Measurements In Science A MEASUREMENT includes both a

Measurements In Science
• A MEASUREMENT includes both a number that identifies how many units there and a unit of measure. • Example: 2 cups

• Most scientific studies and experiments use a standard system of metric units called Le Système International d’Unités, or SI for short. • SI - modern version of the metric system based on a decimal system that uses the number 10 as the base unit. SI Base Units
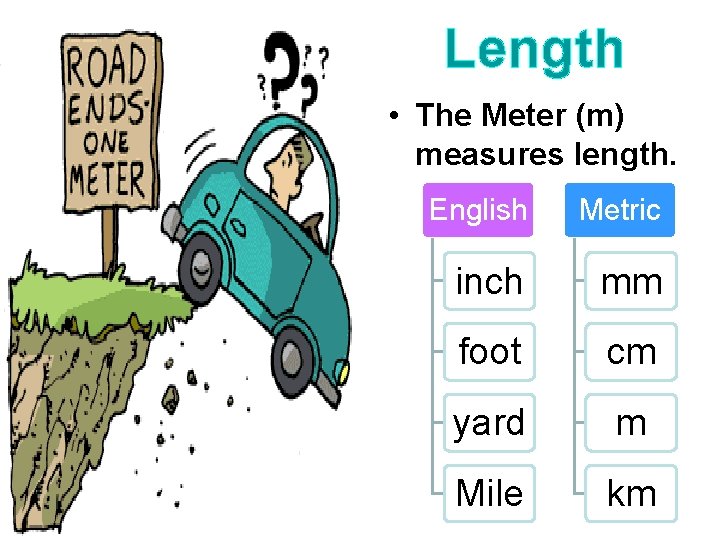
Length • The Meter (m) measures length. English Metric inch mm foot cm yard m Mile km

§ The meter can be divided into 100 equal parts called Centimeters (cm). § A millimeter (mm) is smaller than a centimeter. There is 10 mm in 1 cm. § Long distances are measured in kilometers (1000 m). 100 cm § How many mm’s are in a meter? X 10 mm 1000 mm

Weight • measure of the gravitational force on an object. § varies with location (ex. Moon). § Metric Unit = Grams (g), milligrams (mg).

If you weighed 100 lbs… Mercury Venus Earth Moon Mars 37. 8 90. 7 100 16. 6 37. 9 Jupiter Saturn Uranus Neptune Pluto 236. 4 106. 4 88. 9 112. 5 6. 7
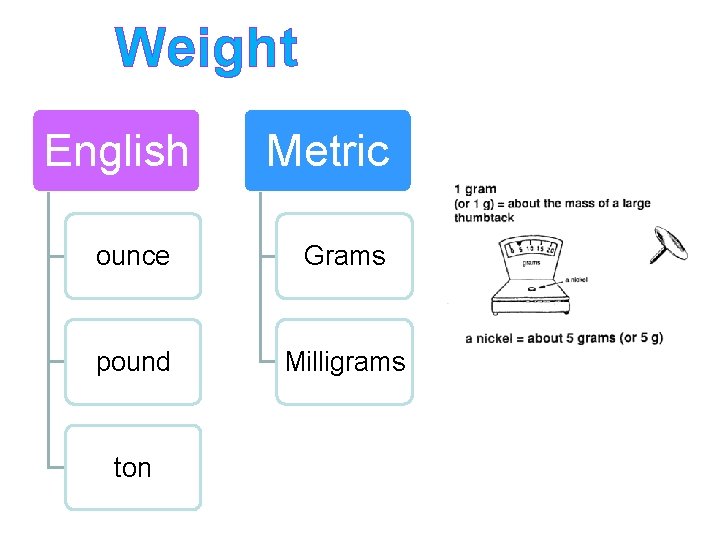
Weight English Metric ounce Grams pound Milligrams ton

Mass • amount of matter in an object. • Depends on the numbers and kind of atoms that make up an object. • Does not change with an object’s location.

• The SI unit of mass is the kilogram (kg). English pounds Metric milligrams Kilogram
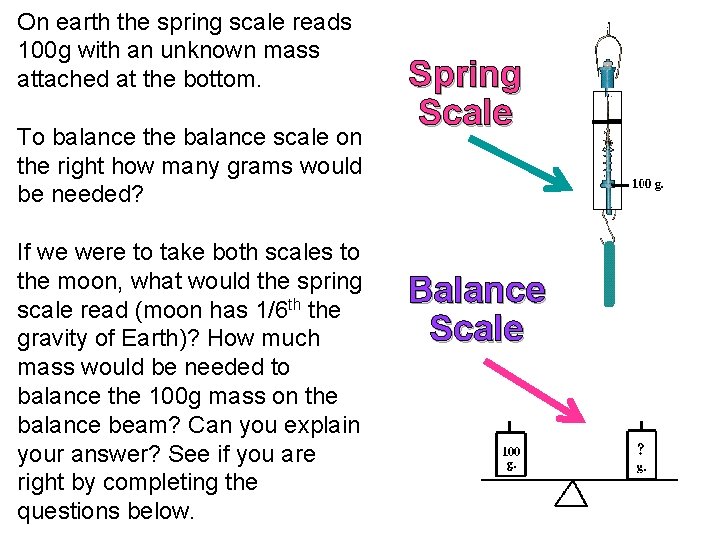
On earth the spring scale reads 100 g with an unknown mass attached at the bottom. To balance the balance scale on the right how many grams would be needed? If we were to take both scales to the moon, what would the spring scale read (moon has 1/6 th the gravity of Earth)? How much mass would be needed to balance the 100 g mass on the balance beam? Can you explain your answer? See if you are right by completing the questions below. Spring Scale Balance Scale

