Measurements make observations meaningful International System of Units

Measurements make observations meaningful

International System of Units (SI Units) • Uses the metric system – Based on units of 10
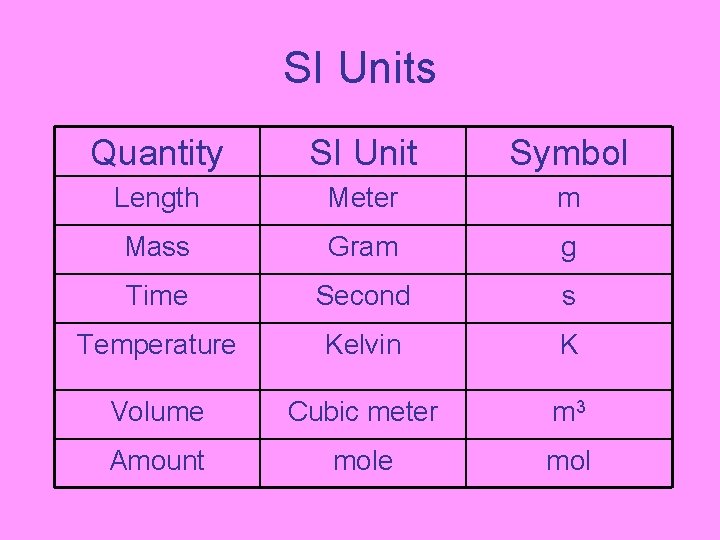
SI Units Quantity SI Unit Symbol Length Meter m Mass Gram g Time Second s Temperature Kelvin K Volume Cubic meter m 3 Amount mole mol

Mass • Mass and weight are NOT the same thing • Weight is dependant upon gravity, mass is not

Temperature • Kelvin is the SI unit, but Celsius (C) is often used • K = C + 273 Practice: 1. Convert 25 o. C to K. 2. Convert 352 K to o. C. 3. Convert -15 o. C to K. • Heat and temperature are not the same thing – – Heat is a type of energy Temperature is a measurement of energy

Important Temperatures • Freezing Points for water Ø 0 o. C = 273 K = 32 o. F • Boiling Points for water Ø 100 o. C = 373 K = 212 o. F • Absolute zero Ø 0 K (-273 o. C) ØTheoretically, all movement stops at this temperature
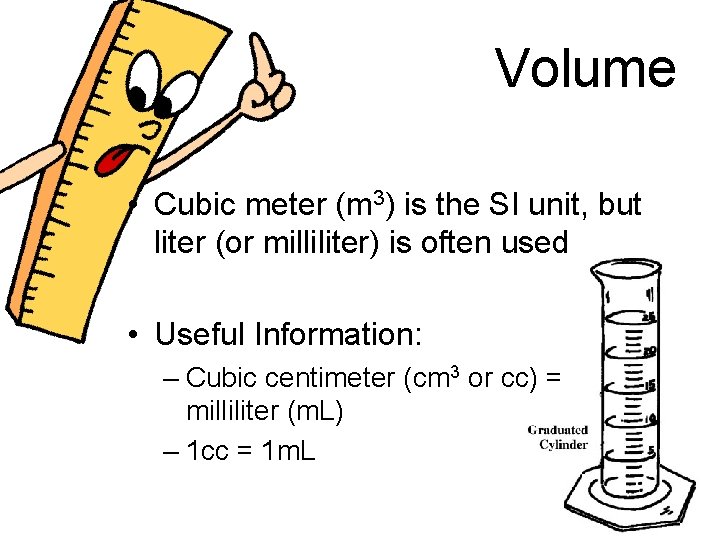
Volume • Cubic meter (m 3) is the SI unit, but liter (or milliliter) is often used • Useful Information: – Cubic centimeter (cm 3 or cc) = milliliter (m. L) – 1 cc = 1 m. L

Moles • Used to measure the amount (quantity) of something – 1 mole = 6. 02 x 1023 particles

Density • • • How much “stuff” in a given area Density of water (at 250 C) = 1. 00 g/m. L D = m/v

