Measurement Scientific measurement Number vs Quantity Quantity number

Measurement Scientific measurement

Number vs. Quantity � Quantity - number + unit UNITS MATTER!! Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

2. 1 Types of measurement � Quantitative- use numbers to describe � Qualitative- use description without numbers � 4 feet � extra large � Hot � 100ºF

2. 1 Scientists prefer. . � Quantitative - easy to check � Easy to agree upon, no personal bias � The measuring instrument limits how good the measurement is.

2. 2 How good are the measurements? � Scientists use two word to describe how good the measurements are � Accuracy- how close the measurement is to the actual value � Precision- how well can the measurement be repeated
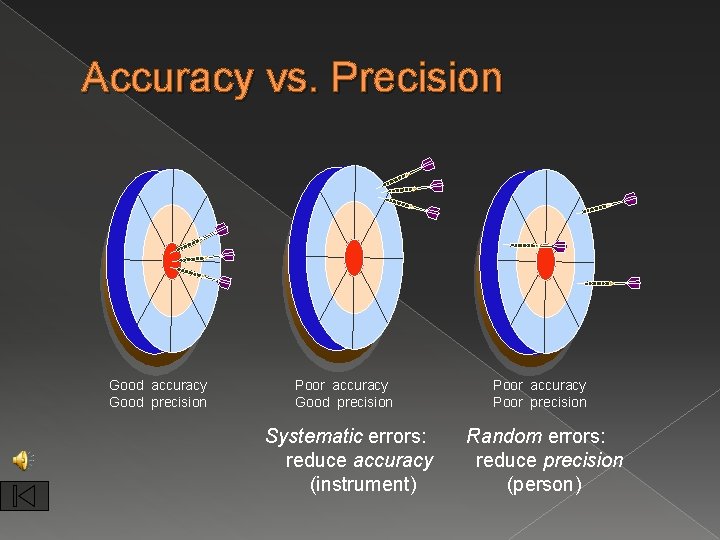
Accuracy vs. Precision Good accuracy Good precision Poor accuracy Good precision Systematic errors: reduce accuracy (instrument) Poor accuracy Poor precision Random errors: reduce precision (person)

2. 2 Differences � Accuracy can be true of an individual measurement or the average of several � Precision requires several measurements before anything can be said about it � examples

Let’s use a golf anaolgy

Accurate? No Precise? Yes 10
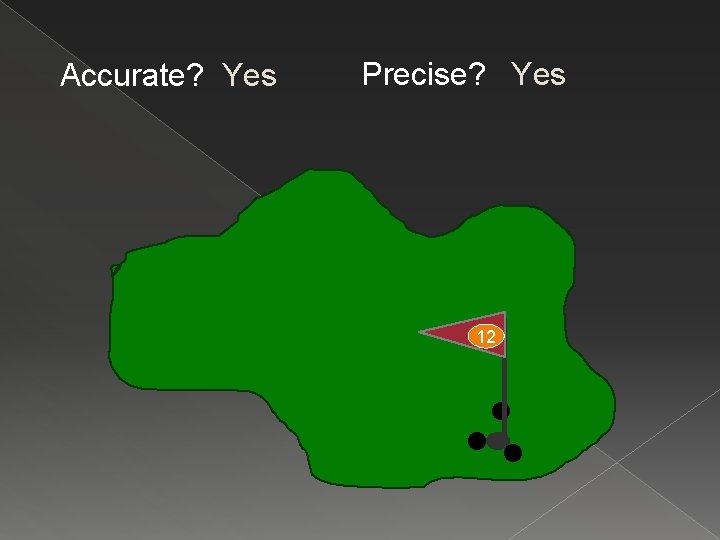
Accurate? Yes Precise? Yes 12
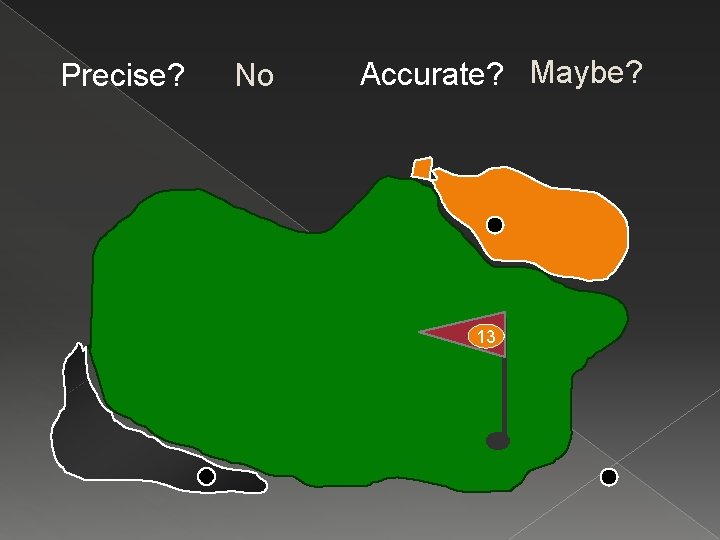
Precise? No Accurate? Maybe? 13

