Measurements make observations meaningful International System of Units

Measurements make observations meaningful

International System of Units (SI Units) • Uses the metric system – Based on units of 10
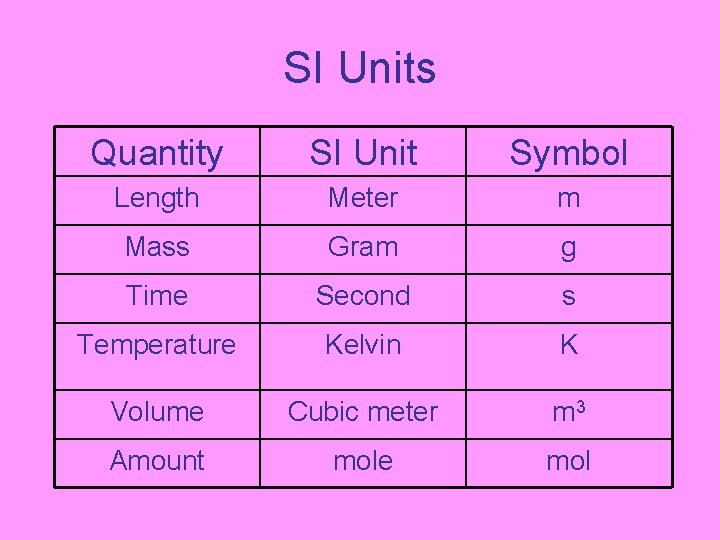
SI Units Quantity SI Unit Symbol Length Meter m Mass Gram g Time Second s Temperature Kelvin K Volume Cubic meter m 3 Amount mole mol

Mass • Mass and weight are NOT the same thing • Weight is dependant upon gravity, mass is not

Temperature • Kelvin is the SI unit, but Celsius (C) is often used • K = C + 273 Practice: 1. Convert 25 o. C to K. 2. Convert 352 K to o. C. 3. Convert -15 o. C to K. • Heat and temperature are not the same thing – – Heat is a type of energy Temperature is a measurement of energy

Important Temperatures • Freezing Points for water Ø 0 o. C = 273 K = 32 o. F • Boiling Points for water Ø 100 o. C = 373 K = 212 o. F • Absolute zero Ø 0 K (-273 o. C) ØTheoretically, all movement stops at this temperature
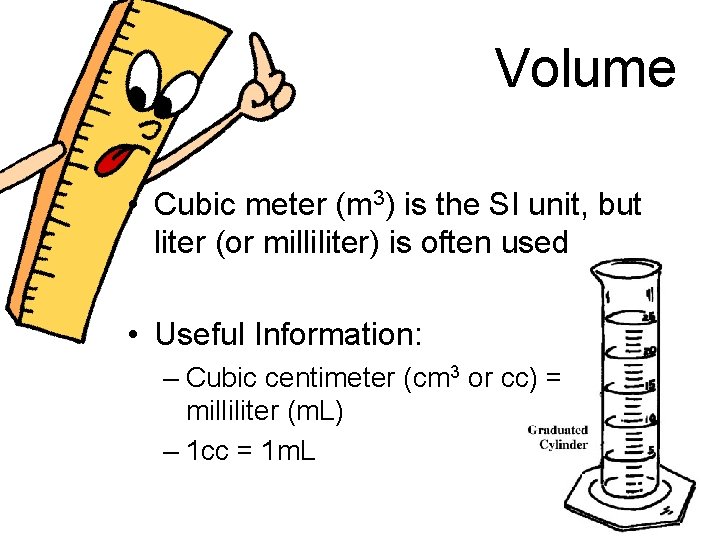
Volume • Cubic meter (m 3) is the SI unit, but liter (or milliliter) is often used • Useful Information: – Cubic centimeter (cm 3 or cc) = milliliter (m. L) – 1 cc = 1 m. L

Moles • Used to measure the amount (quantity) of something – 1 mole = 6. 02 x 1023 particles

Density • • • How much “stuff” in a given area Density of water (at 250 C) = 1. 00 g/m. L D = m/v

