Measurements and Calculations World of Chemistry Numbers and

Measurements and Calculations World of Chemistry

Numbers and measurements • Quantitative observations like measurements must have units • Measurements can be very small or very large • Example: – Distance from Earth to Sun = 93, 000 miles – Average size of eukaryotic cell: 0. 0000062 meters • For very large and small measurements, use scientific notation

• In writing in scientific notation, you will 1) Make the number between 1 and 10 (move the decimal) 2) Determine the power of 10 - if the regular number is >1, move decimal to the right, positive exponent - if the regular number is <1, move decimal to left, negative exponent • Example 1) 93, 000 miles = 9. 3 x 106 miles 2) 0. 0000062 metes = 6. 2 x 10 -6 meters

Put the following numbers into standard scientific notation: 1) 2) 3) 4) 5) 238, 000 = 1, 500, 000 = 24. 7 = 0. 135 = 0. 0024 =

Units • All measurements must have units • USA – English system, most international countries – metric system • Science community – International System (SI) • when it comes to scale, use prefixes
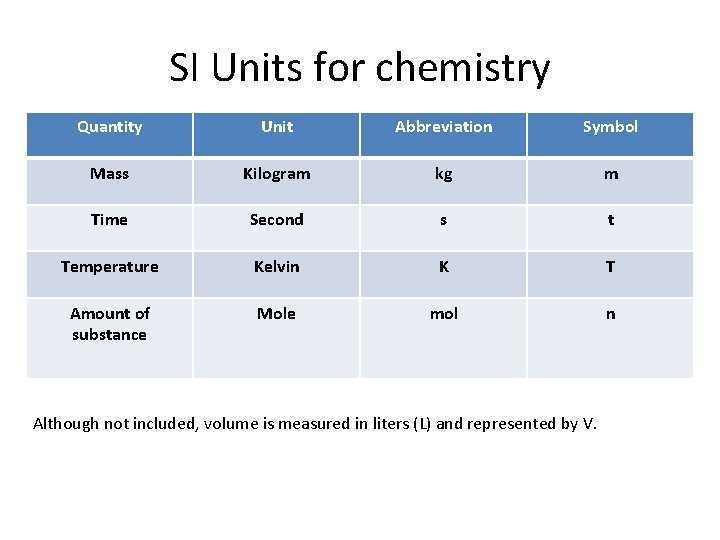
SI Units for chemistry Quantity Unit Abbreviation Symbol Mass Kilogram kg m Time Second s t Temperature Kelvin K T Amount of substance Mole mol n Although not included, volume is measured in liters (L) and represented by V.
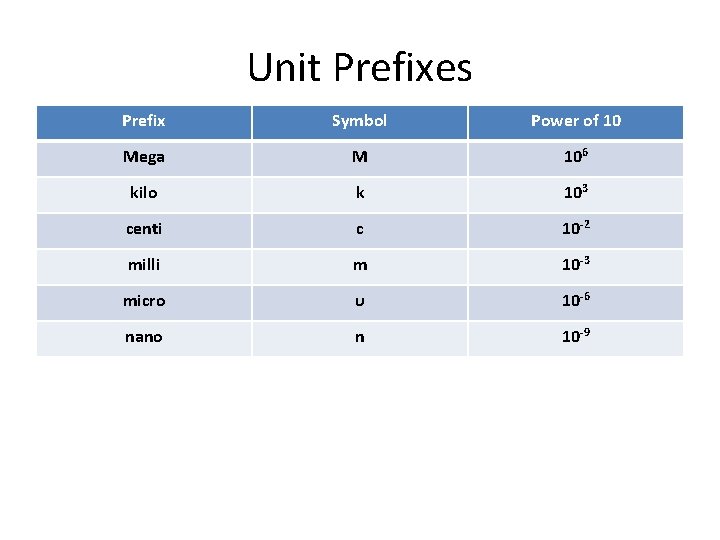
Unit Prefixes Prefix Symbol Power of 10 Mega M 106 kilo k 103 centi c 10 -2 milli m 10 -3 micro υ 10 -6 nano n 10 -9

Unit prefixes Prefixes can be used to replace powers of 10 in scientific notation Examples: – 5000 m = 5 x 103 m = 5 km – 0. 00315 L = 3. 15 x 10 -3 L = 3. 15 m. L – 0. 0000465 s = 4. 65 x 10 -9 s = 4. 65 ns – 33000 m = 33 km – 0. 00000056 L = 0. 56υL (microliters)

Practice Problems Convert the following 1) 382 g = _____kg 2) 0. 0056 g = _____mg 3) 490 m. L = _____L 4) 6, 560, 000 m = _____Mm (Megameters) 5) 99 Mg = ______g 6) 8. 8υg = ______g

Measurement Uncertainty • Measuring anything (especially mass, volume, length): certain and uncertain numbers meter stick
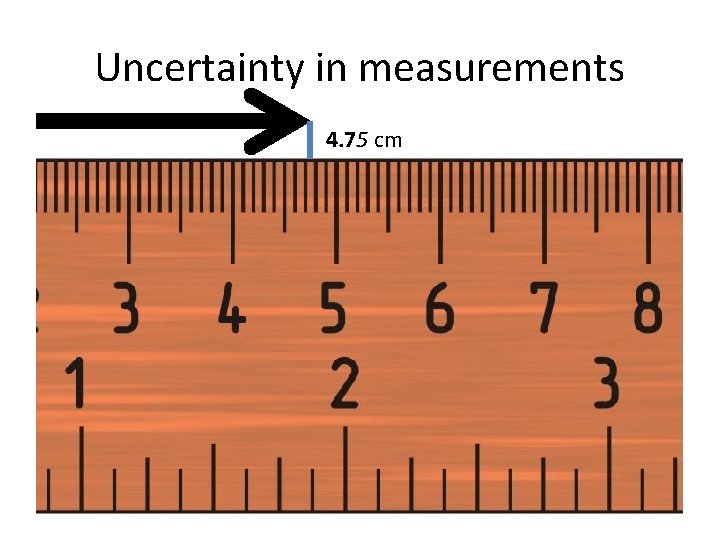
Uncertainty in measurements 4. 75 cm