Accurate? Yes Precise? We cant say! 18

REVIEW: Accuracy vs. Precision � Accuracy - how close a measurement is to the accepted value � Precision - how close a series of measurements are to each other ACCURATE = CORRECT PRECISE = CONSISTENT Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

2. 3 Scientific Notation 65, 000 kg 6. 5 × 104 kg � Converting into Scientific Notation: › Move decimal until there’s 1 digit to its left. Places moved = exponent. › Large # (>1) positive exponent Small # (<1) negative exponent › Only include sig figs. Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
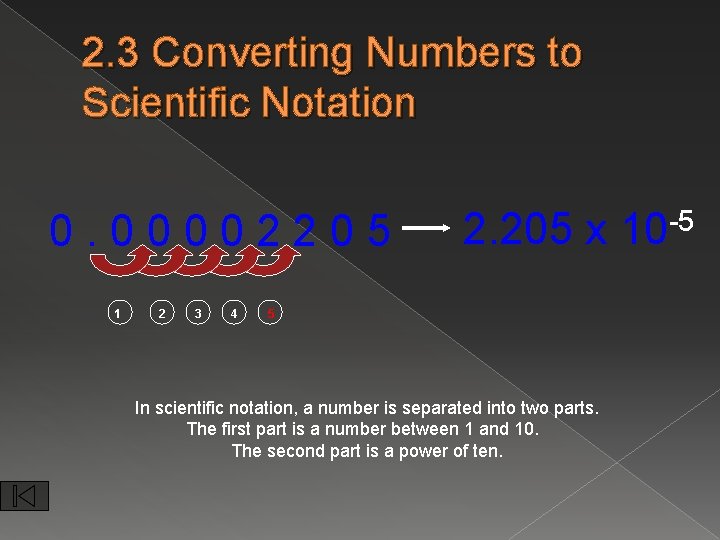
2. 3 Converting Numbers to Scientific Notation 0. 00002205 1 2 3 4 2. 205 x 5 In scientific notation, a number is separated into two parts. The first part is a number between 1 and 10. The second part is a power of ten. -5 10

Form: (# from 1 to 9. 999) x 10 exponent 800 2531 = 8 x 102 = 2. 531 x 10 = 2. 531 x 103 0. 0014 = 1. 4 / 10 = 1. 4 x 10 -3

2. 3 Scientific Notation Practice Problems 1. 2, 400, 000 g 2. 4 106 g 2. 0. 00256 kg 2. 56 10 -3 kg 3. 7 10 -5 km 0. 00007 km 4. 6. 2 104 mm 62, 000 mm Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Using the Exponent Key on a Calculator EE EXP

EE or EXP means “times 10 to the…” 23: : How to to type out 6. 02 xx 10 1023 6 0 . 2 EE EE 2 3 Don’t do it like this… 6 WRONG! 0 . 2 yx 2 3 WRONG! …or like this… 6 . 0 2 x 1 …or like this: 6 . 0 EE 2 3 TOO MUCH WORK. 0 2 x 1 0 yx 2 3
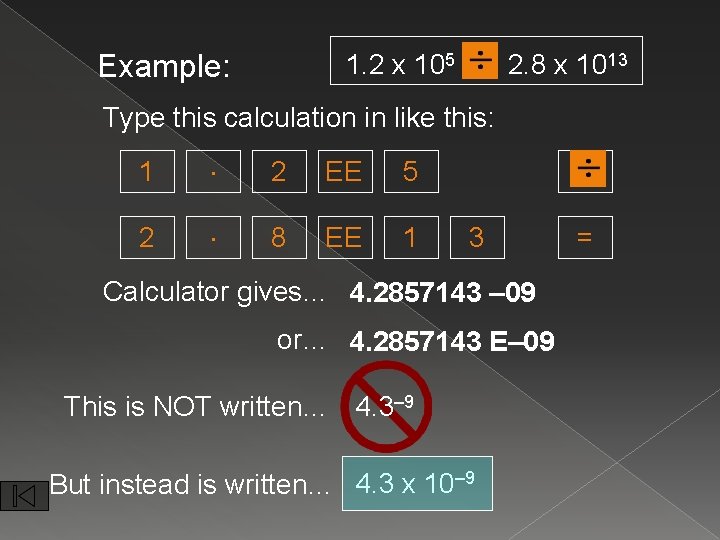
Example: 1. 2 x 105 2. 8 x 1013 Type this calculation in like this: 1 . 2 EE 5 2 . 8 EE 1 3 Calculator gives… 4. 2857143 – 09 or… 4. 2857143 E– 09 This is NOT written… 4. 3– 9 But instead is written… 4. 3 x 10– 9 =

