Mn 2+ c. Create a reaction coordinate diagram for the reaction mechanism shown with the following guidelines: • • • The Ea for step 1 is 50 k. J The Ea for step 2 is 50 k. J The Ea for step 3 is 100 k. J The overall Ea for the whole reaction is 150 k. J The reaction has a ∆H = +50 k. J/mol

Mechanisms Review: A reaction occurs by the following mechanism. step 1: Ce 4+ + Mn 2+ Ce 3+ + Mn 3+ step 2: Ce 4+ + Mn 3+ Ce 3+ + Mn 4+ step 3: Tl+ + Mn 4+ Tl 3+ + Mn 2+ d. If the un-catalyzed reaction occurs in 1 step, why is it predicted to be so slow?

Stoichiometry can only be used to write rate laws for mechanism steps!!! (NOT OVERALL REACTIONS) Rates of Mechanism Steps: Each individual step has its own rate, and therefore its own rate law. Mechanism Step A product A + B product 2 A product Molecularity Unimolecular Bimolecular Rate law Rate = k [A] [B] Rate = k [A]2 2 A + B product Termolecular Rate = k [A]2 [B]

Stoichiometry can only be used to write rate laws for mechanism steps!!! (NOT OVERALL REACTIONS) Rates of Mechanism Steps: Each individual step has its own rate, and therefore its own rate law. Mechanism Step A product A + B product 2 A product Molecularity Unimoleuclar Bimolecular Rate law Rate = k [A] [B] Rate = k [A]2 2 A + B product Termolecular Rate = k [A]2 [B]

Stoichiometry can only be used to write rate laws for Rates of Mechanism Steps: mechanism steps!!! Each individual step has its own rate, and therefore its (NOT REACTIONS) own rate OVERALL law. Mechanism Step A product A + B product 2 A product Molecularity Unimoleuclar Bimolecular Rate law Rate = k [A] [B] Rate = k [A]2 2 A + B product Termolecular Rate = k [A]2 [B]

Stoichiometry can only be used to write rate laws for Rates of Mechanism Steps: Each individual step has steps!!! its own rate, and therefore its mechanism own rate law. (NOT OVERALL REACTIONS) Mechanism Step A product A + B product 2 A product Molecularity Unimoleuclar Bimolecular Rate law Rate = k [A] [B] Rate = k [A]2 2 A + B product Termolecular Rate = k [A]2 [B]

Stoichiometry can only be used to write rate laws for mechanism steps!!! (NOT OVERALL Rates of Mechanism Steps: Each individual step has its own rate, and therefore its own rate law. Mechanism Step A product A + B product 2 A product Molecularity Unimoleuclar Bimolecular Rate law Rate = k [A] [B] Rate = k [A]2 2 A + B product Termolecular Rate = k [A]2 [B]

Rate Determining Steps: General Rule: The rate of an overall reaction can never be faster than the rate of the slowest step! Step 1: A + B Step 2: M + A Overall: slow fast X + M Rate Determining Step Y The rate law for the slow step will match the rate law of the overall reaction!

Rate Determining Steps: Net reaction: NO 2 + CO NO + CO 2 Step 1: 2 NO 2 NO 3 + NO (slow) Step 2: NO 3 + CO NO 2 + CO 2 (fast) Use the proposed mechanism to determine the rate law for the overall reaction.

Rate Determining Steps: 2 NO 2 + F 2 2 FNO 2 rate = k[NO 2][F 2] Proposed mechanism: Step 1: NO 2 + F 2 FNO 2 + F Step 2: NO 2 + F FNO 2 In the mechanism proposed above, which step is likely to be the rate determining step?

Rate Determining Steps: Net reaction: NO 2 + CO NO + CO 2 rate = k [NO 2]2 Can the reaction occur in one bimolecular mechanism step? How do you know?
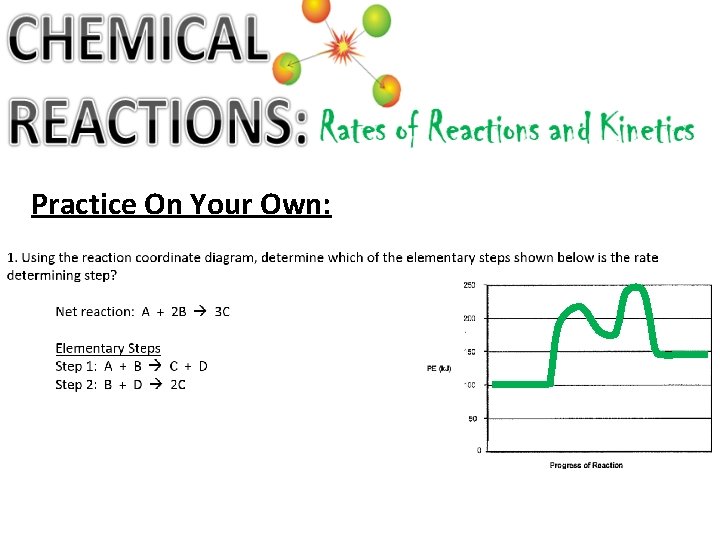
Practice On Your Own:

Practice On Your Own:
- Slides: 14