Measurement Lab Equipment Graphing and Lab Safety Section

Measurement, Lab Equipment, Graphing and Lab Safety Section 1. 4

A Common Measurement System • It is important for scientists to be able to replicate each other’s experiments. • Most experiments require measuring. • Therefore scientists need a common system of measurement. • Most scientists use the metric system when collecting data and performing experiments.

Metric System • Metric system – a decimal system of measurement whose units are based on certain physical standards and are scaled on multiples of 10. • The revised version of the original metric system is called the International System of Units, or SI. • Because it is based on 10 it is easy to use.
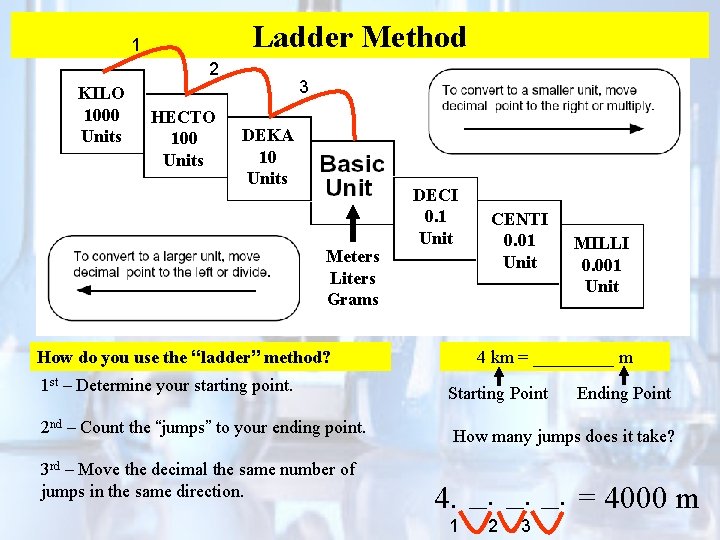
Ladder Method 1 2 KILO 1000 Units HECTO 100 Units 3 DEKA 10 Units Meters Liters Grams DECI 0. 1 Unit How do you use the “ladder” method? 1 st – Determine your starting point. 2 nd – Count the “jumps” to your ending point. 3 rd – Move the decimal the same number of jumps in the same direction. CENTI 0. 01 Unit MILLI 0. 001 Unit 4 km = _____ m Starting Point Ending Point How many jumps does it take? 4. __. __. = 4000 m 1 2 3
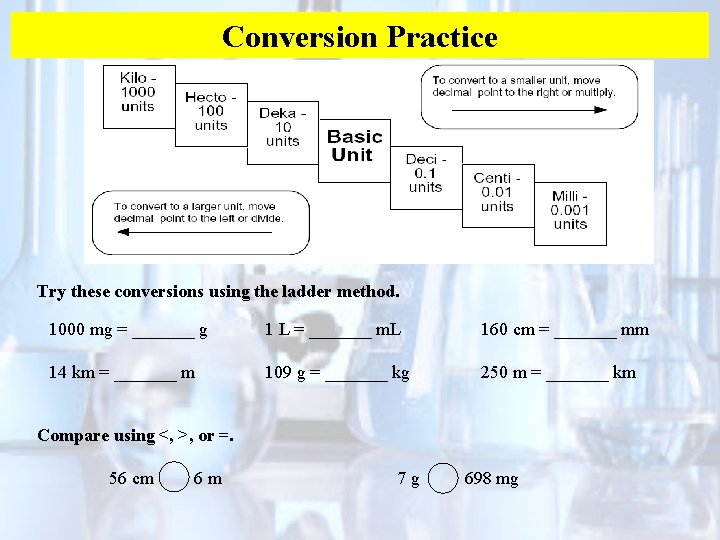
Conversion Practice Try these conversions using the ladder method. 1000 mg = _______ g 1 L = _______ m. L 160 cm = _______ mm 14 km = _______ m 109 g = _______ kg 250 m = _______ km Compare using <, >, or =. 56 cm 6 m 7 g 698 mg
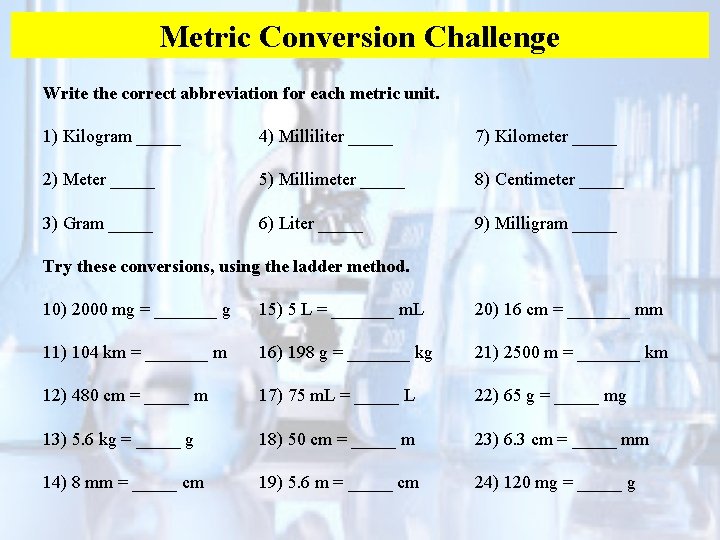
Metric Conversion Challenge Write the correct abbreviation for each metric unit. 1) Kilogram _____ 4) Milliliter _____ 7) Kilometer _____ 2) Meter _____ 5) Millimeter _____ 8) Centimeter _____ 3) Gram _____ 6) Liter _____ 9) Milligram _____ Try these conversions, using the ladder method. 10) 2000 mg = _______ g 15) 5 L = _______ m. L 20) 16 cm = _______ mm 11) 104 km = _______ m 16) 198 g = _______ kg 21) 2500 m = _______ km 12) 480 cm = _____ m 17) 75 m. L = _____ L 22) 65 g = _____ mg 13) 5. 6 kg = _____ g 18) 50 cm = _____ m 23) 6. 3 cm = _____ mm 14) 8 mm = _____ cm 19) 5. 6 m = _____ cm 24) 120 mg = _____ g