Scientific Notation � Calculating with Scientific Notation (5. 44 × 107 g) ÷ (8. 1 × 104 mol) = Type on your calculator: 5. 44 EXP 7 EE = 671. 6049383 ÷ 8. 1 = 670 g/mol EXP EE 4 EXE ENTER = 6. 7 × 102 g/mol Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
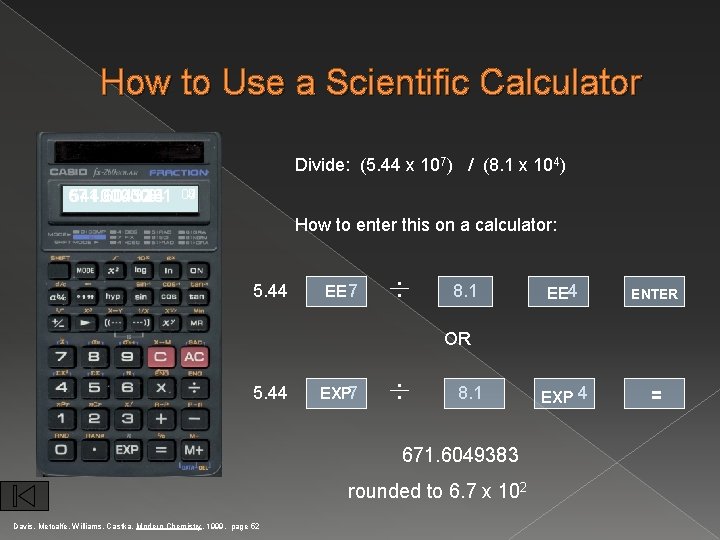
How to Use a Scientific Calculator Divide: (5. 44 x 107) / (8. 1 x 104) 671. 604938 5. 44 8. 1 54400000. 07 04 00 How to enter this on a calculator: 5. 44 EE 7 . . 8. 1 EE 4 ENTER EXP 4 = OR 5. 44 EXP 7 . . 8. 1 671. 6049383 rounded to 6. 7 x 102 Davis, Metcalfe, Williams, Castka, Modern Chemistry, 1999, page 52

2. 3 How reliable are Measurements? � How do you make a measurement? › With most measuring devices, you should be able to estimate to one decimal place more than the smallest division on the device. The smallest division is a _____ tenth of a centimeter, so hundredth you can guess to the _______ (or ___ 2 decimal places like 1. 24).
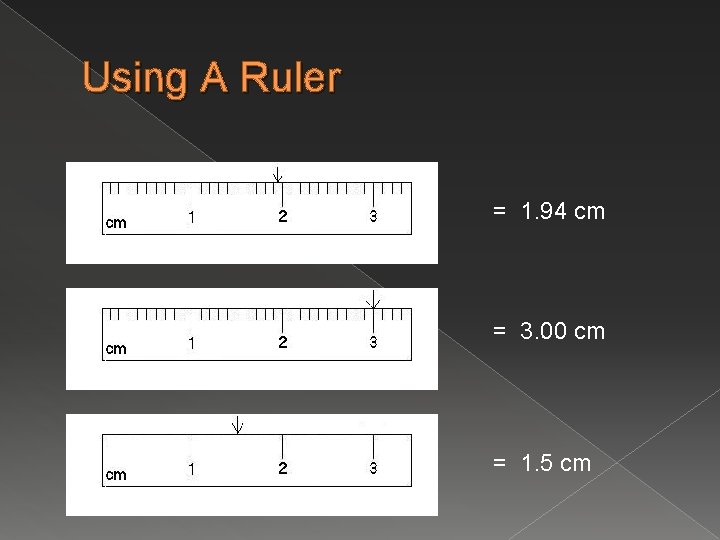
Using A Ruler = 1. 94 cm = 3. 00 cm = 1. 5 cm
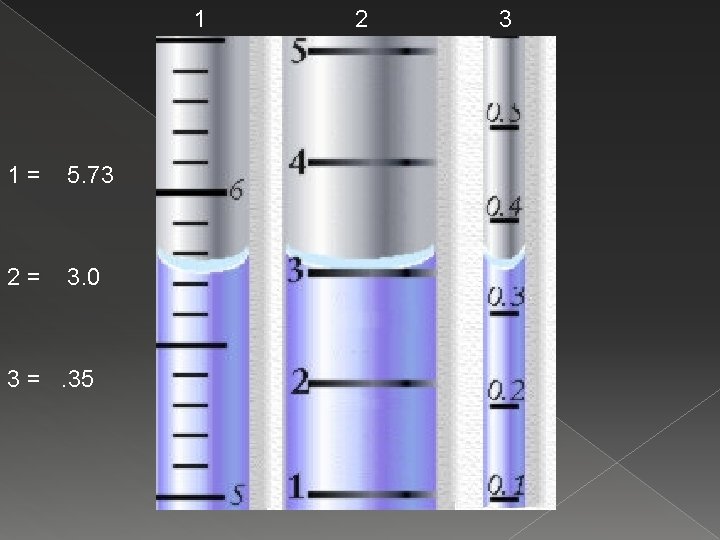
1 1= 5. 73 2= 3. 0 3 =. 35 2 3