Density Practice 1. A rock has a mass of 3. 5 kg and a volume of 7. 0 m 3. What is the rock’s density? 2. An object’s density is 8. 0 g/cm 3 and its mass is 1. 5 g. What is the object’s volume? 3. What would the mass be of a 25 m. L sample of an object with a density of 0. 047 g/m. L?
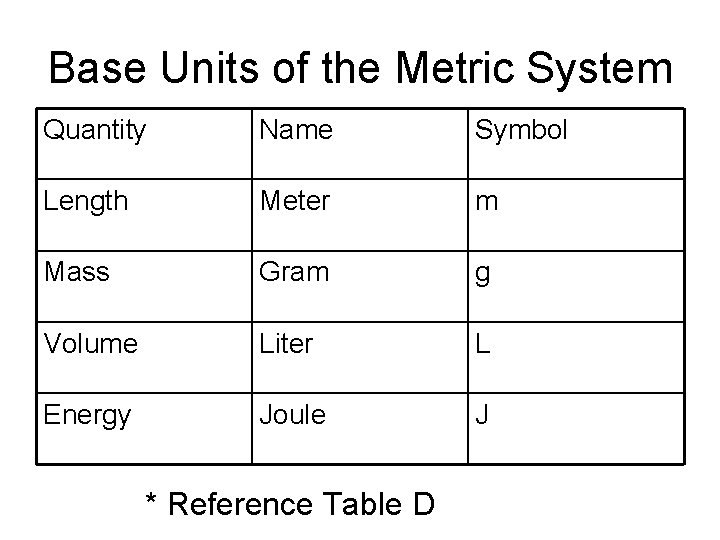
Base Units of the Metric System Quantity Name Symbol Length Meter m Mass Gram g Volume Liter L Energy Joule J * Reference Table D
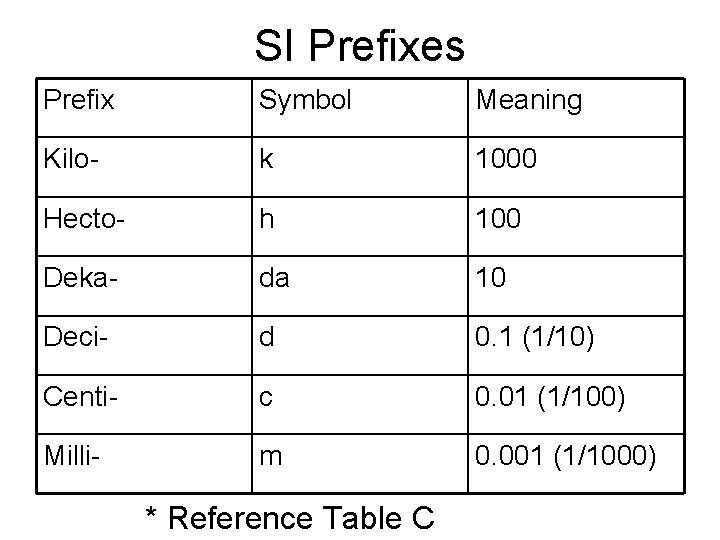
SI Prefixes Prefix Symbol Meaning Kilo- k 1000 Hecto- h 100 Deka- da 10 Deci- d 0. 1 (1/10) Centi- c 0. 01 (1/100) Milli- m 0. 001 (1/1000) * Reference Table C

Metric System Conversions • Kangaroos Hop Down Large Green Mountains During Christmas Morning – As you move left, move the decimal to the left – As you move right, move the decimal to the right

Convert each measurement 1. 2. 3. 4. 5. 873 cm 0. 05 L 1200 mg 75 dag 560 dm m m. L kg hg km

Significant Figures • Indicate the precision of a number • Used for measurements

Rules for determining Sig Figs 1. All non-zero numbers are significant 2. Zeros sandwiched between significant figures are always significant. 3. Zeros before the first non-zero number are not significant. These zeros can be thought of as “place holders” 4. Zeros at the end of a number are only significant when they are decimals.

• If the decimal is Present in a measurement, start on the Pacific side (left side of the number) with the first nonzero digit. All the following digits are significant. Atlantic • If the decimal is Absent in a measurement, start on the Atlantic side (right side of the number) with the first nonzero digit. All the preceding digits are significant. Pacific Atlantic – Pacific Rule

Sig Fig Practice 1. 2. 3. 4. 5. 6. 7. 803 60. 56 5. 780 0. 0025 0. 08150 200. 1. 50 x 1021

Exact Numbers • Exact numbers, such as the number of people in a room, have an infinite number of significant figures. • Exact numbers are counting up how many of something are present, they are not measurements made with instruments. • Exact numbers are defined numbers, such as 1 foot = 12 inches. There are exactly 12 inches in one foot. • Therefore, if a number is exact, it DOES NOT affect the accuracy of a calculation nor the precision of the expression.

Rounding Addition/Subtraction • Round your final answer to the same number of decimal places as the figure with the least number of decimal places Practice 1. 2. 1 g 12. 59 g + 34. 73 g 2. 109. 05 g - 62. 4 g

Rounding Multiplication/Division • Round your final answer to the same number of significant figures as the number with the least number of significant figures Practice 2. The mass of a solid is 1. 3. 127 3. 60 g and its volume is x 8. 01 1. 8 cm 3. What is the density of the solid?

Scientific Notation • Used as a shorthand for writing very small or very large numbers • Always written in the form a x 10 b Ø 1 a < 10 Ø Exponent will be positive for numbers greater than 1 Ø Exponent will be negative for numbers less than 1

Practice 1. 2. 3. 4. 103, 000 2 x 106 0. 6842 8. 56 x 10 -4 = =

Adding/Subtracting • Must have the same exponent first! Change the smaller exponent into the larger one • Add/Subtract the non-exponent • Keep the same exponent Examples: 1. 2. 7 x 103 + 3. 2 x 102 2. 7. 58 x 1020 – 6. 2 x 1021

Multiplying/Dividing • • Multiply/Divide non-exponent Add/Subtract exponent Examples: • (7. 2 x 10 -2) (3. 4 x 104) = • 7. 5 x 106 = 2. 5 x 102

Percent Error Measured – Accepted x 100 Accepted * Also given on Reference Table T Example: Methyl alcohol boils at 65 o. C, a student measures it to be 68 o. C. What is the percent error?

Dimensional Analysis • • • You can multiply anything by 1 and not change the value of the number Multiplying by conversion factors is the same as multiplying by 1 Just keep track of your units!!!!! Examples: 1. How many seconds are there in 5. 00 days? 2. Calculate the number of minutes in 2. 0 years? Express your answer in scientific notation.
- Slides: 27