Analyzing Biological Data • Sometimes the simplest way to organize data is in the form of a table or graph. • A graph can make a pattern much easier to recognize and understand.
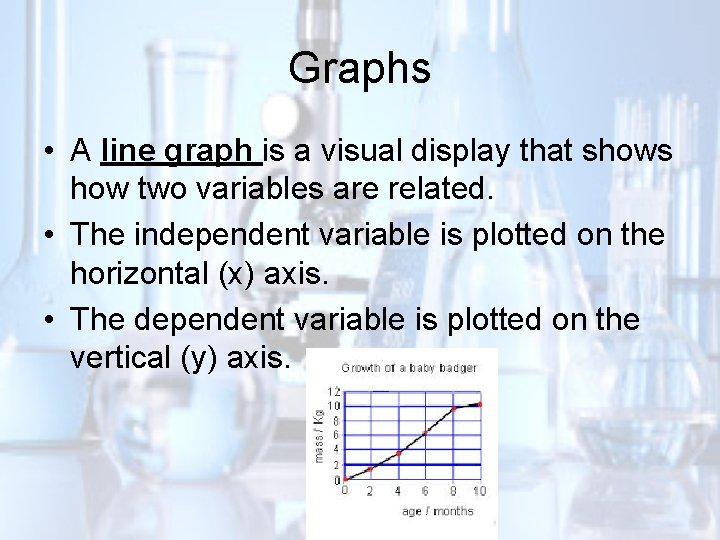
Graphs • A line graph is a visual display that shows how two variables are related. • The independent variable is plotted on the horizontal (x) axis. • The dependent variable is plotted on the vertical (y) axis.
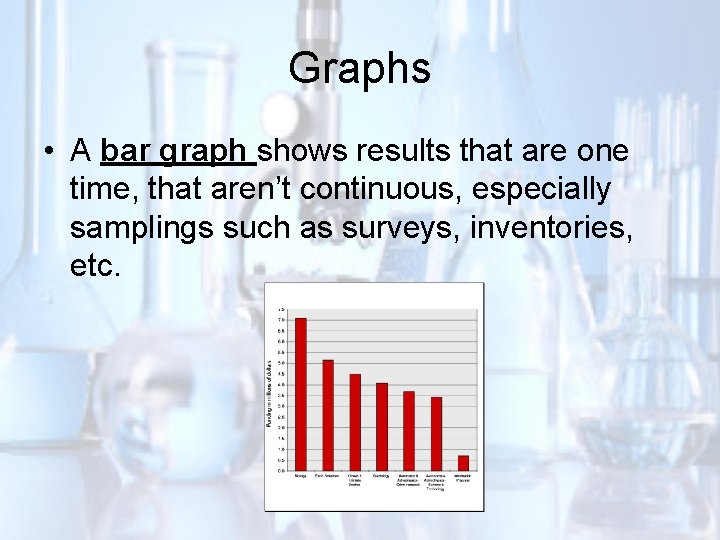
Graphs • A bar graph shows results that are one time, that aren’t continuous, especially samplings such as surveys, inventories, etc.

