Measuring Trends in the Development of New Drugs


















- Slides: 41

Measuring Trends in the Development of New Drugs: Time, Costs, Risks and Returns Joseph A. Di. Masi, Ph. D. Director of Economic Analysis Tufts Center for the Study of Drug Development Tufts University SLA Pharmaceutical & Health Technology Division Spring Meeting Boston, MA, March 19, 2007

Agenda u New Drug development times u Risks in new drug development u R&D costs and returns for new drugs u Pace of competitive development u Impact of improvements to the R&D process u Trends in new drug pipelines

New Drug Development Times

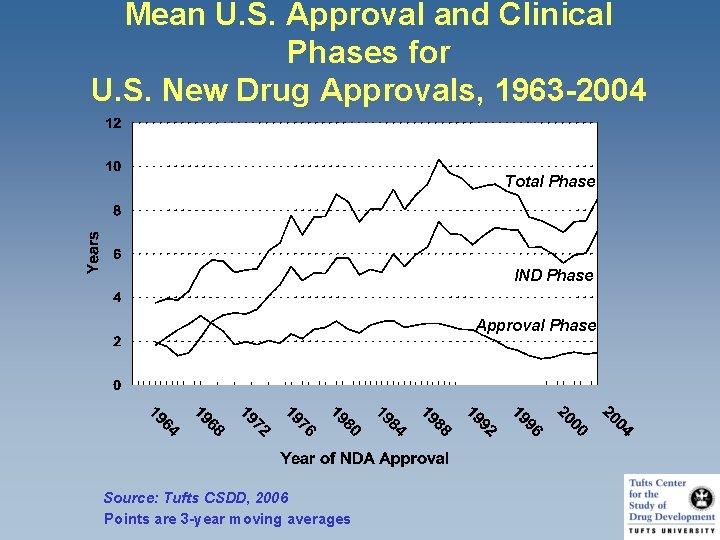
Mean U. S. Approval and Clinical Phases for U. S. New Drug Approvals, 1963 -2004 Total Phase IND Phase Approval Phase Source: Tufts CSDD, 2006 Points are 3 -year moving averages

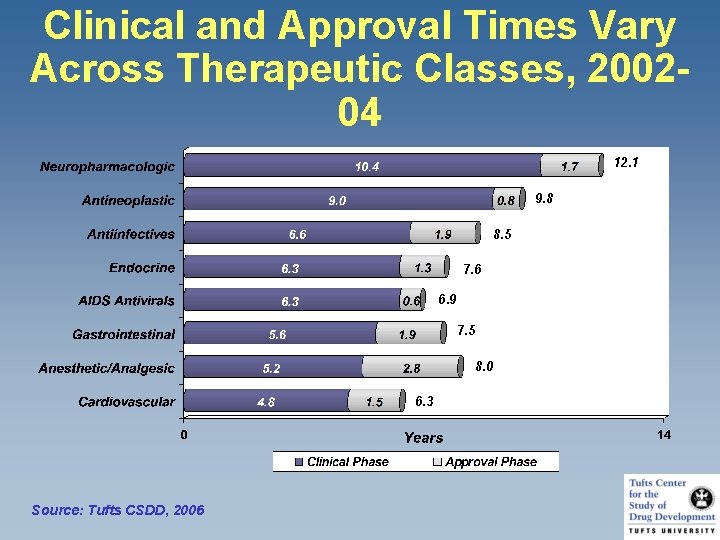
Clinical and Approval Times Vary Across Therapeutic Classes, 200204 12. 1 9. 8 8. 5 7. 6 6. 9 7. 5 8. 0 6. 3 Source: Tufts CSDD, 2006

New Drug Development Risk

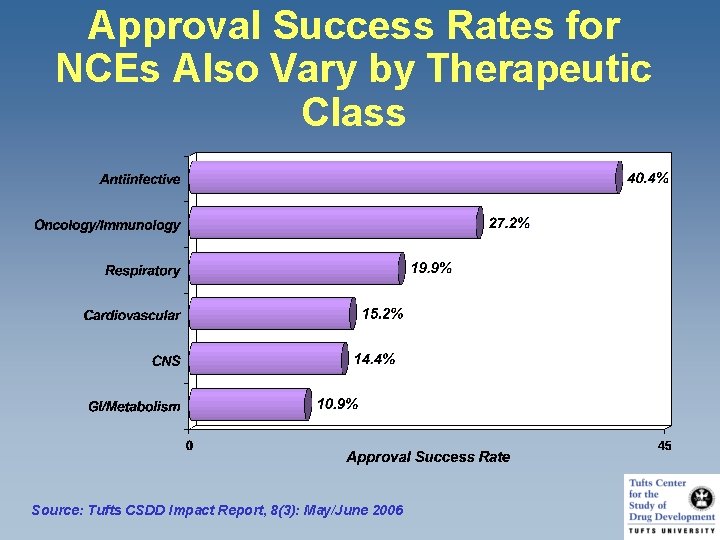
Approval Success Rates for NCEs Also Vary by Therapeutic Class Source: Tufts CSDD Impact Report, 8(3): May/June 2006