2. 3 Significant Figures � Indicate precision of a measurement. � Recording Sig. Figs. › Sig. figs. in a measurement include the known digits plus a final estimated digit 1. 19 cm 0 1 2 3 4 Centimeters Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem 5

2. 3 Measurement – Significant Figures � Significant Figures › All of the known digits plus the estimated digit are significant – they are not placeholders. › When we measured the volume of cylinder 1 on the last slide we got: 5. 73 m. L known estimated › This would mean 3 significant figures.
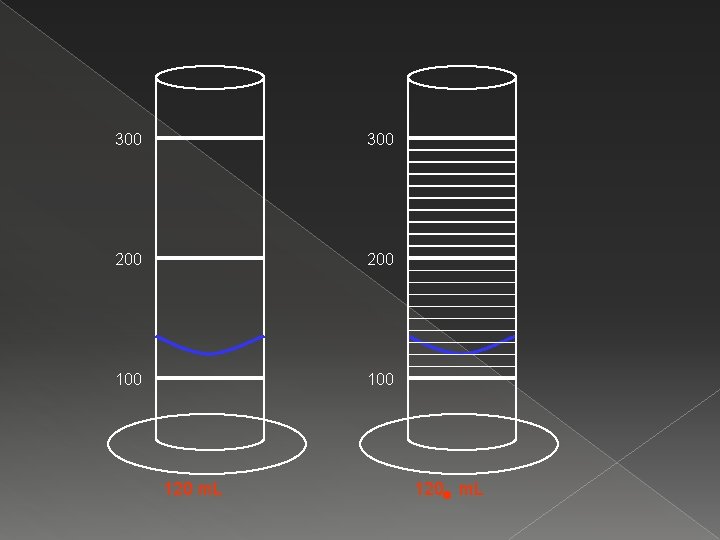
300 200 100 120 m. L
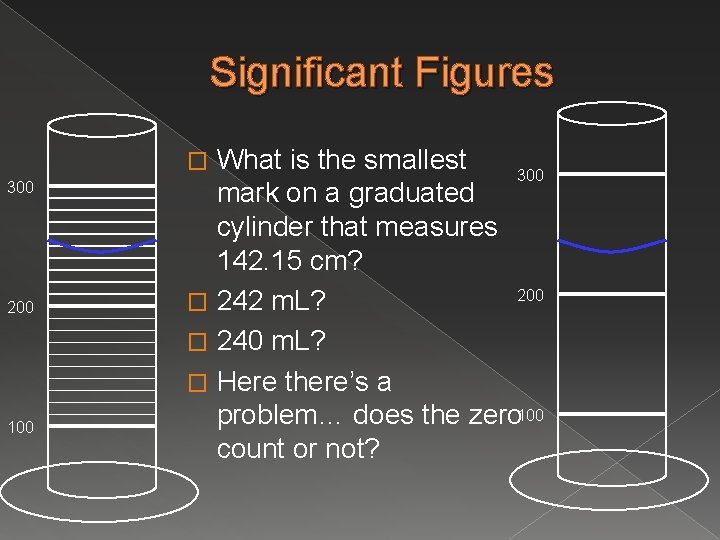
Significant Figures What is the smallest 300 mark on a graduated cylinder that measures 142. 15 cm? 200 � 242 m. L? � 240 m. L? � Here there’s a problem… does the zero 100 count or not? � 300 200 100