Density Practice 1. A rock has a mass of 3. 5 kg and a volume of 7. 0 m 3. What is the rock’s density? 2. An object’s density is 8. 0 g/cm 3 and its mass is 1. 5 g. What is the object’s volume? 3. What would the mass be of a 25 m. L sample of an object with a density of 0. 047 g/m. L?
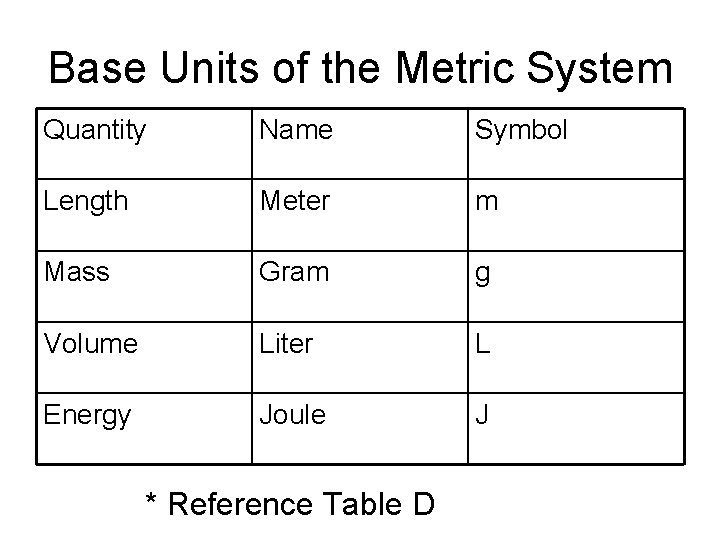
Base Units of the Metric System Quantity Name Symbol Length Meter m Mass Gram g Volume Liter L Energy Joule J * Reference Table D
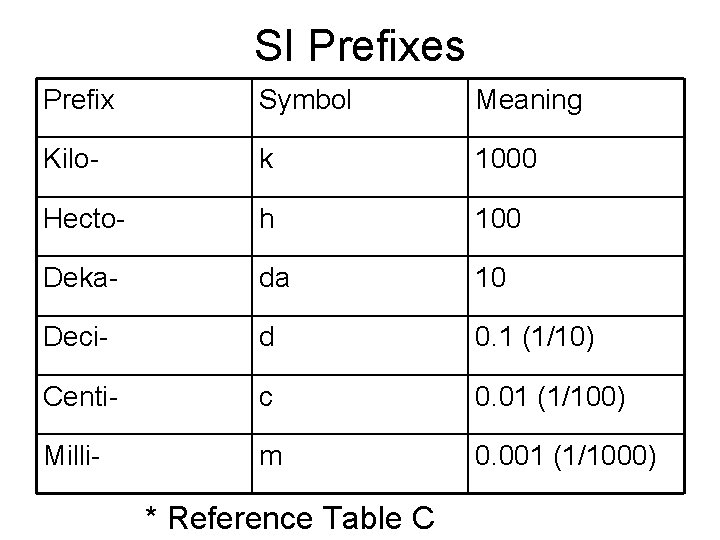
SI Prefixes Prefix Symbol Meaning Kilo- k 1000 Hecto- h 100 Deka- da 10 Deci- d 0. 1 (1/10) Centi- c 0. 01 (1/100) Milli- m 0. 001 (1/1000) * Reference Table C

