Measuring Matter Avogadros Number Mole Molar Mass How

Measuring Matter Avogadro’s Number Mole Molar Mass

How Scientists Keep Track of Atoms �A ream of paper is 500 sheets: ◦ 1 ream = 500 sheets �A counting unit appropriate for objects based on their size/function ◦ Always constant �Easier to sell by ream than by sheet �Scientist could count atoms ◦ Atoms are extremely small �The number of individual particles even in a small sample is REALLY SMALL!

The Mole � A ‘chemical’ counting unit � SI base unit � Measures amount of a pure substance � Abbreviated ‘mol’ � Scientist developed � One mole contains Avogadro’s number of particles ◦ This number is named in honor of Amedeo Avogadro (1776 – 1856), 1856) who studied quantities of gases and discovered that no matter the identity of the gas, there were the same number of molecules present

How much is the mole? �Similar to a dozen except instead of 12, �A mole = 602 000 000 or 6. 02 x 1023 = Avogadro’s Number. �It is equal to that number no matter what kind of particles: �Atoms �Molecules �Formula unit

How Do We Use The Mole? �We’d never use the mole to describe macroscopic or real world objects. � 1 mole (6. 02 x 1023) of watermelon seeds would be found inside a watermelon the size of the moon. � 1 mole (6. 02 x 1023) of donut holes would cover the earth and would be 5 miles deep.

How do we use the mole? �Mole is a huge number of particles ◦ only used to describe the amount of things that are very, very small. ◦ 1 mole (6. 02 x 1023) of water molecules would barely fill a shot glass

A Mole of Particles Contains 6. 02 x 1023 particles 1 mole C = 6. 02 x 1023 C atoms 1 mole H 2 O = 6. 02 x 1023 H 2 O molecules 23 Na. Cl formula units = 6. 02 x 10 1 mole Na. Cl

Avogadro’s Number as Conversion Factor 6. 02 x 1023 particles 1 mole or 1 mole 6. 02 x 1023 particles Note: a particle could be an atom, a molecule or a formula unit

Practice How many moles of silver are in 3. 01 x 1023 atoms of silver? 3. 01 x 1023 atoms Ag Given: Find: Moles of Ag 6. 02 x 1023 atoms = 1 mol Ag 3. 01 x 1023 Ag atoms 1 mol Ag 6. 02 x 1023 Ag atoms = 0. 5 mol Ag = 0. 500 mol Ag

Molar Mass �The mass of one mole of a pure substance UNIT: (g/mol) �Equal to the numerical value of the average atomic mass (PT) 1 mole of C atoms = 12. 01 g 1 mole of Mg atoms = 24. 31 g 1 mole of Cu atoms = 63. 55 g

Other Names Related to Molar Mass � Molecular � Formula �THE Mass/Molecular Weight Mass/Formula Weight: POINT: You may hear all of these terms which mean the SAME Number from the periodic table!!

Find the molar mass A. 1 mole of Br B. 1 mole of Sn Learning Check! = = 79. 90 g 118. 71 g

Molar Mass of Compounds Mass in grams of 1 mole equal numerically to the sum of the atomic masses 1 mole of Ca. Cl 2 = 110. 98 g Ca. Cl 2 1 mole Ca x 40. 08 g/mol + 2 moles Cl x 35. 45 g/mol 1 mole of N 2 O 4 = 92. 02 g = 70. 90 g/mol

Molar Mass of Compounds Mass in grams of 1 mole equal numerically to the sum of the atomic masses 1 mole of Ca. Cl 2 =
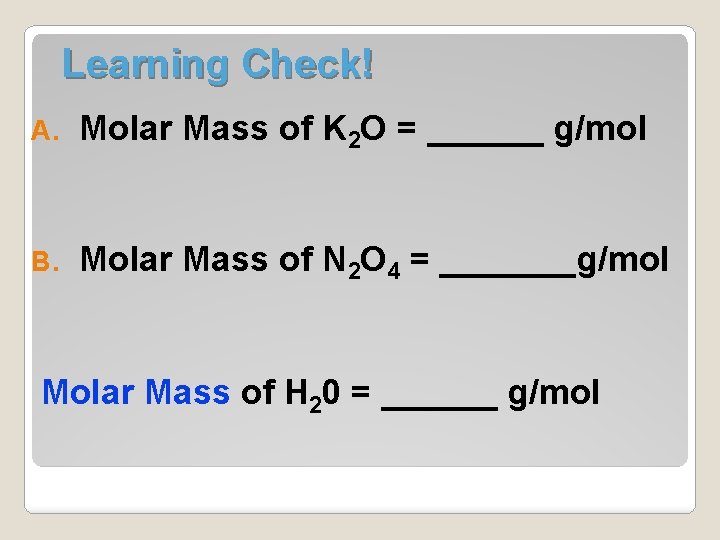
Learning Check! A. Molar Mass of K 2 O = ______ g/mol B. Molar Mass of N 2 O 4 = _______g/mol Molar Mass of H 20 = ______ g/mol