Pharmaceutical R&D Productivity

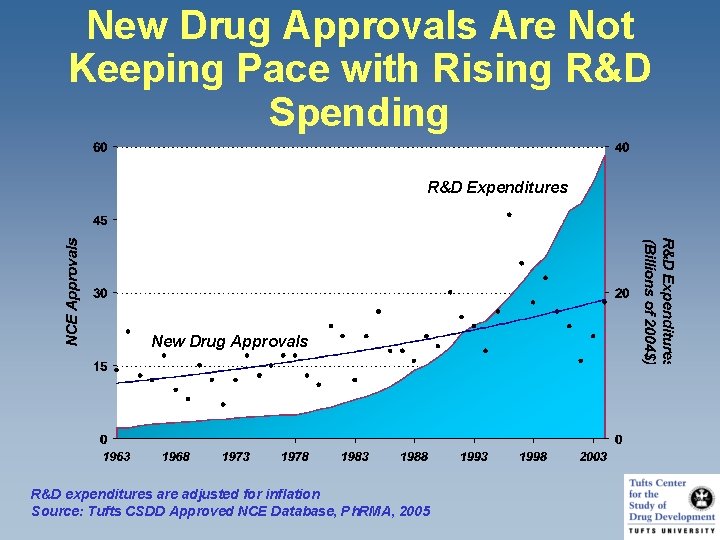
New Drug Approvals Are Not Keeping Pace with Rising R&D Spending R&D Expenditures New Drug Approvals R&D expenditures are adjusted for inflation Source: Tufts CSDD Approved NCE Database, Ph. RMA, 2005

Recent Productivity Decline in the Drug Industry: Is this a Unique Phenomenon? “In 1960 the trade press of the U. S. drug industry began to refer to the last few years as constituting a “research gap, ” commenting that the flow of important new drug discoveries has for some inexplicable reason diminished. ” Source: U. S. Senate, Report of the Subcommittee on Antitrust and Monopoly, 87 th Congress, 1 st Session, “Study of Administered Prices in the Drug Industry, ” June 27, 1961, p. 136

Pharmaceutical R&D Costs and Returns

Opportunity Cost for Investments u Consider two investment projects, A and B u Both projects require the same out-of- pocket expenditure (say, $400 million) u However, returns to A are realized immediately, but investors must wait 10 years before returns to B are realized u Rational investors would conclude that B is effectively much costlier than A

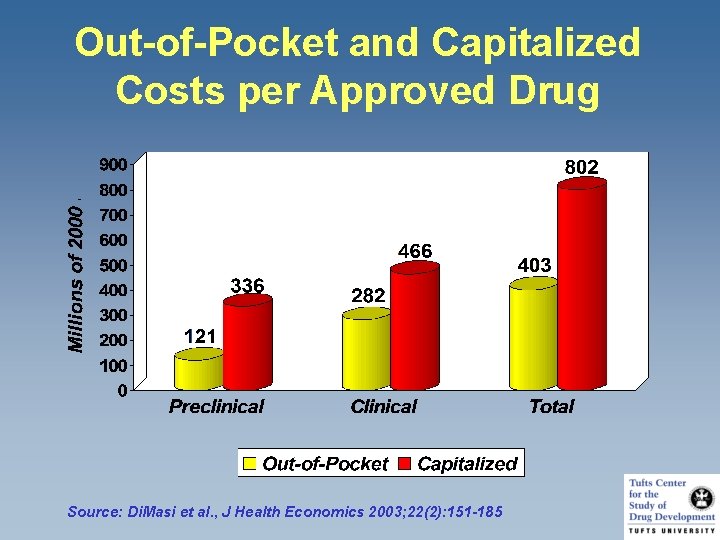
Out-of-Pocket and Capitalized Costs per Approved Drug Source: Di. Masi et al. , J Health Economics 2003; 22(2): 151 -185