2. 3 Measurement – Significant Figures Significant Figure Rules Every nonzero is significant. 1. • 4 sig figs Zeros between nonzero digits are significant. 2. • 3. 123. 2 g 1004 m 4 sig figs Zeros to left of nonzero are NOT significant. • 0. 01 g 1 sig fig Zeros to the right of a nonzero number if there is no decimal point are NOT significant 4. • 1200 g 2 sig figs

Sig figs. How many sig figs in the following measurements? � 458 g � 4085 g � 4850 g � 0. 0485 g � 0. 004085 g � 40. 004085 g �

Significant Figures � Counting Sig Figs REVIEW › Count all numbers EXCEPT: �Leading zeros -- 0. 0025 �Trailing zeros without a decimal point -- 2, 500 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Significant Figures Practice Counting Sig. Figs. Examples 1. 23. 50 4 sig figs 2. 402 3 sig figs 3. 5, 280 3 sig figs 4. 0. 080 2 sig figs Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Sig Figs. � 405. 0 g � 4050 g � 0. 450 g � 4050. 05 g � 0. 0500060 g � Next we learn the rules for calculations

Rounding rules � Look at the number behind the one you’re rounding. � If it is 0 to 4 don’t change it � If it is 5 to 9 make it one bigger � Round 45. 462 to four sig figs � to three sig figs � to two sig figs � to one sig fig

Rounding Practice � Round › › the following to 3 significant figures 55. 8954 m 527, 254 g 4. 998 m. L 959, 600 m

Scientific Notation Quiz Write in Standard form � 1. 6. 32 x 105 2. 5. 05682 x 103 � Write in Scientific Notation � 3) 9384000 g 4) 0. 00000034623 m � Calculate~ Answers need to be in scientific notation! � 5. (6. 02 X 1023) X (9. 54 x 10 -13) � 6. (5. 23 X 10 -21) / (1. 23 X 1023) �

2. 5 Significant Figures in Calculations � An answer can’t have more significance than the measurements upon which it is based. � YOUR ANSWER IS ONLY AS GOOD AS YOUR WORST MEASUREMENT!

Significant Figures � Calculating with Sig Figs › Multiply/Divide - The # with the fewest sig figs determines the # of sig figs in the answer. (13. 91 g/cm 3)(23. 3 cm 3) = 324. 103 g 4 SF 324 g Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Multiplication and Division � Same rules for division Practice � 4. 5 / 6. 245 � 4. 5 x 6. 245 � 9. 8764 x. 043 � 3. 876 / 1983 � 16547 / 714

Significant Figures � Calculating with Sig Figs (con’t) › Add/Subtract - The # with the lowest decimal value determines the place of the last sig fig in the answer. 224 g 3. 75 m. L + 130 g + 4. 1 m. L 7. 85 m. L 7. 9 m. L 354 g 350 g Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

For example 27. 93 + 6. 4 34. 33 First line up the decimal places Then do the adding Find the estimated numbers in the problem This answer must be rounded to the tenths place

Significant Figures Practice Problems 1. (15. 30 g) ÷ (6. 4 m. L) 4 SF 2 SF = 2. 390625 g/m. L 2. 4 g/m. L 2. 18. 9 g - 0. 84 g 18. 06 g 18. 1 g Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem 2 SF

Problems � 500 is only 1 significant figure � If it really has two, how can I write it? � A zero at the end only counts after the decimal place � Scientific notation � 5. 0 x 102 � Now the zero counts.

