Measuring mass Mass describes the amount of matter

Measuring mass ØMass describes the amount of matter in an object. ØThe SI unit for mass is the kilogram (kg). ØThe kilogram is too large a unit to be convenient for small masses. One gram (g) is one-thousandth of a kilogram. What is the estimated mass of ONE zinc nut?

Matter ØMatter is anything that has mass and takes up space. ØAll matter has mass. ØSteel, plastic, rubber, and glass are different kinds of matter. A car has a lot more of each kind of matter than a bike.

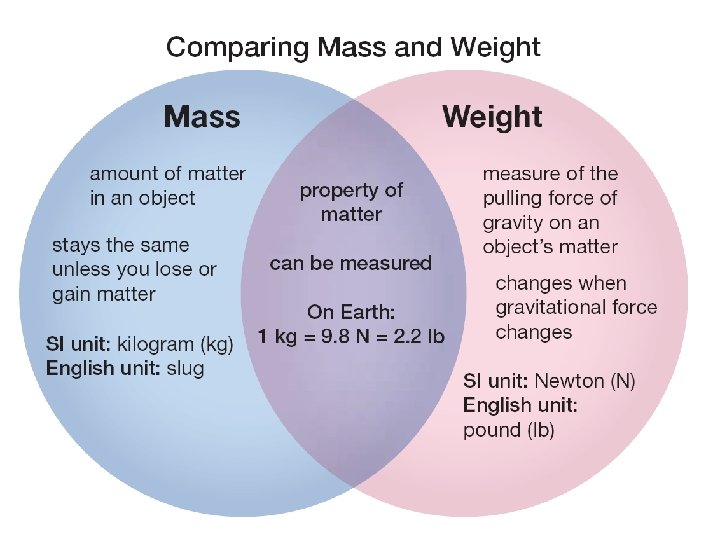
Mass and weight are different Ø We tend to use the terms mass and weight interchangeably, but they are not the same thing. Ø Mass is the amount of matter in an object. Ø Weight is a measure of the pulling force of gravity on an object.

Mass and weight are different Ø A 2. 3 kg bag of flour has a mass of 2. 3 kilograms no matter where it is in the universe. Ø The weight of the bag of flour is less on the moon. The 5 lb bag of flour on Earth weighs only. 8 lbs on the moon!

Volume ØVolume is the amount of space an object takes up. ØThe fundamental unit of volume in SI is the cubic meter (m 3). ØMore convenient smaller units are cubic centimeters (cc or cm 3), liters (L) and milliliters (m. L).

Volume ØMeasuring the volume of liquids is easy. ØPour the liquid into a graduated cylinder and read the meniscus at eye level.

Displacement ØYou can find the volume of an irregular shape using a technique called displacement. ØPut the irregularly shaped object in water and measuring the amount of water displaced.

Comparing mass and volume ØMass and volume are two different properties of matter. ØSize does not always indicate an object’s mass! ØHow the matter is packed into space is more important.

Review Questions pg. 35 ØAnswer the following questions on page 35 Ø#’s 1, 3, 5, 6

Density ØDensity describes how much mass is in a given volume of a material.