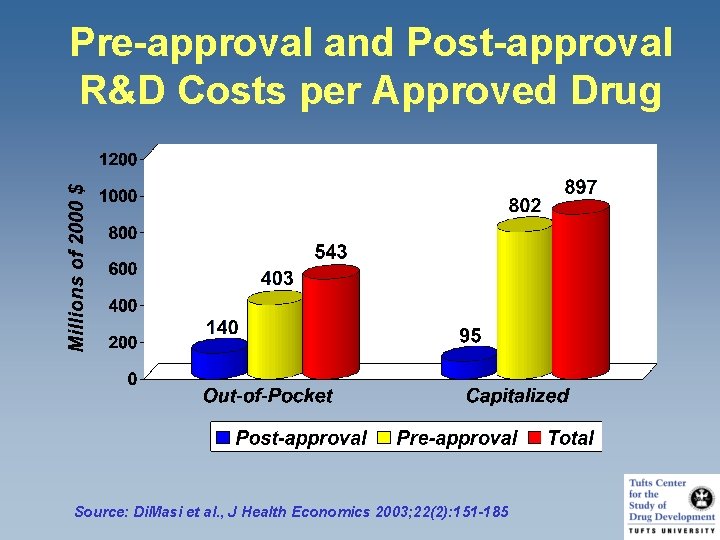
Pre-approval and Post-approval R&D Costs per Approved Drug Source: Di. Masi et al. , J Health Economics 2003; 22(2): 151 -185

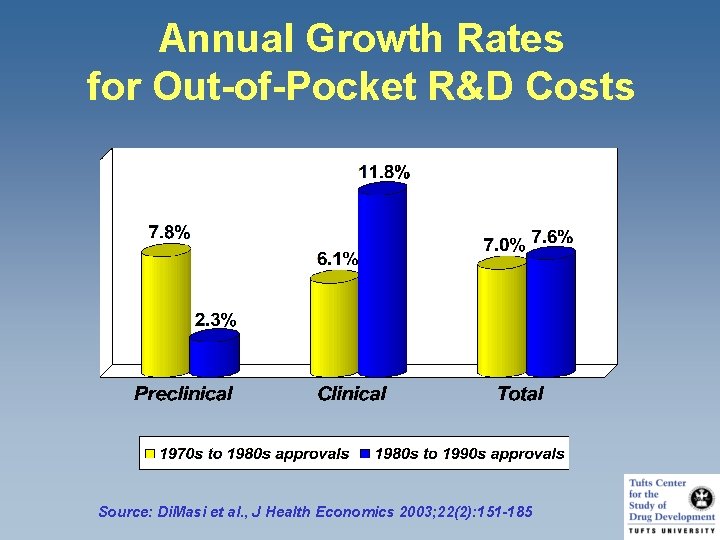
Annual Growth Rates for Out-of-Pocket R&D Costs Source: Di. Masi et al. , J Health Economics 2003; 22(2): 151 -185

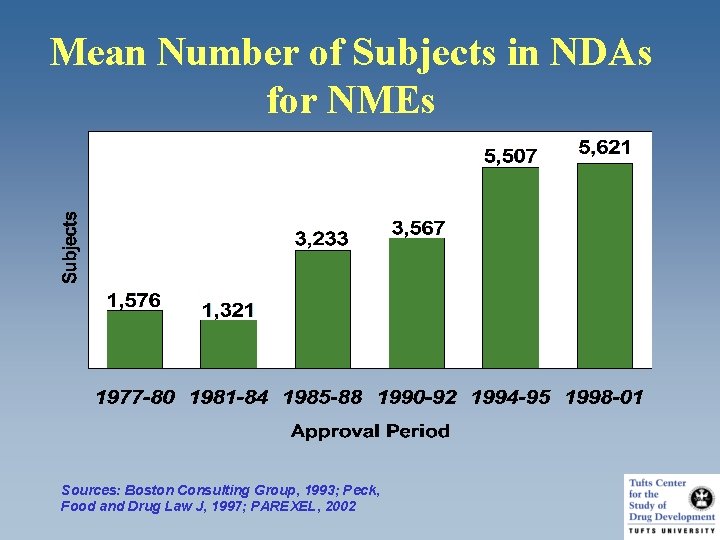
Mean Number of Subjects in NDAs for NMEs Sources: Boston Consulting Group, 1993; Peck, Food and Drug Law J, 1997; PAREXEL, 2002

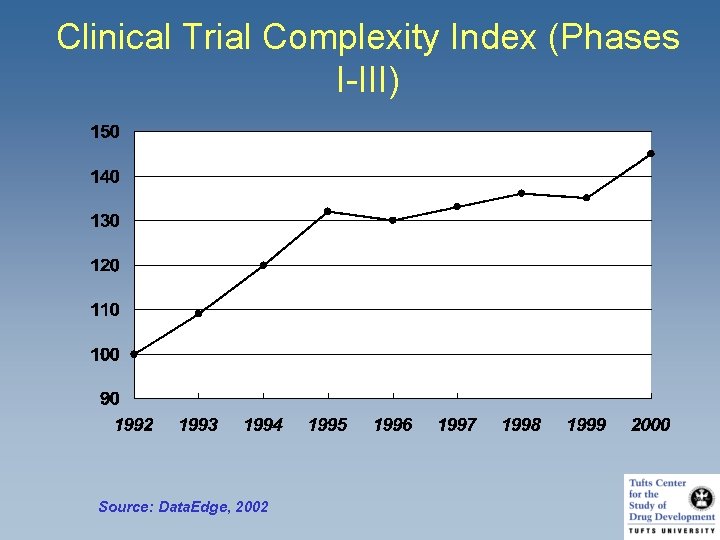
Clinical Trial Complexity Index (Phases I-III) Source: Data. Edge, 2002

