MECE251 Thermo Lesson 2 Properties of Pure Substances

MECE-251 Thermo. , Lesson 2: Properties of Pure Substances A pure substance must have the same molecular composition throughout. Water is a pure substance. Air is not a pure substance. In some cases “dry air” may be treated like a pure substance. Moist air is a mixture of water and dry air – we’ll come back to this later. R·I·T MECE-251 Read Sections 2. 1 – 2. 6 Mon Solve Problems Tue View Lectures and Case Studies Wed Thu Solve Case Study Fri Complete On-line Lesson Quiz Sat or Sun 1
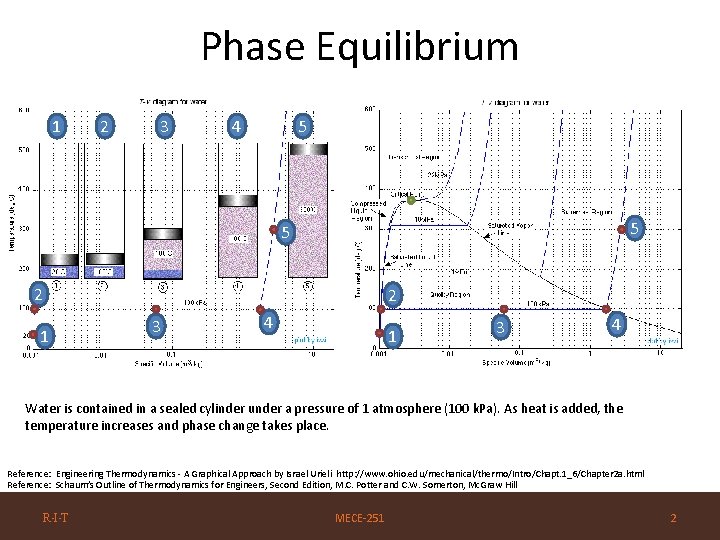
Phase Equilibrium 1 2 3 4 5 5 5 2 1 2 3 4 1 3 4 Water is contained in a sealed cylinder under a pressure of 1 atmosphere (100 k. Pa). As heat is added, the temperature increases and phase change takes place. Reference: Engineering Thermodynamics - A Graphical Approach by Israel Urieli http: //www. ohio. edu/mechanical/thermo/Intro/Chapt. 1_6/Chapter 2 a. html Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 2
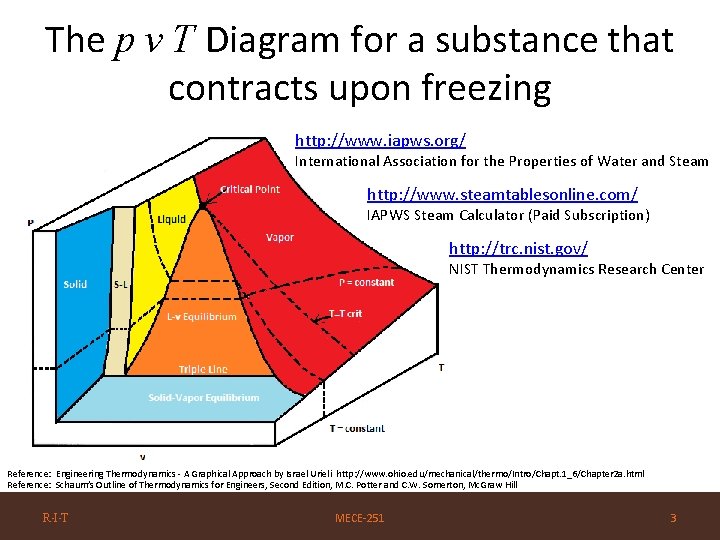
The p v T Diagram for a substance that contracts upon freezing http: //www. iapws. org/ International Association for the Properties of Water and Steam http: //www. steamtablesonline. com/ IAPWS Steam Calculator (Paid Subscription) http: //trc. nist. gov/ NIST Thermodynamics Research Center Reference: Engineering Thermodynamics - A Graphical Approach by Israel Urieli http: //www. ohio. edu/mechanical/thermo/Intro/Chapt. 1_6/Chapter 2 a. html Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 3