Metric System Conversions • Kangaroos Hop Down Large Green Mountains During Christmas Morning – As you move left, move the decimal to the left – As you move right, move the decimal to the right
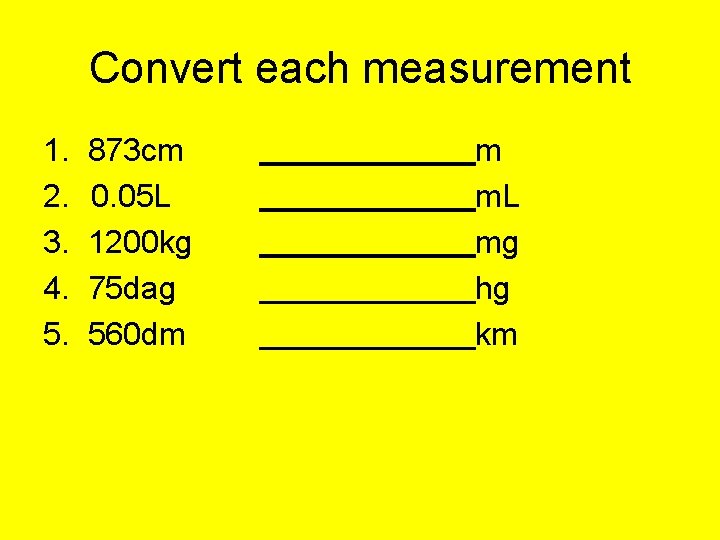
Convert each measurement 1. 2. 3. 4. 5. 873 cm 0. 05 L 1200 kg 75 dag 560 dm m m. L mg hg km

Significant Figures • Indicate the precision of a number • Used for measurements

Rules for determining Sig Figs 1. All non-zero numbers are significant 2. Zeros sandwiched between significant figures are always significant. 3. Zeros before the first non-zero number are not significant. These zeros can be thought of as “place holders” 4. Zeros at the end of a number are only significant when they are decimals.

• If the decimal is Present in a measurement, start on the Pacific side (left side of the number) with the first nonzero digit. All the following digits are significant. Atlantic • If the decimal is Absent in a measurement, start on the Atlantic side (right side of the number) with the first nonzero digit. All the preceding digits are significant. Pacific Atlantic – Pacific Rule

Sig Fig Practice 1. 2. 3. 4. 5. 6. 7. 803 60. 56 5. 780 0. 0025 0. 08150 200. 1. 50 x 1021

Exact Numbers • Exact numbers, such as the number of people in a room, have an infinite number of significant figures. Exact numbers are counting up how many of something are present, they are not measurements made with instruments. Another example of this are defined numbers, such as 1 foot = 12 inches. There are exactly 12 inches in one foot. Therefore, if a number is exact, it DOES NOT affect the accuracy of a calculation nor the precision of the expression. Some more examples: • There are 100 years in a century. • 2 molecules of hydrogen react with 1 molecule of oxygen to form 2 molecules of water.

Addition/Subtraction • Round your final answer to the same number of decimal places as the figure with the least number of decimal places Practice 1. 2. 1 g 12. 59 g + 34. 73 g 2. 109. 05 g - 62. 4 g

Multiplication/Division • Round your final answer to the same number of significant figures as the number with the least number of significant figures Practice 2. The mass of a solid 1. 3. 127 is 3. 60 g and its x 8. 01 volume is 1. 8 cm 3. What is the density of the solid?

Scientific Notation • Used as a shorthand for writing very small or very large numbers • Always written in the form a x 10 b Ø 1 a < 10 Ø Exponent will be positive for numbers greater than 1 Ø Exponent will be negative for numbers less than 1

Practice 1. 2. 3. 4. 103, 000 2 x 106 0. 6842 8. 56 x 10 -4 = =

Adding/Subtracting • Must have the same exponent first! Change the smaller exponent into the larger one • Add/Subtract the non-exponent • Keep the same exponent Examples: 1. 2. 7 x 103 + 3. 2 x 102 2. 7. 58 x 1020 – 6. 2 x 1021

Multiplying/Dividing • • Multiply/Divide non-exponent Add/Subtract exponent Examples: • (7. 2 x 10 -2) (3. 4 x 104) = • 7. 5 x 106 = 2. 5 x 102

Percent Error Measured – Accepted x 100 Accepted * Also given on Reference Table T Example: Methyl alcohol boils at 65 o. C, a student measures it to be 68 o. C. What is the percent error?

Dimensional Analysis • • • You can multiply anything by 1 and not change the value of the number Multiplying by conversion factors is the same as multiplying by 1 Just keep track of your units!!!!! Examples: 1. How many seconds are there in 5. 00 days? 2. Calculate the number of minutes in 2. 0 years? Express your answer in scientific notation.
- Slides: 27