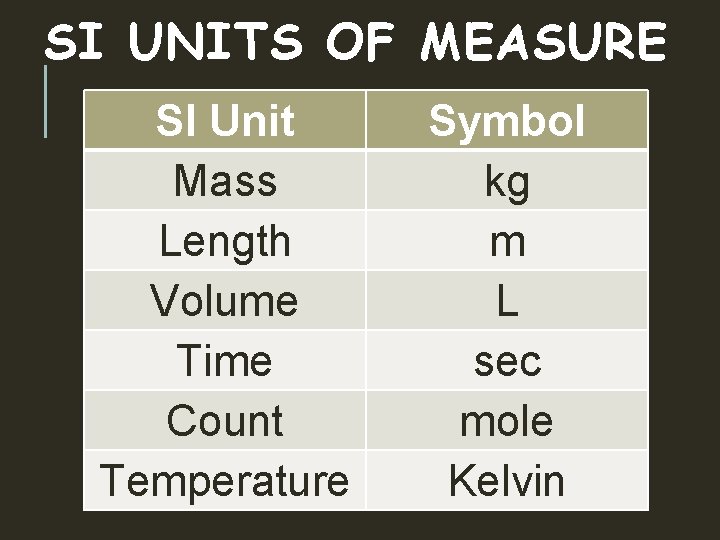
MEASUREMENTS SI UNITS OF MEASURE SI Unit Mass





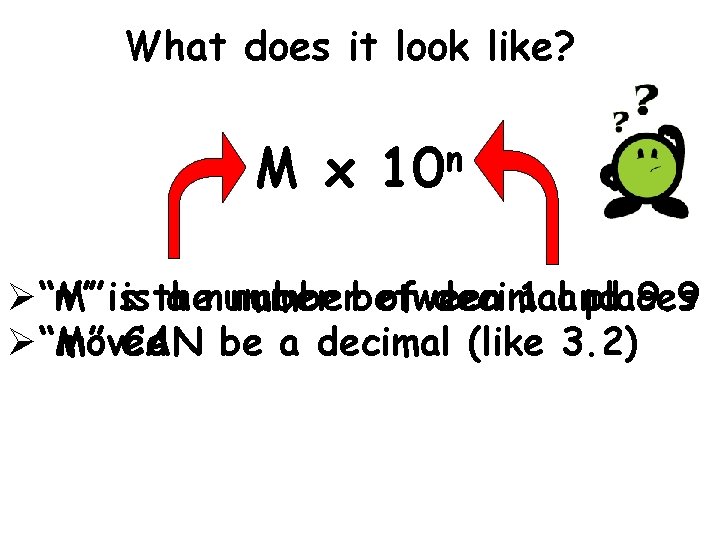

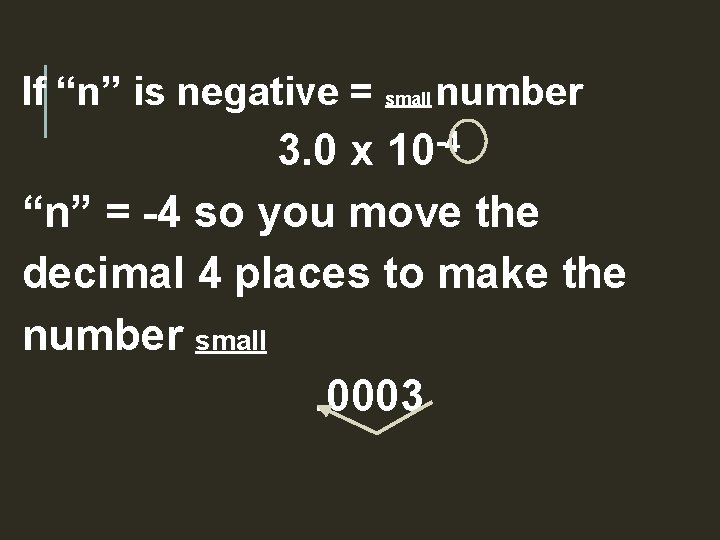








- Slides: 20

MEASUREMENTS

SI UNITS OF MEASURE SI Unit Mass Length Volume Time Count Temperature Symbol kg m L sec mole Kelvin

READING MEASUREMENTS Always read one place past the graduation markings. This value is called an estimated digit.