Practice 1. 4. 8 + 6. 8765 2. 520 + 94. 98 3. 0. 0045 + 2. 113 4. 6. 0 x 102 - 3. 8 x 103 5. 5. 4 - 3. 28 6. 6. 7 -. 542 7. 500 -126 8. 6. 0 x 10 -2 - 3. 8 x 10 -3

Significant Figures � Calculating with Sig Figs (con’t) › Exact Numbers do not limit the # of sig figs in the answer. �Counting numbers: 12 students �Exact conversions: 1 m = 100 cm �“ 1” in any conversion: 1 in = 2. 54 cm Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

2. 5 Significant Figures in Calculations REVIEW � Addition Subtraction › Round your answer to the same number of decimal places as your least significant number. › Think of it as the leftmost uncertainty. 124. 0 m + . 12 m 420 m 544. 12 m 540 m

2. 5 Significant Figures in Calculations REVIEW � Multiplication and Division › Round answer to the same number of significant digits as the measurement with the least number of significant digits. 238. 63 m 5 × 12. 0 m 2863. 56 m 2 3 2860 m 2

2. 6 Units of Measurement – Metric 2. Metric – Developed in France in 1790. • • • Simple base units Interchangeable prefixes Decimal (base 10) system
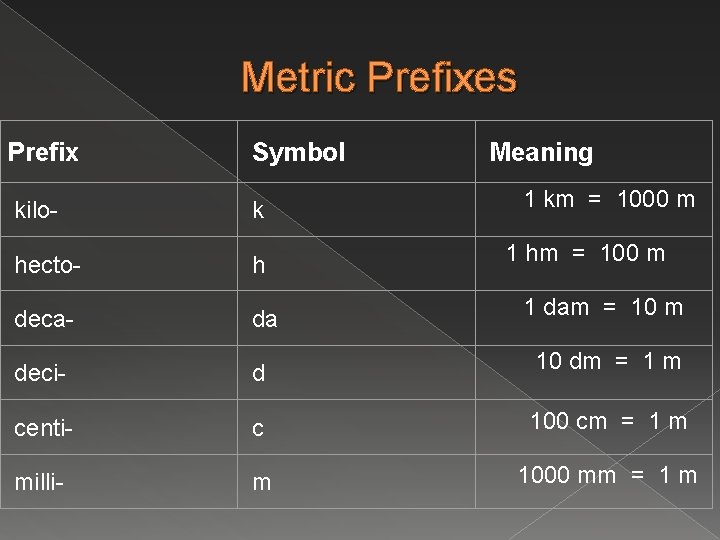
Metric Prefixes Prefix Symbol Meaning kilo- k hecto- h deca- da deci- d centi- c 100 cm = 1 m milli- m 1000 mm = 1 m 1 km = 1000 m 1 hm = 100 m 1 dam = 10 m 10 dm = 1 m
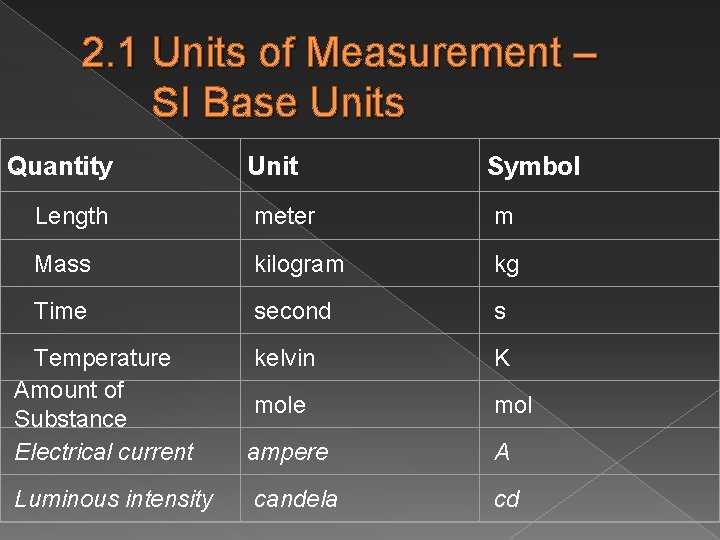
2. 1 Units of Measurement – SI Base Units Quantity Unit Symbol Length meter m Mass kilogram kg Time second s kelvin K Temperature Amount of Substance Electrical current mole mol ampere A Luminous intensity candela cd

No Cussing! The following 4 -Letter words are forbidden here: Inch Foot Yard Mile Pint Acre And we never swear the BIG F (useo. C) Please keep it clean and Metric