Measurement Uncertainty • Determine the “certain” numbers of the measurement • Meter stick broken down into centimeters and millimeters 4. 7 cm • Determine “uncertain” numbers (estimate) – value between millimeters – 4. 75 cm • Every measuring device (ruler, graduated cylinder, balance) has some degree of uncertainty…except for digital measuring devices

Significant Figures In a measurement, specific numbers are considered significant figures What counts as sig fig? 1) All regular integers (1457 = 4 sig figs) 2) Trapped zeros (12059 = 5 sig figs) 3) Trailing zeros (only if there is a decimal involved) 100. 3 sig figs 78. 0 = 3 sig figs 100 = 1 sig fig What does not count as sig fig? Leading zeros (0. 00025 = 2 sig figs)

Practice Problems Determine the number of sig figs: 1) 2) 3) 4) 5) 6) 7) 8) 9) 10) 0. 000304 g 1. 270 x 102 m. 125 g 10 A 0. 09020 L 6. 5 x 103 g 6. 5 mg 9. 02 g 10. 0 m. L 21. 40 s

Calculating using Sig Figs 1) Multiplication/Division: # of sig figs in answer is the same as the measurement with smallest # of sig figs Example: 4. 56 x 1. 4 = 6. 384 = 6. 4 3 sig figs 2 sig figs Round off 2 sig figs

Calculating using Sig Figs Example: 8. 315 ÷ 298 = 0. 0279027 = 4 sig figs 3 sig figs 0. 0279 3 sig figs Round off

Calculating using Sig Figs Examples 5. 4 / 3 = 2. 5 x 5. 230 = (8. 62 x 103) / (33) = (0. 54 / 6. 4) x 100. 00 =

Calculating using Sig Figs 2) Addition/subtraction: sig figs in answer determined by measurement with smallest number of decimal places Example: 12. 11 + 18. 0 + 1. 013 = 31. 123 31. 1 1 decimal place

Calculating using Sig Figs Example: 0. 6875 – 0. 1 = 0. 5875 0. 6 1 decimal place

Calculations Examples • Multiplication and division – count sig figs • Addition and subtraction – count decimal places 1) 8. 90 + 63. 45 = 1) 103. 4 – 34. 94 = 1) (1945)(17. 1) = 1) 32. 789 / 270 = 1) (13. 3 x 105)(45. 45) =

Dimensional Analysis • Dimensional analysis is used to convert units Ex: g mol g L • Use conversion factors as bridges • Examples of conversion factors include. . 1 lb = 453 g 1 in = 2. 54 cm 1 mol carbon = 12. 01 g • Use railroad method to cancel out units

Dimensional Analysis Convert the following 12 ft into cm 3. 2 L into ounces 32. 0 ft in km

5. 7 Temperature • Temperature is the measure of heat • Three scales: – Celsius (°C) – Fahrenheit (°F) – Kelvin (K)

Celsius • Anders Celsius • Original 1742 “Centigrade” scale – 100°C water boils – 0°C water freezes • 100 degree scale

Fahrenheit • Daniel Gabriel Fahrenheit • Scale based on three fixed points: – “Brine” mixture: water, ice, ammonium chloride = 0°F – Water and ice mixture = 32°F – Human body = 96°F • 180 degree scale – 32°F water freezes – 212°F water boils

Kelvin • • • William Thomson (Lord Kelvin) SI unit 273 K water freezes 373 K water boils 100 degree scale Absolute zero = 0 K – all molecular motion ceases – has never been reached

Temperature Conversion Equations °C to °F T°F = (1. 8)T°C + 32 °F to °C T°C = (T°F - 32) x (0. 56) K to °C TK =T°C + 273

5. 8 Density • Physical property, specific for a pure substance • Ratio of mass and volume • Density = mass volume • Volume units – solids cm 3 – liquid m. L – gas L
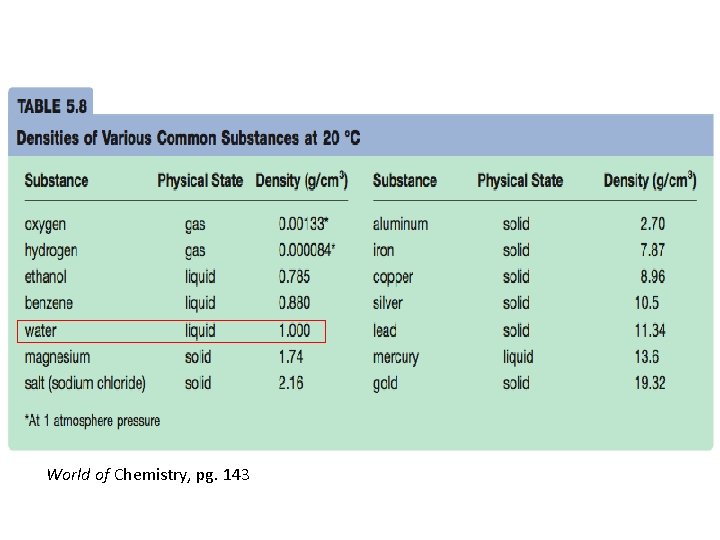
World of Chemistry, pg. 143

Determining Density Example: Mass = 55. 64 g Volume = 10. 0 m. L Density = 55. 64 g = 5. 56 g/m. L 10. 0 m. L
- Slides: 30