METRIC CONVERSIONS M. . k h D b d c m. . μ. . n. . P King Henry Died by drinking chocolate milk

SCIENTIFIC NOTATION

WHY DO WE USE IT? To work with LARGE numbers like: 602, 000, 000, 000 To work with small numbers like: 0. 000 000 920 It’s easier (and less confusing) to put them in scientific notation
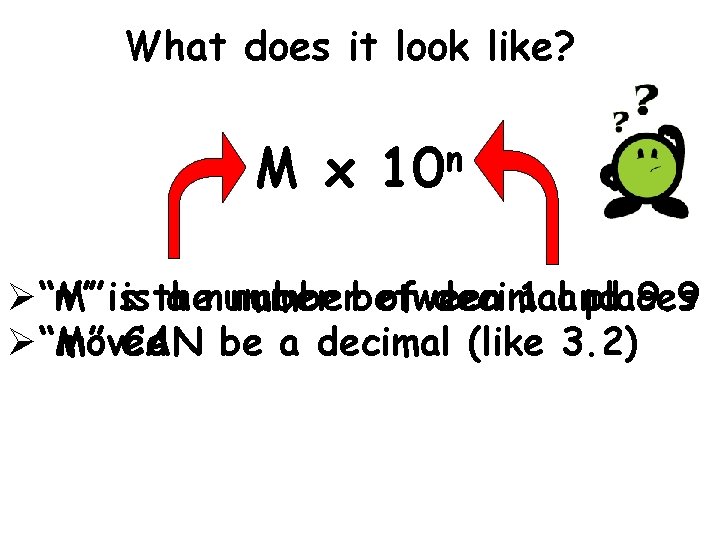
What does it look like? M x n 10 Ø“M” “n” isis the a numberbetween of decimal 1 and places 9. 9 Ø“M” moved CAN be a decimal (like 3. 2)

WHAT DO YOU MEAN… MOVE THE DECIMAL? ? ? If “n” is positive = BIG number 3. 0 x 104 “n” = 4 so you move the decimal 4 places to make the number BIG 30000.
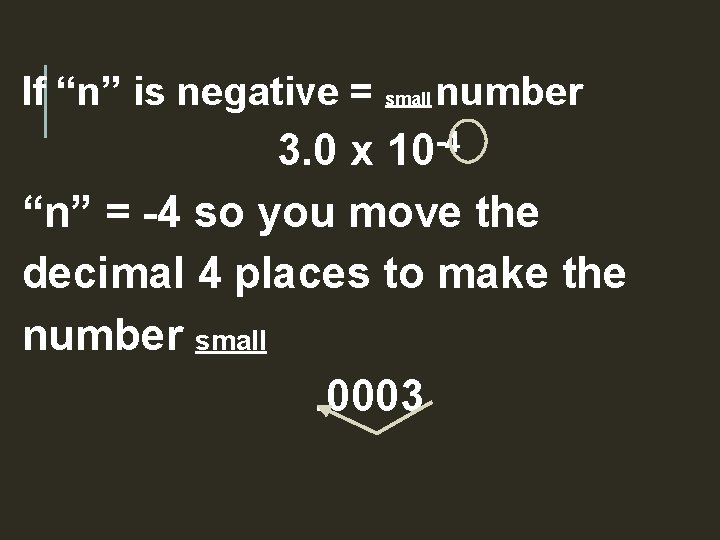
If “n” is negative = small number 3. 0 x 10 -4 “n” = -4 so you move the decimal 4 places to make the number small. 0003

LET’S PRACTICE. . . 1) 7 x 103 = 2) 6. 8 x 106 = 3) 1. 356 x 10 -4 = 4) 7. 42 x 10 -3 = 7, 000 6, 800, 0001356. 00742

HOW ABOUT IN REVERSE? Can you change a number like 50, 000 into scientific notation?

FIRST Place a decimal in the number that would make it between 1 and 9. 9 In this case: 5. 0

SECOND Look at the original number: Is it big or small? ? ? If it’s LARGER than 1 positive “n” If it’s SMALLER than 1 negative “n”

LAST How many places did you move the decimal point? this will be your “n” The decimal moved from: 5. 0000 It moved 4 places, so the exponent is 4 5. 0 x 104

LET’S TRY THESE! 1) 2) 3) 6, 800, 000 6. 8 x 106 13, 430, 000 1. 343 x 107 0. 03459 3. 459 x 10 -2

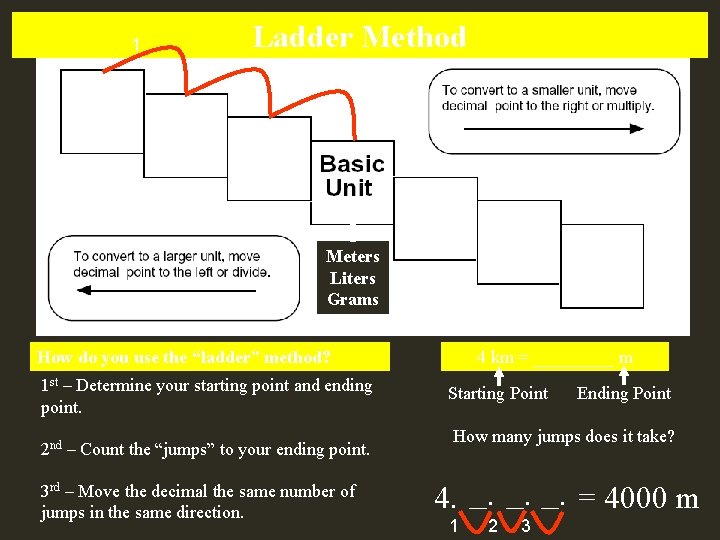
Metric Conversions Ladder Method T. Trimpe 2008 http: //sciencespot. net/