Summary for R&D Costs u R&D costs have grown substantially, even in inflation-adjusted terms u The growth rate for discovery and preclinical development costs has decreased substantially u Conversely, clinical costs have grown at a much more rapid rate u New discovery and development technologies (e. g. , genomics) hold the promise of lower costs in the long-run (but perhaps higher costs in the short -run)

Summary for R&D Costs (cont. ) u Evidence and conjectures regarding factors affecting growth in clinical costs § More clinical trial subjects § Increased complexity: more procedures per patient § Patient recruitment and retention § Treatments associated with chronic and degenerative diseases § Testing against comparator drugs

Returns to New Drug Development

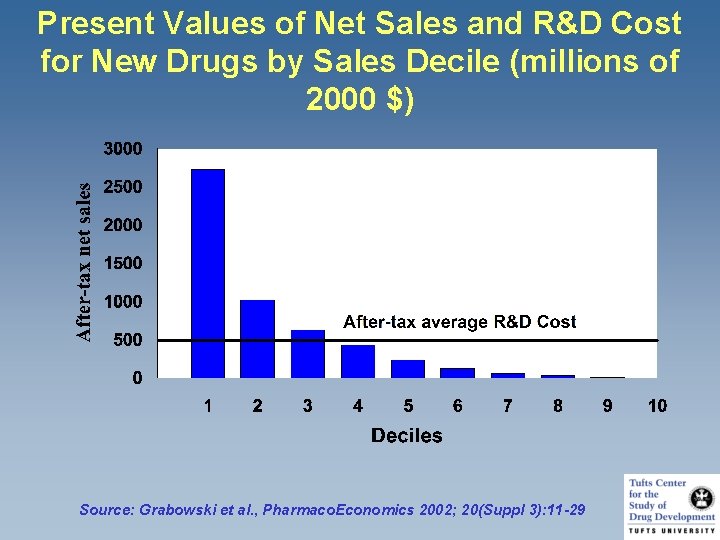
Present Values of Net Sales and R&D Cost for New Drugs by Sales Decile (millions of 2000 $) Source: Grabowski et al. , Pharmaco. Economics 2002; 20(Suppl 3): 11 -29

Biopharmaceutical R&D Costs

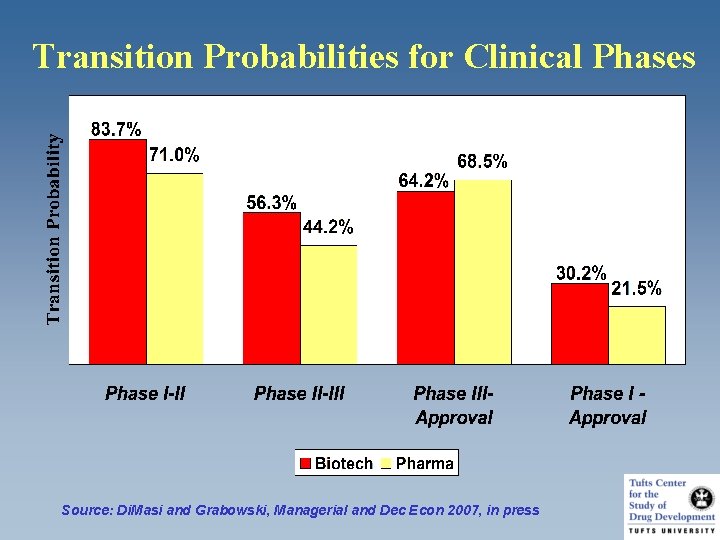
Transition Probabilities for Clinical Phases Source: Di. Masi and Grabowski, Managerial and Dec Econ 2007, in press