Graphs • A pie graph is good to use when looking at parts of a whole. • Percentages.

Lab Equipment

Safety Gloves

Safety Glasses

Beaker

Flask

Forceps

Funnel

Incubator

Graduated Cylinders

Concave Microscope Slides

Light Microscope

Electron Microscope

Microscopes • Light microscopes produce magnified images by focusing visible light rays. • Electron microscopes produce magnified images by focusing beams of electrons. • Compound light microscopes allow light to pass through the specimen and use two lenses to form an image.

Test tubes

Thermometer

Test tube rack

Petri dish

Disposable Pipette

Pipette pump

Hot plate
Medicine Dropper

Laboratory Techniques • Biologists use many techniques to study cells. • Cell Cultures – a group of cells that develops from a single original cell. • Cell Fractionation – to separate the different cell parts.

Lab Safety

General Safety Rules 1. Listen to or read instructions carefully before attempting to do anything. 2. Wear safety goggles to protect your eyes from chemicals, heated materials, or things that might be able to shatter. 3. Notify your teacher if any spills or accidents occur.

General Safety Rules 4. After handling chemicals, always wash your hands with soap and water. 5. During lab work, keep your hands away from your face. 6. Tie back long hair.

General Safety Rules 7. Roll up loose sleeves. 8. Know the location of the fire extinguisher, fire blanket, eyewash station, and first aid kit. 9. Keep your work area uncluttered. Take to the lab station only what is necessary.

General Safety Rules 10. It is suggested that you wear glasses rather than contact lenses. 11. Never put anything into your mouth during a lab experiment. 12. Clean up your lab area at the conclusion of the laboratory period. 13. Never “horse around” or play practical jokes in the laboratory.

Glassware Safety 1. 2. 3. Chipped or cracked glassware should not be used. Show it to the teacher. Broken glassware should not be disposed of in a classroom trashcan. There is a special glass disposal container for it. When pouring liquids into glassware, make sure the container you are pouring into is resting on a table at least a hands breadth from the edge.

Glassware Safety 4. Pour down a glass stirring rod to prevent liquids from splattering. 5. If a piece of glassware gets broken, do not try to clean it up by yourself. Notify the teacher. 6. When inserting glass tubing into a rubber stopper, apply a lubricant like glycerin to the glass and use a twisting motion.

Glassware Safety 7. Do not place hot glassware in water. Rapid cooling may make it shatter.

Chemical Safety 1. 2. 3. Wear protective goggles and a lab apron whenever heating or pouring hazardous chemicals. Never mix chemicals together unless you are told to do so (and then only in the manner specified). Never taste any chemicals (you should never taste anything in the lab).

Chemical Safety 4. If you need to smell the odor of a chemical, waft the fumes toward your nose with one hand. Do not put your nose over the container and inhale the fumes. 5. Never pour water into a concentrated acid. Acid should be poured slowly into water.

Chemical Safety 6. 7. Follow the instructions of your teacher when disposing of all chemicals. Wash your hands after handling hazardous chemicals.

Electrical Safety 1. 2. 3. Lay electrical cords where no one can trip on them or get caught in them. Be sure your hands and your lab area are dry before using electrical equipment. Never poke anything into electrical outlets.

Electrical Safety 4. 5. Unplug cords by pulling the plug and not the cord. Unplug all electrical equipment at the end of the lab period.
- Slides: 45