MEASUREMENTS AND SIG FIGS Chemistry 82814 DRILL Get














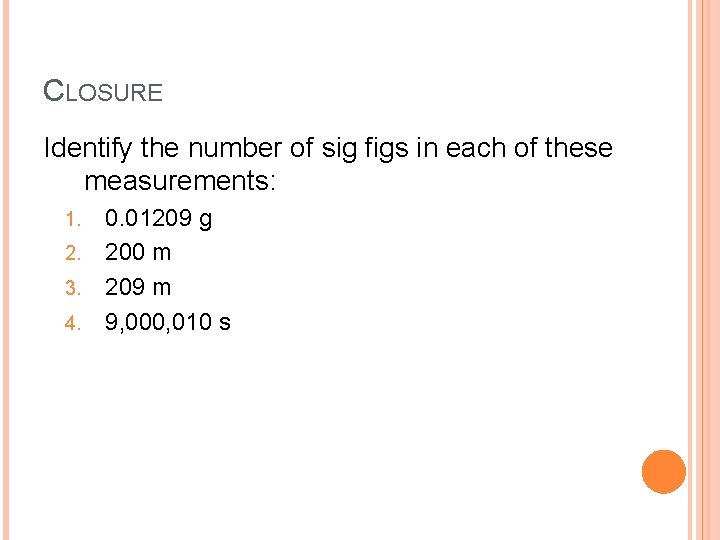
- Slides: 16

MEASUREMENTS AND SIG FIGS Chemistry 8/28/14

DRILL Get a ruler. Measure the length of your agenda book. How long is it? What unit did you choose? How many digits does your measurement have? WHY? ? HW: Significant Figures WS Front & Back

OBJECTIVE IWBAT � Measure with great precision � Use the rules to determine the number of sig figs in a measurement � Use the rules to add, subtract, multiply, and divide and have the correct number of sig figs in my answer Collect: � Bubble Lab

MEASUREMENT LAB Do all of your measurements have the same number of digits? What determines the number of digits you write down?

WHAT ARE SIGNIFICANT FIGURES? ? Please work with a partner to complete “What are Significant Figures? ? ” WS. You will be deriving rules to govern numbers!

RULES FOR DETERMINING THE NUMBER OF SIGNIFICANT FIGURES IN A GIVEN MEASUREMENT If you are given a measurement (i. e. YOU did not measure it), you follow these rules to calculate the number of sig. figs. : �All non-zero digits are sig. figs (ex. 1. 2300, 3004) �Final zeros to right of decimal are sig. figs (ex. 1. 0, 1. 2300) �Zeros surrounded by significant figures are significant (ex. 1. 2300, 102, 3004)

MEASUREMENTS DETERMINE SIGNIFICANCE Read Is the temperature on thermometer. 80 it: � 75°C? 70 � 74°C? 60 � 74. 2°C? 50 � How 40 carefully CAN you read it? 30 instrument itself determines the 20 significance. The 10 0

RECORD NUMBER OF SIGNIFICANT FIGURES IN EACH MEASUREMENT

PRACTICE Use Significant Figures WS to practice with determining numbers of Sig Figs

RULE FOR ADDITION AND SUBTRACTION WITH SIG. FIGS. Round the sum or difference so that it has the SAME number of DECIMAL PLACES as the measurement having the FEWEST decimal places. ex. 2. 7 + 3. 52 = 6. 22 = 6. 2 ex. 73 - 2. 341 = 70. 659 = 71 ex. 1001 - 0. 099 = 1000. 901 = 1001 ex. 2. 94 + 0. 06 = 3. 00 correct answer shown in red box; what you get on your calculator shown in italics

RULE FOR MULTIPLICATION & DIVISION WITH SIG. FIGS. Express a product or quotient to the same number of significant figures as the multiplied or divided measurement having the fewer total significant figures. ex. 32. 43 • 2. 17 = 70. 3731= 70. 4 (3 sig. figs. ) ex. 9574 45 = 212. 7555556 = 210 (2 sig. figs. ) correct answer shown in red box; what you get on your calculator shown in italics

1) 4. 375 g 14. 62 g 327. 9 g A. 2) 3. 0024 L - 2. 3 L = A. + 347 g B. 346. 895 g C. 346. 9 g 0. 7024 L B. 0. 7 L C. 0. 70 L

Example 1 Example 2 5. 22 m x 82. 7 m = d = 23 g / 4. 44 cm 3 = a) 431. 694 m 2 a) 5. 18018018 g/cm 3 b) 432 m 2 c) 431. 7 m 2 d) 430 m 2 b) 5. 18 g/cm 3 c) 5. 2 g/cm 3 d) 5 g/cm 3

INFINITE NUMBER OF SIGNIFICANT DIGITS Some quantities have an infinite number of significant figures because they are definitions rather than measurements. Example, by definition 1 meter = 100. 000 00…. . cm

MEASUREMENT LAB – PART II Do you think your measurements have the correct number of sig figs? Return to the lab. Measure the quantities a second time. Write the number of sig figs in the third column.
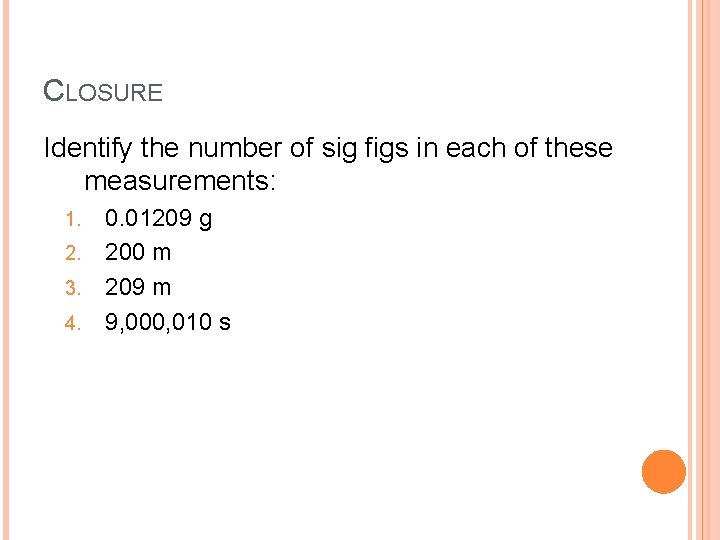
CLOSURE Identify the number of sig figs in each of these measurements: 1. 2. 3. 4. 0. 01209 g 200 m 209 m 9, 000, 010 s