Metric Conversions kilo(k) 1000 hecto (h) 100 deca(da) 10 base unit Meter (m) Liter (L) Gram (g) Second (s) deci(d) 1/10 centi(c) 1/100 To convert from 1 prefix to another, just move the decimal to the left or right that many places! milli(m) 1/1000
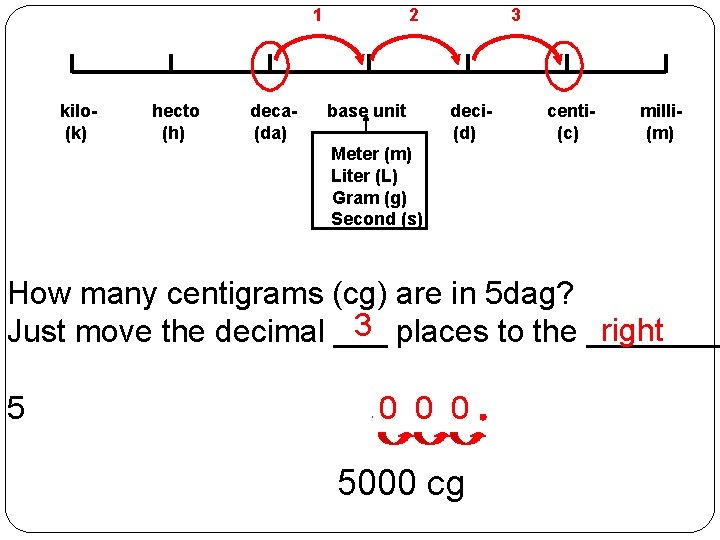
1 kilo(k) hecto (h) deca(da) 2 base unit 3 deci(d) centi(c) milli(m) Meter (m) Liter (L) Gram (g) Second (s) How many centigrams (cg) are in 5 dag? 3 places to the ____ right Just move the decimal ___ 5 0 0 0 5000 cg
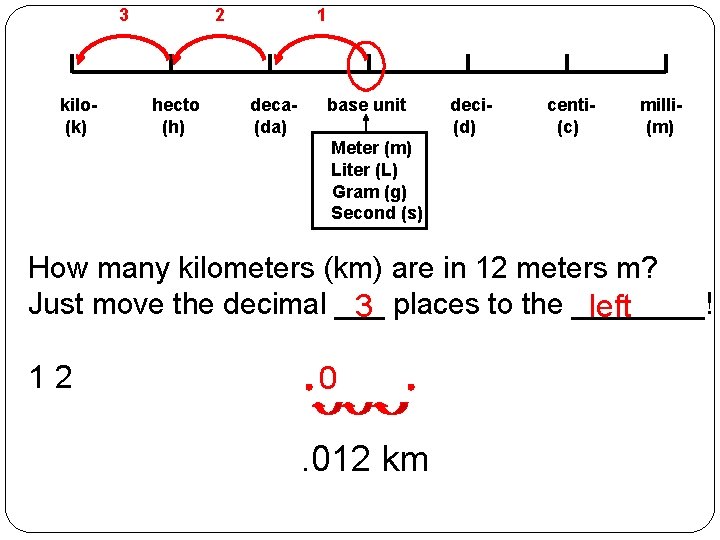
3 kilo(k) 2 hecto (h) 1 deca(da) base unit deci(d) centi(c) milli(m) Meter (m) Liter (L) Gram (g) Second (s) How many kilometers (km) are in 12 meters m? Just move the decimal ___ 3 places to the ____! left 12 0 . 012 km

Volume � calculated by multiplying L x W x H � Liter the volume of a cube 1 dm (10 cm) on a side � so 1 L = 10 cm x 10 cm � 1 L = 1000 cm 3 � 1/1000 L = 1 cm 3 � 1 m. L = 1 cm 3

Measuring Volume: Tank of Water Zumdahl, De. Coste, World of Chemistry 2002, page 143

Person Submerged in Water Zumdahl, De. Coste, World of Chemistry 2002, page 143

Mass � 1 kg = 2. 5 lbs � 1 g = 1 paper clip � 1 mg = 10 grains of salt or 2 drops of water.
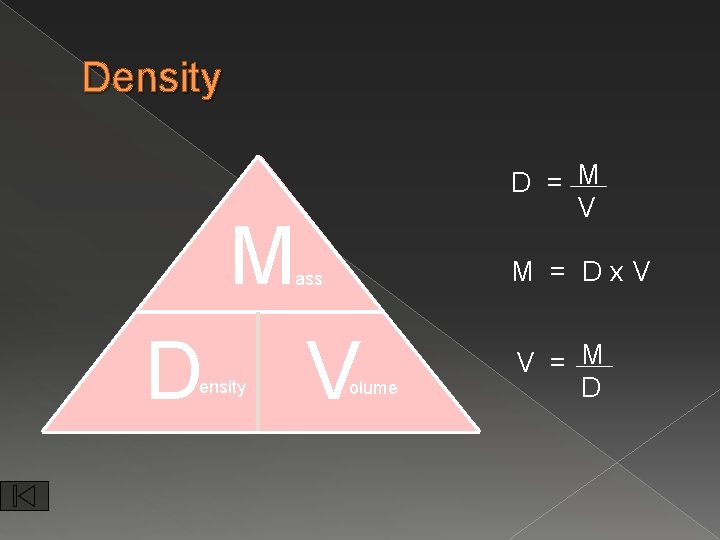
Density D = M V M M = Dx. V ass D ensity V olume V = M D