ØSolids, liquids and gases are matter, so they all have density. ØThe density of water is about one gram per cubic centimeter.
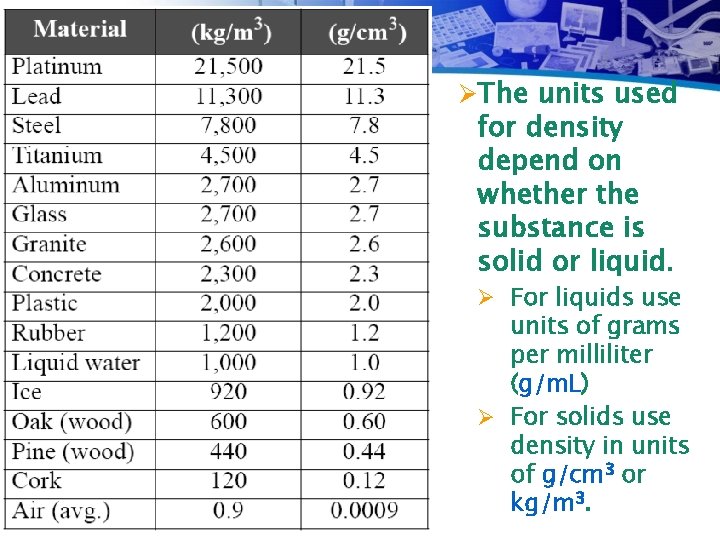
ØThe units used for density depend on whether the substance is solid or liquid. Ø For liquids use units of grams per milliliter (g/m. L) Ø For solids use density in units of g/cm 3 or kg/m 3.
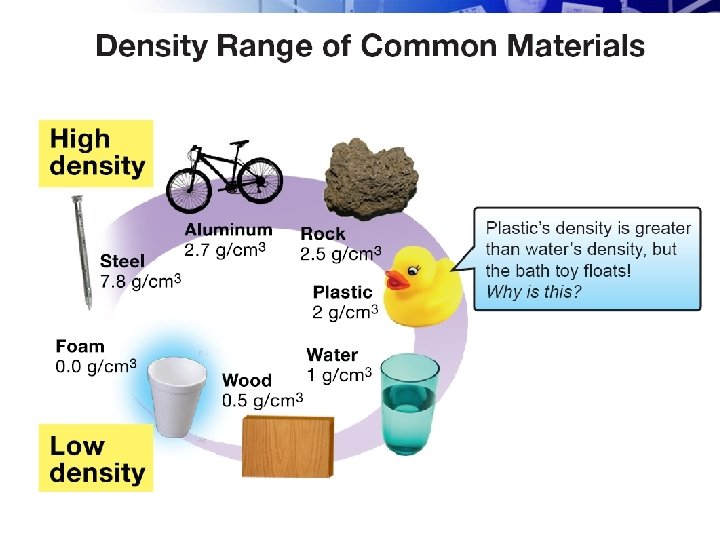

ØDensity is a property of material independent of quantity or shape.

ØLiquids tend to be less dense than solids of the same material. Ø Ex. solder (“sodder)

ØWater is an exception to this rule. ØThe density of solid water (ice) is less than the density of liquid water.

Ø To find the density of a material, you need to know the mass and volume of a solid sample of the material. 1. Mass is measured with a balance or scale. 2. Use the displacement method or calculate the volume.

Density changes for different substances because: 1. Atoms have different masses. 2. Atoms may be “packed” tightly or loosely.


Calculating Density 1. Looking for: Ø …the density of the candle 2. Given: Ø …mass = 1500 g; volume = 1700 m. L 3. Relationship: Ø D = m/V 4. Solution: Ø 1, 500 g ÷ 1, 700 m. L = 0. 8823529 g/m. L # Sig. fig =. 88 g/m. L