Thermodynamic Properties of Water (aka Steam Tables) Schaum’s Appendix C. “The Vapor Dome” Saturation Temperature (boiling point) of Water is 100 C at 101. 3 k. Pa. Saturation Temperature (boiling point) of Water is 180 C at 1 MPa (10 atm. ) Critical Point of Water is T = 374 C and P = 22. 1 MPa Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 4
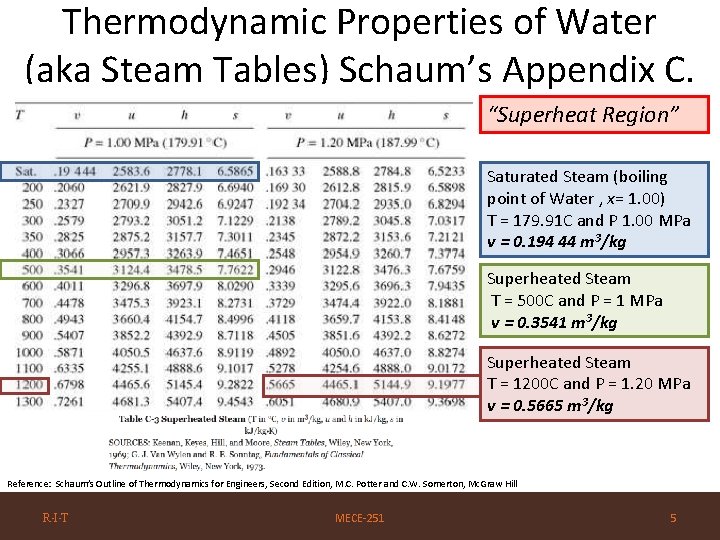
Thermodynamic Properties of Water (aka Steam Tables) Schaum’s Appendix C. “Superheat Region” Saturated Steam (boiling point of Water , x= 1. 00) T = 179. 91 C and P 1. 00 MPa v = 0. 194 44 m 3/kg Superheated Steam T = 500 C and P = 1 MPa v = 0. 3541 m 3/kg Superheated Steam T = 1200 C and P = 1. 20 MPa v = 0. 5665 m 3/kg Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 5

Thermodynamic Properties of Water (aka Steam Tables) Schaum’s Appendix C. “Compressed Liquid” Compressed Liquid T = 0 C and P 5. 00 MPa v = 0. 000 997 7 m 3/kg Compressed Liquid T = 120 C and P = 5 MPa v = 001 057 6 m 3/kg Compressed Liquid T = 260 C and P = 10 MPa v = 0. 001 264 5 m 3/kg Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 6
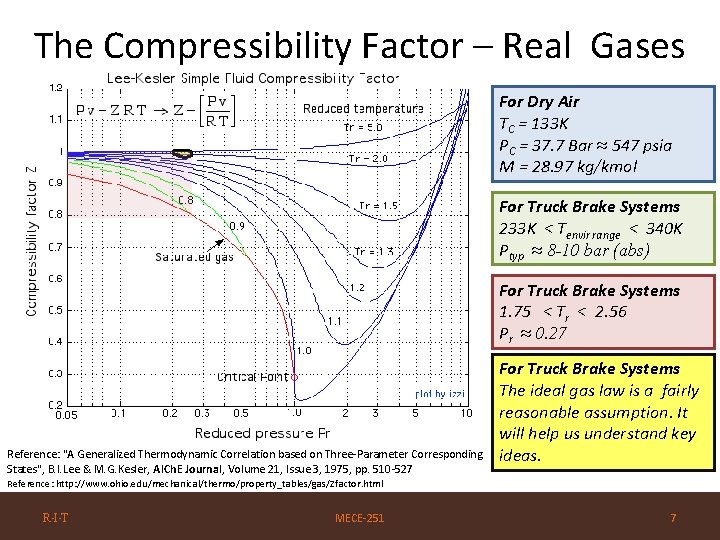
The Compressibility Factor – Real Gases For Dry Air TC = 133 K PC = 37. 7 Bar ≈ 547 psia M = 28. 97 kg/kmol For Truck Brake Systems 233 K < Tenvir range < 340 K Ptyp ≈ 8 -10 bar (abs) For Truck Brake Systems 1. 75 < Tr < 2. 56 Pr ≈ 0. 27 Reference: "A Generalized Thermodynamic Correlation based on Three-Parameter Corresponding States", B. I. Lee & M. G. Kesler, AICh. E Journal, Volume 21, Issue 3, 1975, pp. 510 -527 For Truck Brake Systems The ideal gas law is a fairly reasonable assumption. It will help us understand key ideas. Reference: http: //www. ohio. edu/mechanical/thermo/property_tables/gas/Zfactor. html R·I·T MECE-251 7

Equations of State for Dry Air For Dry Air TC = 133 K = 239 R PC = 37. 7 Bar ≈ 547 psia M = 28. 97 kg/kmol = 28. 97 lbm/lbmol R = 0. 287 k. J/Kg·K = 53. 3 ft·lbf / lbm·R Ideal Gas, Z ≈ 1 Compressible Gas, Z < 1 Pv = RT Z=Pv PV =m. RT RT P = ρ RT P V = Nmoles Runiversal T Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 8

Next Steps L 2 Task 3 B: Please review the example problems on line. L 2 Task 3 C: Then, solve the review problem. L 2 Task 4 A: Please review the lecture on line. L 2 Task 4 B: Please review the example problems on line. L 2 Task 4 C: Then, solve the review problem. L 2 Task 5: Solve and TURN IN the case study problem. L 2 Task 6: Take the Lesson 2 quiz. Reference: Schaum’s Outline of Thermodynamics for Engineers, Second Edition, M. C. Potter and C. W. Somerton, Mc. Graw Hill R·I·T MECE-251 9
- Slides: 9