Volume • amount of space an object takes up. • For example, an inflated balloon takes up more space than an empty balloon. (A balloon that is blown up has more volume than one that hasn’t been blown up)

• SI measurements for liquid volumes are usually made in milliliters (m. L) or liters (L). English Volume = Length x Width x Height Metric drop m. L tsp liter tbsp 1 US gallon = 3. 78541178 liters Cup Gallon Pint

Beakers

Erlenmeyer Flasks

Graduated Cylinders


Volumetric Flasks
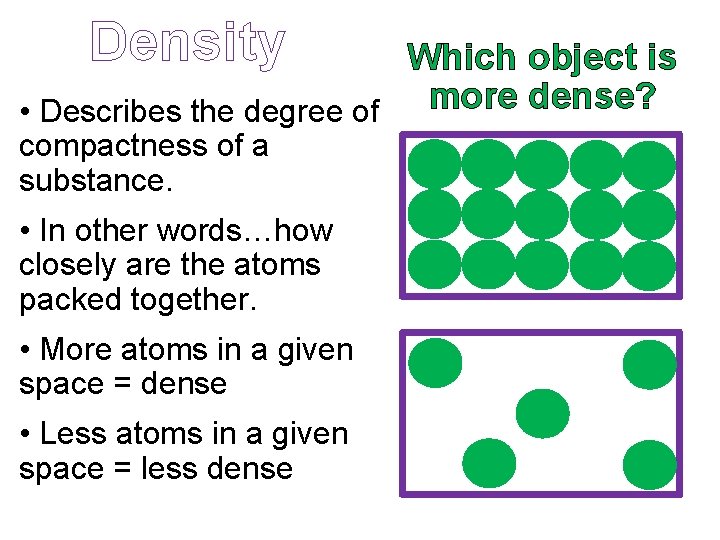
Density Which object is • Describes the degree of more dense? compactness of a substance. • In other words…how closely are the atoms packed together. • More atoms in a given space = dense • Less atoms in a given space = less dense

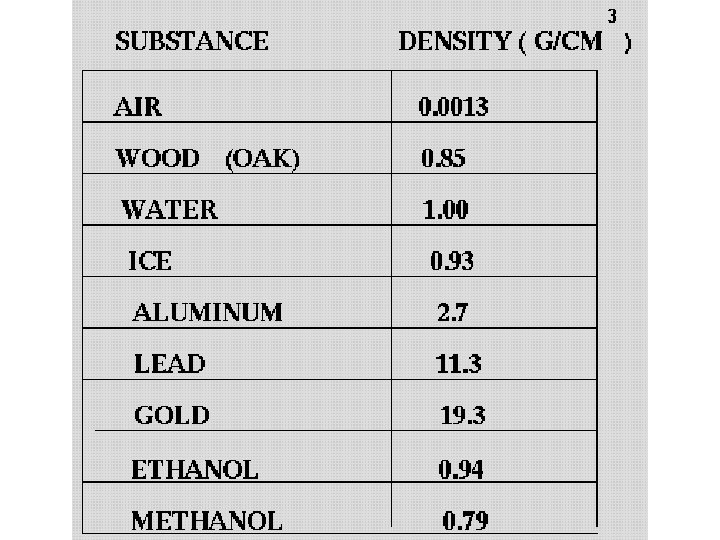
Which two factors determine Density? • Mass = a measure of the amount of atoms • Volume = the space that the atoms take up calculated by dividing the mass of an object by its volume Mass = grams Volume = m. L or cm 3 (1 ml = 1 cm 3) • Expressed in grams per cubic centimeter (g/cm 3)

Time § Time is the interval between 2 events and is measured with a watch or clock. § SI unit of time is the second.

Temperature § Temperature is a English Metric measure of the average vibrations of Fahrenheit Celsius the particles that make up a material. Kelvin (ex. solid, liquid, & gas) To calculate Fahrenheit to § SI unit for temperature is the Kelvin (K) scale. Kelvin: 1 st °C = (°F - 32) / (1. 8) 2 nd K = °C + 273. 15
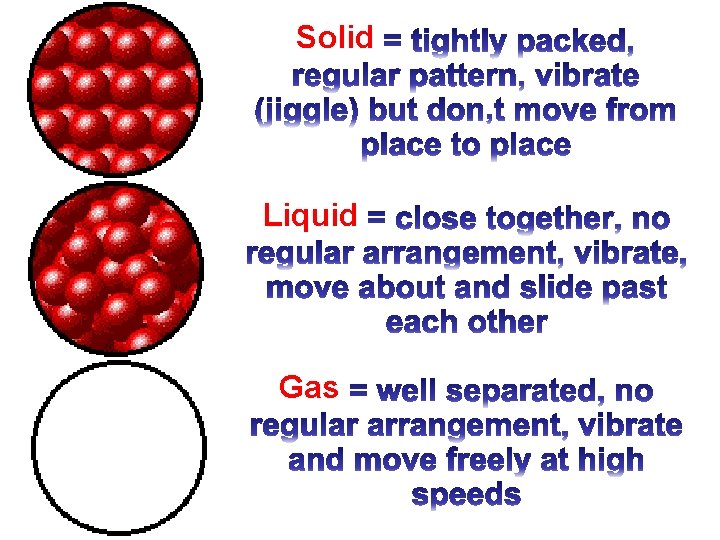
Solid Liquid Gas

To calculate Celsius to Fahrenheit: • °C x 9/5 + 32 = °F To calculate Fahrenheit to Celsius: • (°F - 32) x 5/9 = °C
- Slides: 25