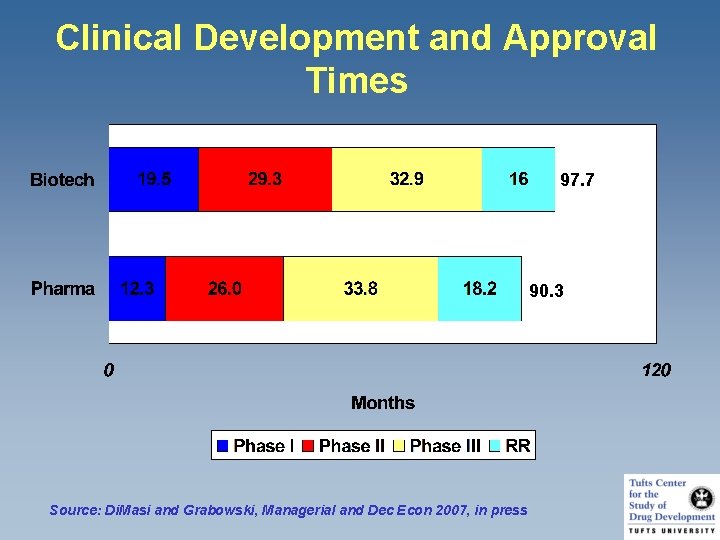
Clinical Development and Approval Times 97. 7 90. 3 Source: Di. Masi and Grabowski, Managerial and Dec Econ 2007, in press

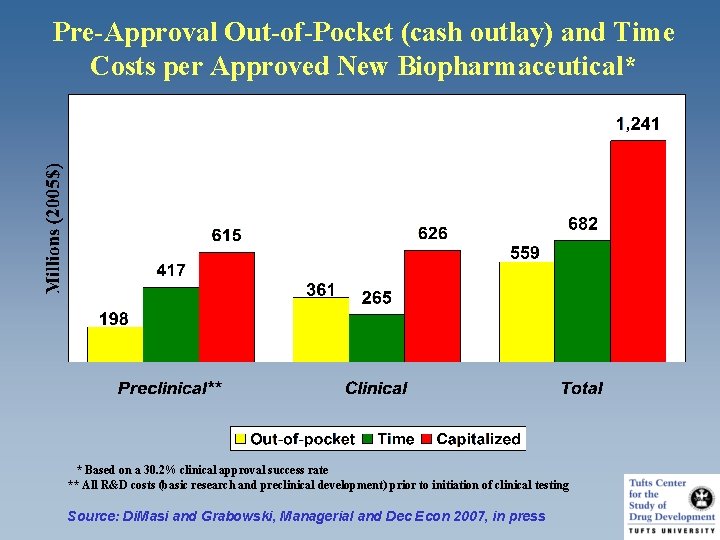
Pre-Approval Out-of-Pocket (cash outlay) and Time Costs per Approved New Biopharmaceutical* * Based on a 30. 2% clinical approval success rate ** All R&D costs (basic research and preclinical development) prior to initiation of clinical testing Source: Di. Masi and Grabowski, Managerial and Dec Econ 2007, in press

Why Might Biopharma Cost Differ? u Biotech firms may be more nimble and creative (different corporate culture) u Replacement therapies may confront fewer safety issues (more relevant to early biotech era development) u However, biotech firms have less experience in clinical development and in interacting with regulatory authorities u Manufacturing process R&D and production of clinical supplies much more expensive for biopharmaceuticals

Biopharmaceutical and Pharma R&D Costs Compared

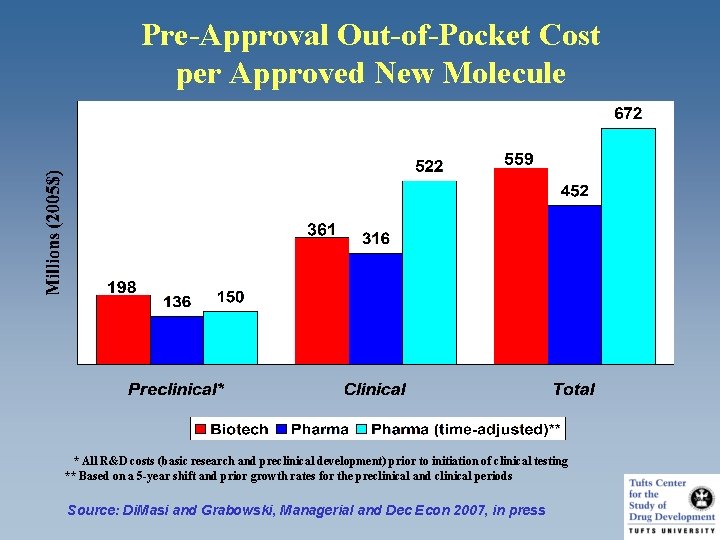
Pre-Approval Out-of-Pocket Cost per Approved New Molecule * All R&D costs (basic research and preclinical development) prior to initiation of clinical testing ** Based on a 5 -year shift and prior growth rates for the preclinical and clinical periods Source: Di. Masi and Grabowski, Managerial and Dec Econ 2007, in press