Answer Review Questions pg. 41 ØNumbers : 1, 2, 6, 7
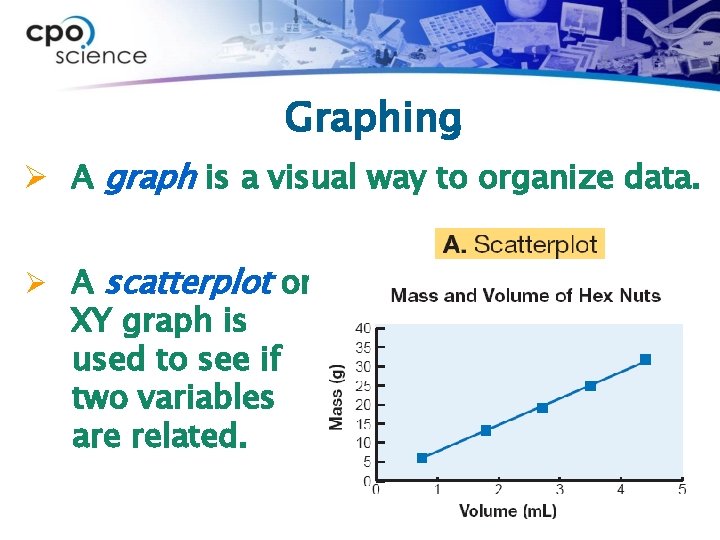
Graphing Ø A graph is a visual way to organize data. Ø A scatterplot or XY graph is used to see if two variables are related.
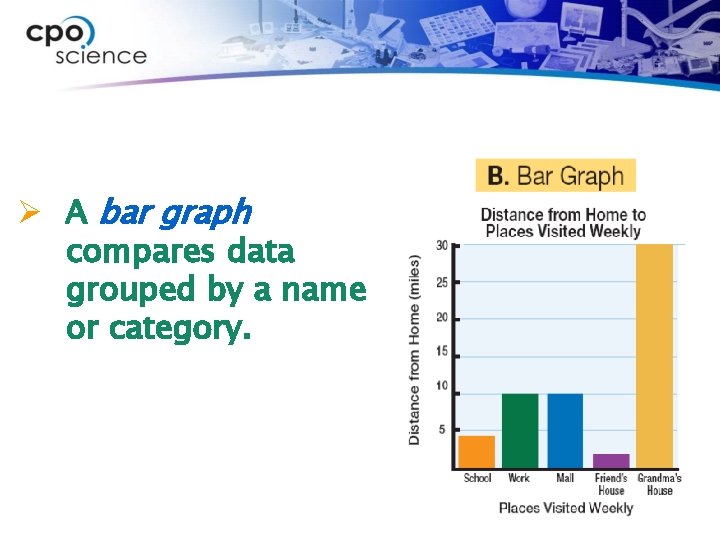
Ø A bar graph compares data grouped by a name or category.