Ladder Method 1 2 KILO 1000 Units HECTO 100 Units 3 DEKA 10 Units Meters Liters Grams DECI 0. 1 Unit How do you use the “ladder” method? 1 st – Determine your starting point and ending point. 2 nd – Count the “jumps” to your ending point. 3 rd – Move the decimal the same number of jumps in the same direction. CENTI 0. 01 Unit MILLI 0. 001 Unit 4 km = _____ m Starting Point Ending Point How many jumps does it take? 4. __. __. = 4000 m 1 2 3

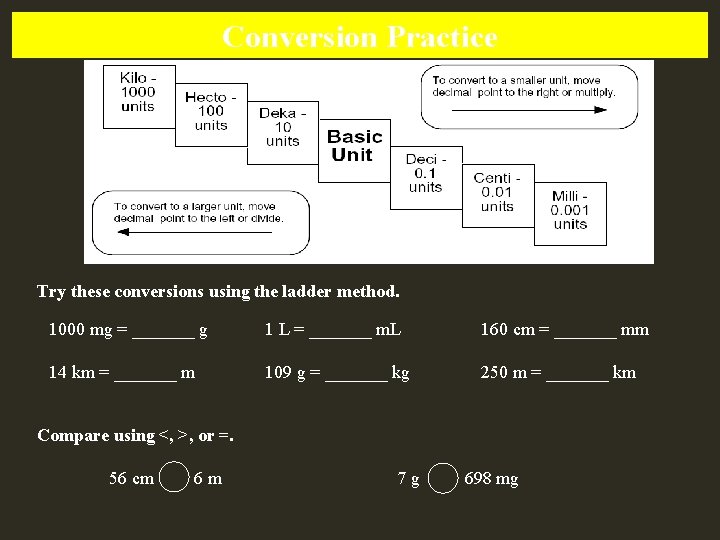
Conversion Practice Try these conversions using the ladder method. 1000 mg = _______ g 1 L = _______ m. L 160 cm = _______ mm 14 km = _______ m 109 g = _______ kg 250 m = _______ km Compare using <, >, or =. 56 cm 6 m 7 g 698 mg

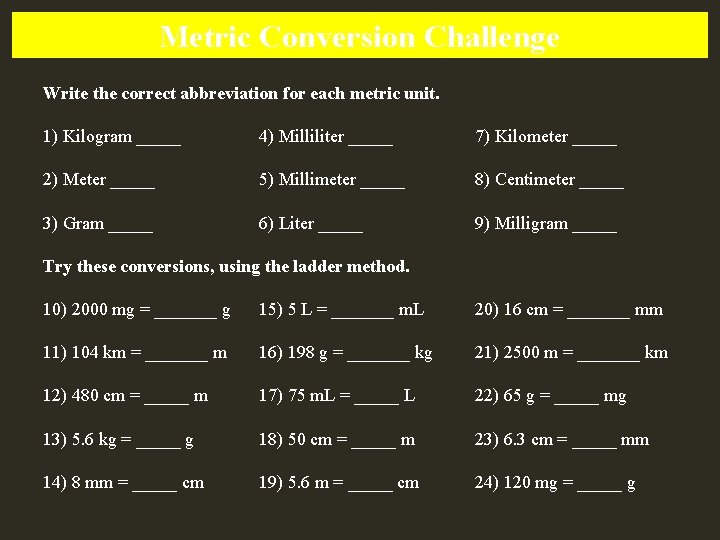
Metric Conversion Challenge Write the correct abbreviation for each metric unit. 1) Kilogram _____ 4) Milliliter _____ 7) Kilometer _____ 2) Meter _____ 5) Millimeter _____ 8) Centimeter _____ 3) Gram _____ 6) Liter _____ 9) Milligram _____ Try these conversions, using the ladder method. 10) 2000 mg = _______ g 15) 5 L = _______ m. L 20) 16 cm = _______ mm 11) 104 km = _______ m 16) 198 g = _______ kg 21) 2500 m = _______ km 12) 480 cm = _____ m 17) 75 m. L = _____ L 22) 65 g = _____ mg 13) 5. 6 kg = _____ g 18) 50 cm = _____ m 23) 6. 3 cm = _____ mm 14) 8 mm = _____ cm 19) 5. 6 m = _____ cm 24) 120 mg = _____ g

Compare using <, >, or =. 25) 63 cm 26) 536 cm 6 m 53. 6 dm 27) 5 g 28) 43 mg 508 mg 5 g 29) 1, 500 m. L 30) 3. 6 m 1. 5 L 36 cm