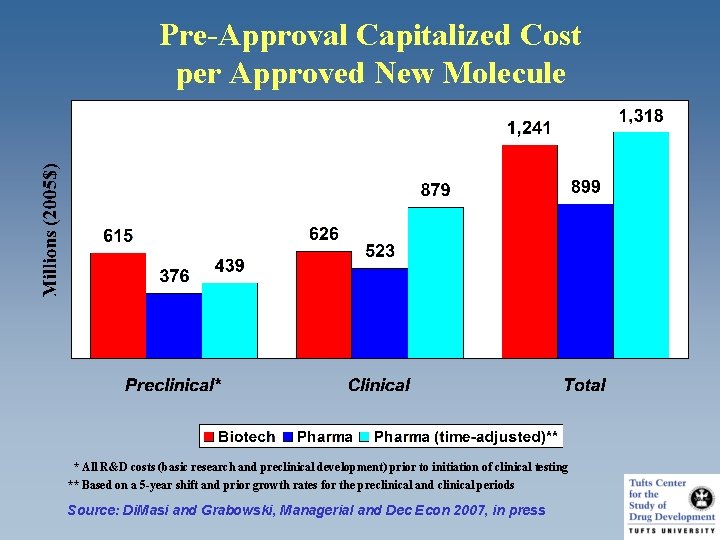
Pre-Approval Capitalized Cost per Approved New Molecule * All R&D costs (basic research and preclinical development) prior to initiation of clinical testing ** Based on a 5 -year shift and prior growth rates for the preclinical and clinical periods Source: Di. Masi and Grabowski, Managerial and Dec Econ 2007, in press

The Pace of Competitive Development

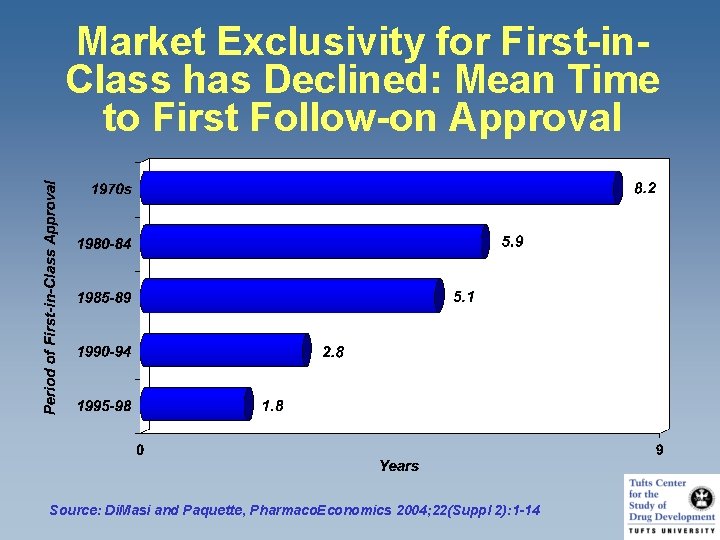
Market Exclusivity for First-in. Class has Declined: Mean Time to First Follow-on Approval Source: Di. Masi and Paquette, Pharmaco. Economics 2004; 22(Suppl 2): 1 -14

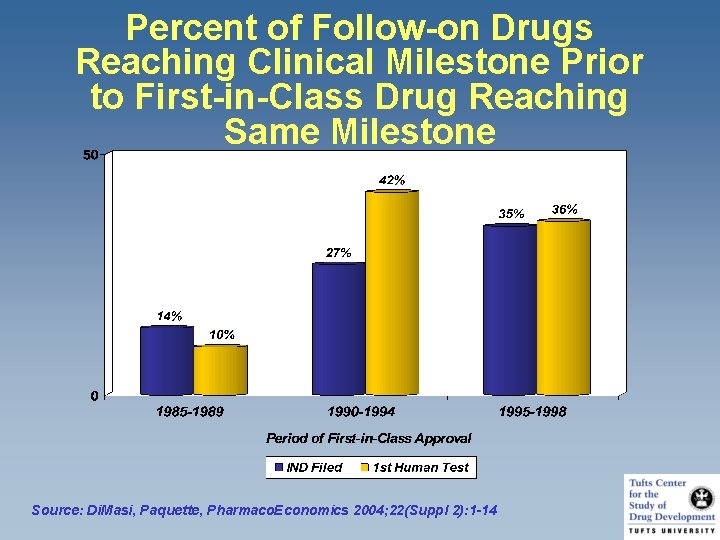
Percent of Follow-on Drugs Reaching Clinical Milestone Prior to First-in-Class Drug Reaching Same Milestone Source: Di. Masi, Paquette, Pharmaco. Economics 2004; 22(Suppl 2): 1 -14