Ø A pie graph shows the amount each part makes of up of the whole (100%).
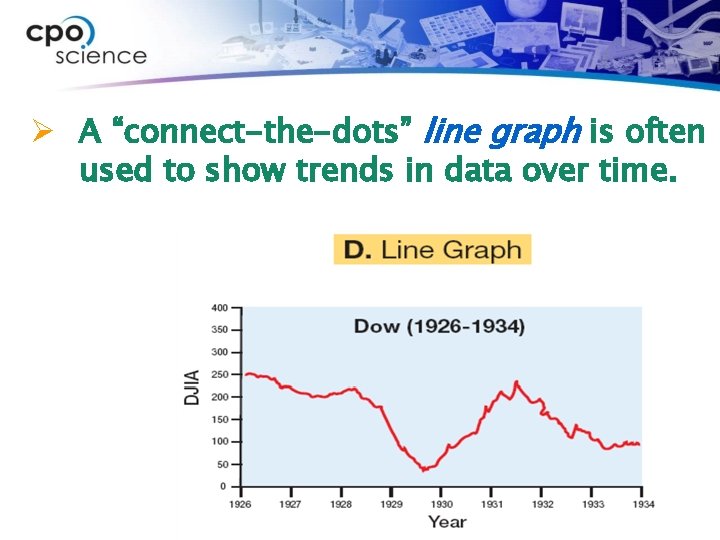
Ø A “connect-the-dots” line graph is often used to show trends in data over time.
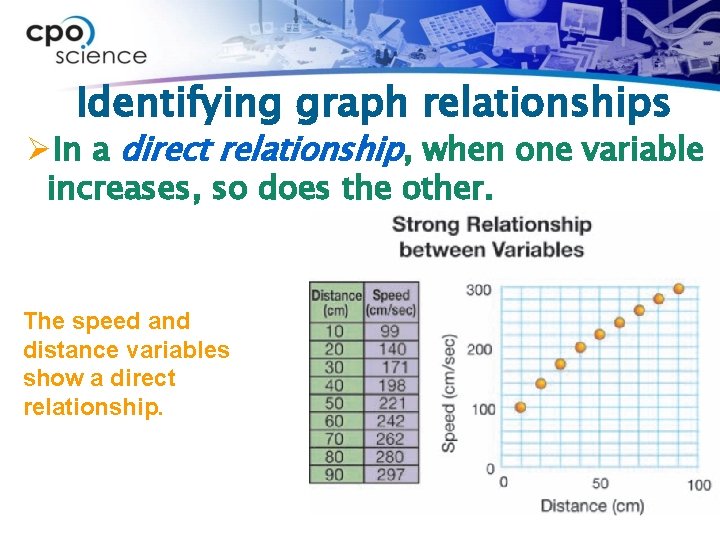
Identifying graph relationships ØIn a direct relationship, when one variable increases, so does the other. The speed and distance variables show a direct relationship.
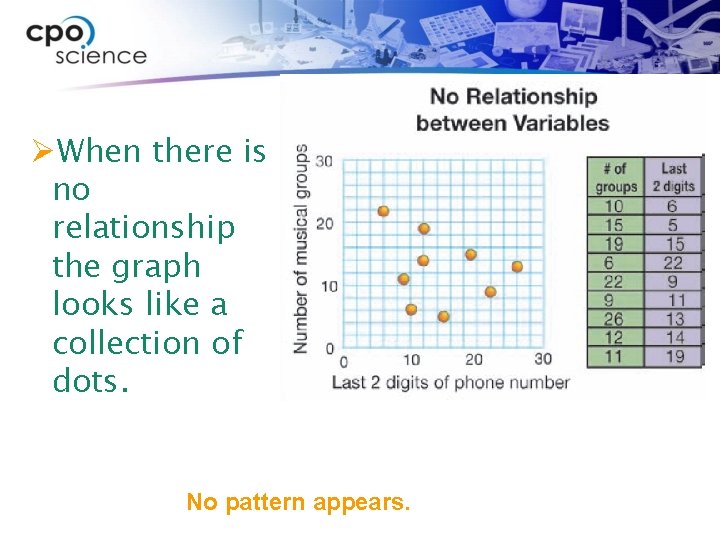
ØWhen there is no relationship the graph looks like a collection of dots. No pattern appears.
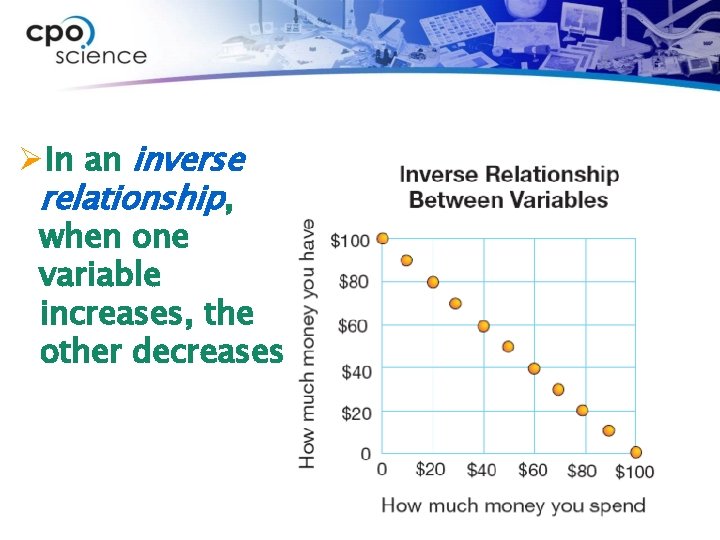
ØIn an inverse relationship, when one variable increases, the other decreases.

ØAnswer questions on pg. 47 numbers 1, 2, 4, 5
- Slides: 30