Grams molar mass Moles Calculations with Molar Mass

Practice Aluminum is often used for the structure of lightweight bicycle frames. How many grams of Al are in 9. 60 moles of Al? G: F: 9. 60 mol Al g of Al 1 mol Al = 26. 98 g
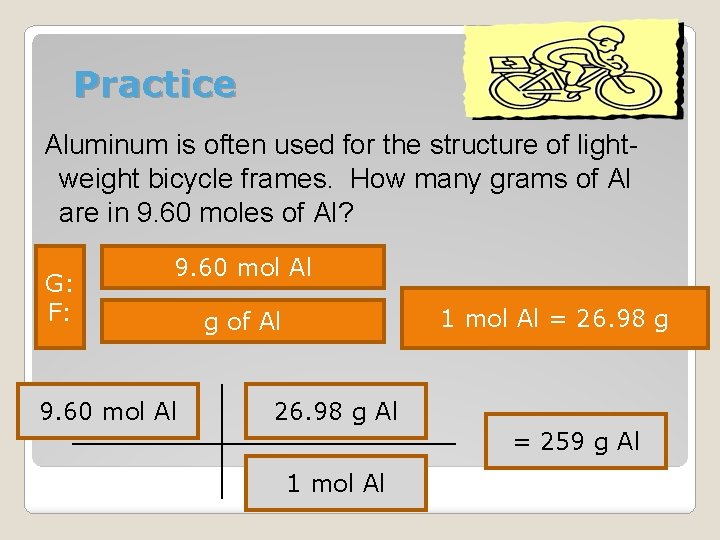
Practice Aluminum is often used for the structure of lightweight bicycle frames. How many grams of Al are in 9. 60 moles of Al? G: F: 9. 60 mol Al 1 mol Al = 26. 98 g g of Al 26. 98 g Al = 259 g Al 1 mol Al

Practice Propane gas is needed to operate gas grills. Your father used 110. 6 grams of propane to grill burgers over the weekend. How many moles of propane did he use up? Propane = C 3 H 8 G: F: 110. 6 g C 3 H 8 mol of C 3 H 8 1 mol. C 3 H 8 = 44. 11 g

Practice Propane gas is needed to operate gas grills. Your father used 110. 6 grams of propane to grill burgers over the weekend. How many moles of propane did he use up? Propane = C 3 H 8 G: F: 110. 6 g C 3 H 8 mol of C 3 H 8 110. 6 g. C 3 H 8 1 mol C 3 H 8 44. 11 g C 3 H 8 1 mol. C 3 H 8 = 44. 11 g = 2. 507 mol C 3 H 8

Remember: There may not always be a direct pathway to the final answer!! molar mass Grams Avogadro’s number Moles particles Calculations using Molar Mass & Avogadro’s Number
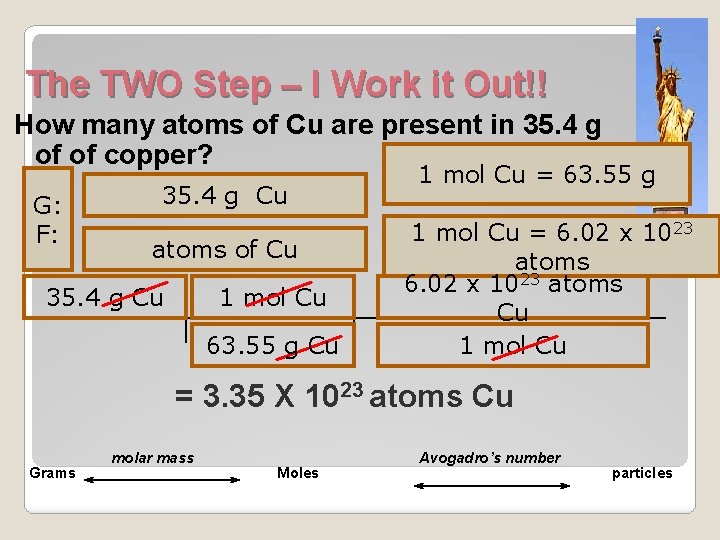
The TWO Step – I Work it Out!! How many atoms of Cu are present in 35. 4 g of of copper? G: F: 35. 4 g Cu atoms of Cu 35. 4 g Cu 1 mol Cu 63. 55 g Cu 1 mol Cu = 63. 55 g 1 mol Cu = 6. 02 x 1023 atoms Cu 1 mol Cu = 3. 35 X 1023 atoms Cu Grams molar mass Moles Avogadro’s number particles

The TWO Step – I Work it Out!! How many atoms of Cu are present in 35. 4 g of of copper? G: F: Grams 35. 4 g Cu atoms of Cu molar mass Moles 1 mol Cu = 63. 55 g 1 mol Cu = 6. 02 x 1023 atoms Avogadro’s number particles

Learning Check! What is the mass (in grams) of 1. 20 X 1024 molecules of glucose (C 6 H 12 O 6)? Grams molar mass Moles Avogadro’s number particles

Now you work it out! How many atoms of O are present in 78. 1 g of oxygen? 78. 1 g O 2 1 mol O 2 6. 02 X 1023 molecules O 2 2 atoms O 32. 0 g O 2 1 molecule O 2
- Slides: 25