Density of Some Common Substances Substance Air Lithium Ice Water Aluminum Iron Lead Gold *at 0 o. C and 1 atm pressure Density (g / cm 3) 0. 0013* 0. 53 0. 917 1. 00 2. 70 7. 86 11. 4 19. 3

Consider Equal Volumes Mass Density = Volume Equal volumes… …but unequal masses The more massive object (the gold cube) has the GREATER density. _____ aluminum Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 71 gold
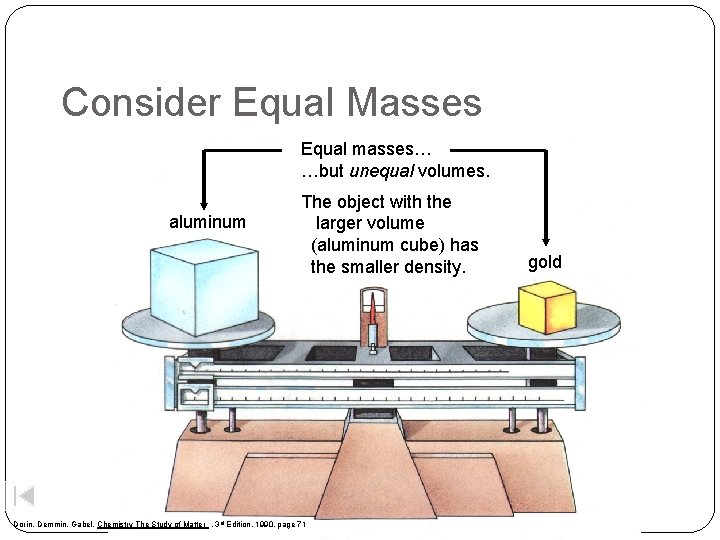
Consider Equal Masses Equal masses… …but unequal volumes. aluminum The object with the larger volume (aluminum cube) has the smaller density. Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 71 gold

Density �An object has a volume of 825 cm 3 and a density of 13. 6 g/cm 3. Find its mass. GIVEN: V = 825 cm 3 D = 13. 6 g/cm 3 M=? WORK: M = DV M = (13. 6 g/cm 3)(825 cm 3) M = 11, 200 g Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Density � A liquid has a density of 0. 87 g/m. L. What volume is occupied by 25 g of the liquid? GIVEN: D = 0. 87 g/m. L V=? M = 25 g WORK: V=M D V= 25 g 0. 87 g/m. L V = 29 m. L Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

2. 12 Temperature � Heat – type of energy transferred because of a difference in temperature. › Can’t be measured directly � Temperature – measure of the average kinetic energy of the particles in a sample of matter. › Determines the direction of heat transfer

2. 12 Temperature What contains more heat? a glass of boiling water or an iceberg What does your body sense? temperature or heat

2. 12 Temperature Scales � Fahrenheit ( F) – zero based on equal mix of snow and ammonium chloride. 32 F = freezing point of water 212 F = boiling point of water • Celsius ( C) – based on water 0 C = freezing point of water 100 C = boiling point of water

2. 12 Temperature Scales � Kelvin (K) - only temperature scales that is proportional to the speed of the particles. 0 K = all particle motion stops 273 K = freezing point of water 373 K = boiling point of water

2. 12 Temperature Conversion � T(K) = t( C) + 273 What is 25 C (room temp. ) in kelvin? T(K) = 25 C + 273 = 298 K • t( C) = T(K) – 273

� Accuracy is very important when making measurements in the lab. � In order to evaluate the accuracy of a measurement, you must be able to compare the experimental value to the accepted value. � Accepted value = the true or correct value based on reliable references � Experimental value = the measured value determined in the experiment in the lab.

Percent Error � Indicates accuracy of a measurement expressed as a percentage Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Percent Error �A student determines the density of a substance to be 1. 40 g/m. L. Find the % error if the accepted value of the density is 1. 36 g/m. L. % error = 2. 9 % Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
- Slides: 73