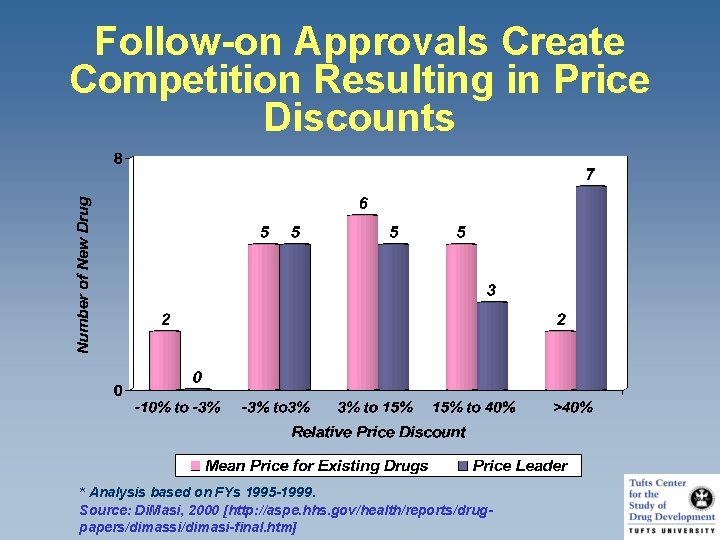
Follow-on Approvals Create Competition Resulting in Price Discounts * Analysis based on FYs 1995 -1999. Source: Di. Masi, 2000 [http: //aspe. hhs. gov/health/reports/drugpapers/dimassi/dimasi-final. htm]

Impact of Improvements in Drug Development Productivity

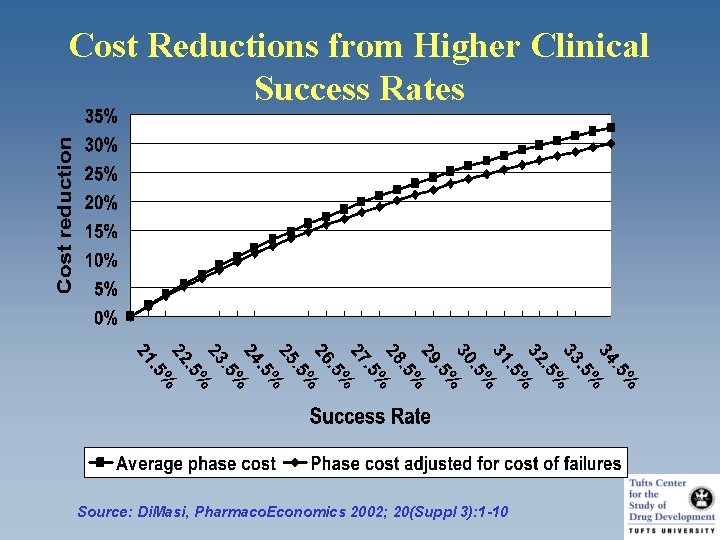
Cost Reductions from Higher Clinical Success Rates Source: Di. Masi, Pharmaco. Economics 2002; 20(Suppl 3): 1 -10

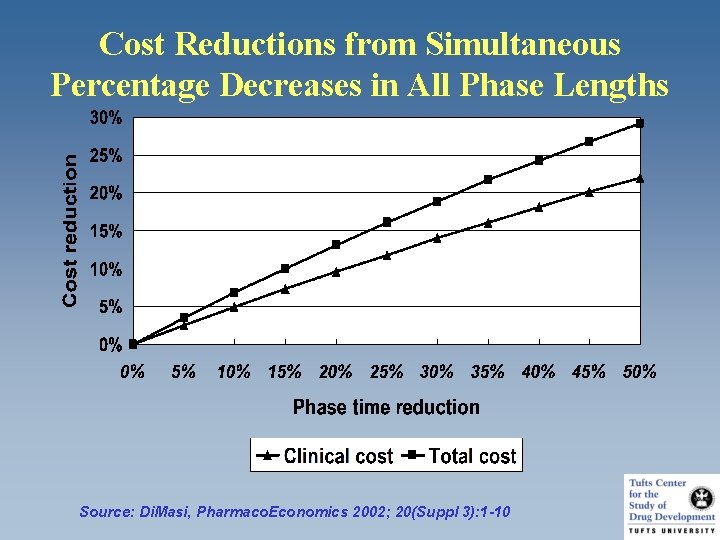
Cost Reductions from Simultaneous Percentage Decreases in All Phase Lengths Source: Di. Masi, Pharmaco. Economics 2002; 20(Suppl 3): 1 -10

Trends in Drug Development Pipelines

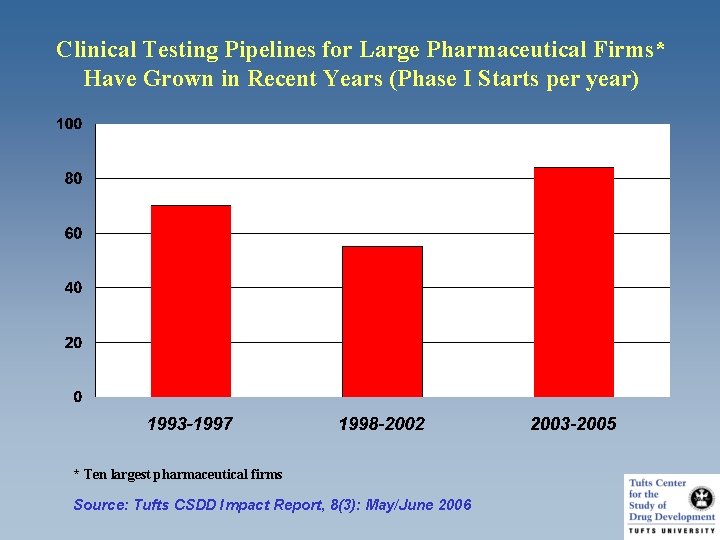
Clinical Testing Pipelines for Large Pharmaceutical Firms* Have Grown in Recent Years (Phase I Starts per year) * Ten largest pharmaceutical firms Source: Tufts CSDD Impact Report, 8(3): May/June 2006

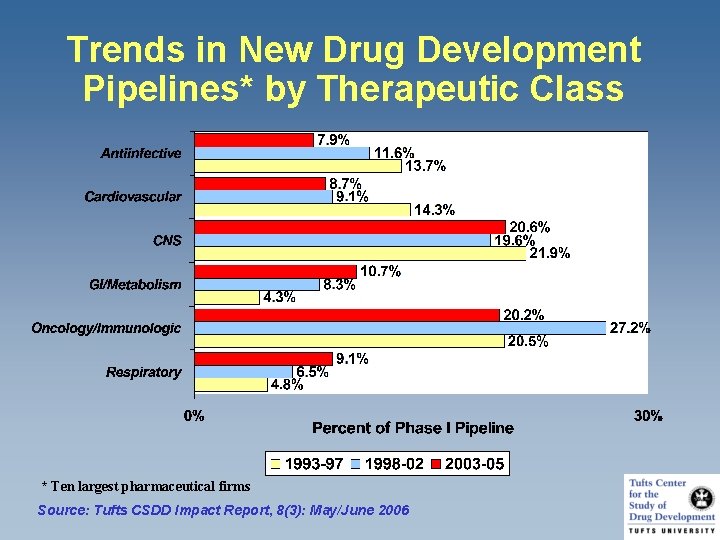
Trends in New Drug Development Pipelines* by Therapeutic Class * Ten largest pharmaceutical firms Source: Tufts CSDD Impact Report, 8(3): May/June 2006

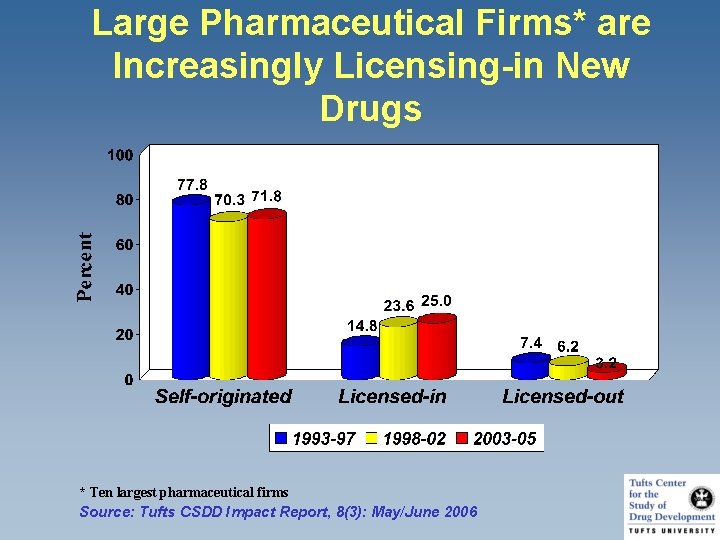
Large Pharmaceutical Firms* are Increasingly Licensing-in New Drugs * Ten largest pharmaceutical firms Source: Tufts CSDD Impact Report, 8(3): May/June 2006

Conclusions u Drug development has been and still is costly, risky, and lengthy u Periods of market exclusivity have shrunk for first-in-class drugs u The potential payoffs for improvements in the development process are substantial u After a period of decline, more new drugs are now entering clinical testing pipelines