MEASUREMENT CHOOSING THE CORRECT TOOL CALCULATED UNITS READING





























- Slides: 34

MEASUREMENT CHOOSING THE CORRECT TOOL CALCULATED UNITS READING MEASUREMENTS TEMPERATURE CONVERSIONS ESTIMATING METRIC CONVERSIONS

Physical quantities consist of both a number and a unit.

SI MEASUREMENT • Le Système international d'unités • The only countries that have not officially adopted SI are Liberia (in western Africa) and Myanmar (a. k. a. Burma, in SE Asia), but now these are reportedly using metric regularly • Metrication is a process that does not happen all at once, but is rather a process that happens over time. • Among countries with non- Information from U. S. Metric Association



LENGTH: CENTIMETERS AND MILLIMETERS centimet ers millimeters



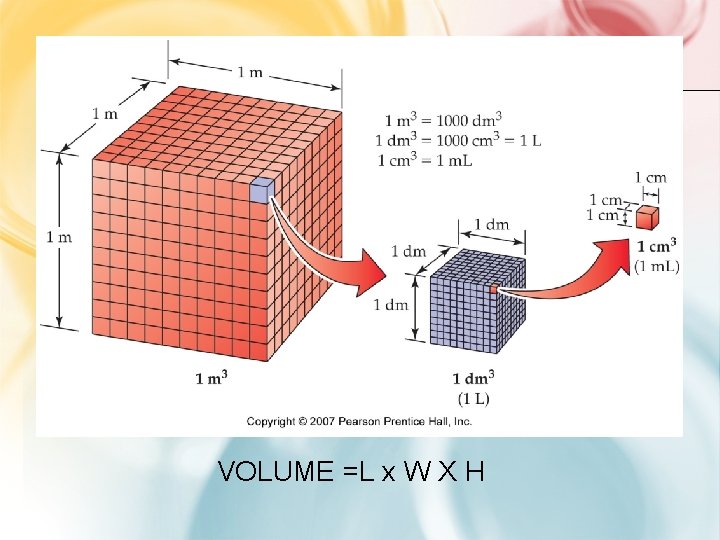
VOLUME: MILLILITERS AND CUBIC CENITMETERS


MEASUREMENT AND SIGNIFICANT FIGURES • Every experimental measurement has a degree of uncertainty. • The volume, V, at right is certain in the 10’s place, 10 m. L<V<20 m. L • The 1’s digit is also certain, 17 m. L<V<18 m. L • A best guess is needed for the tenths





VOLUME =L x W X H

VOLUME DISPLACEMENT A solid displaces a matching volume of water when the solid is placed in water. 33 m. L 25 m. L

MASS – GRAMS & KILOGRAMS


THE TOP LOADING BALANCE • These balances are used when an accuracy of +/- 0. 1 g is sufficient. • Before weighing check that the balance is displaying grams. • Zero or Tare the balance by pressing on the Zero button.


TEMPERATURE - Fahrenheit, Celsius, and Kelvin temperature scales



CALCULATIONS USING TEMPERATURE • Generally require temp’s in kelvins • T (K) = t (˚C) + 273 • Body temp = 37 ˚C + 273 = 310 K • Liquid nitrogen = -196 ˚C + 273 = 77 K

Why is this stuff important?

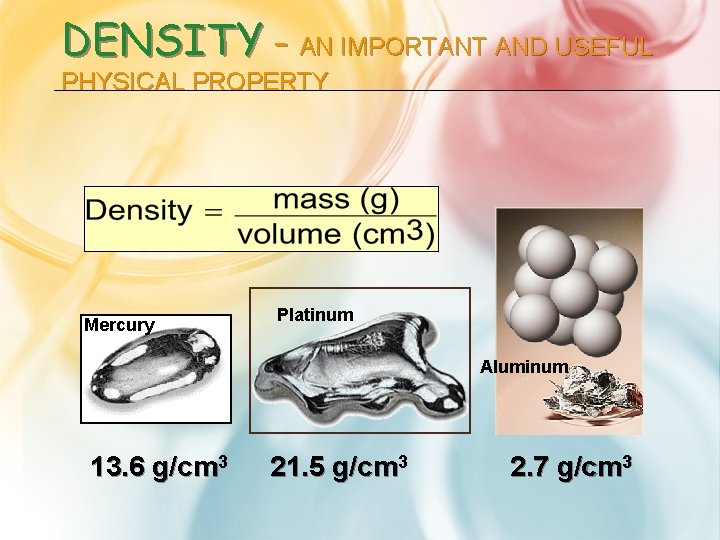
DENSITY - AN IMPORTANT AND USEFUL PHYSICAL PROPERTY Mercury Platinum Aluminum 13. 6 g/cm 3 21. 5 g/cm 3 2. 7 g/cm 3

Problem A piece of copper has a mass of 57. 54 g. It is 9. 36 cm long, 7. 23 cm wide, and 0. 95 mm thick. Calculate density (g/cm 3).

PROBLEM: MERCURY (HG) HAS A DENSITY OF 13. 6 G/CM 3. WHAT IS THE MASS OF 95 ML OF HG IN GRAMS?

LEARNING CHECK Osmium is a very dense metal. What is its density in g/cm 3 if 50. 00 g of the metal occupies a volume of 2. 22 cm 3? 1) 2. 25 g/cm 3 2) 22. 5 g/cm 3 3) 111 g/cm 3

LEARNING CHECK What is the density (g/cm 3) of 48 g of a metal if the metal raises the level of water in a graduated cylinder from 25 m. L to 33 m. L? 1) 0. 2 g/ cm 3 2) 6 g/m 3 3) 252 g/cm 3 33 m. L

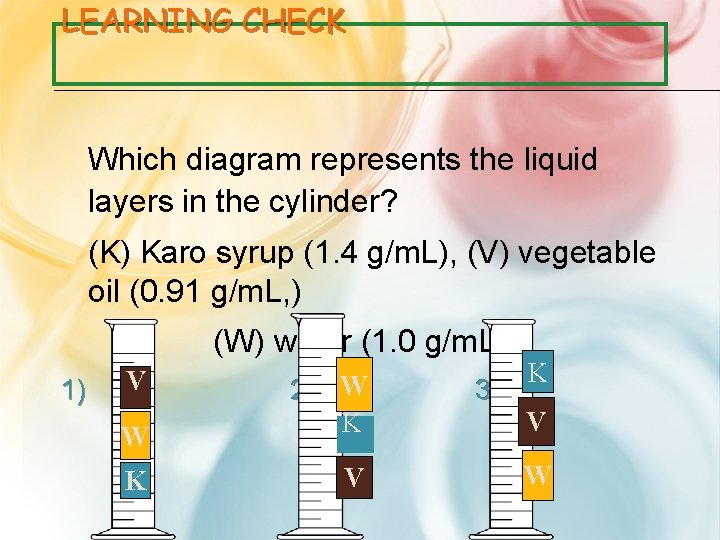
LEARNING CHECK Which diagram represents the liquid layers in the cylinder? (K) Karo syrup (1. 4 g/m. L), (V) vegetable oil (0. 91 g/m. L, ) (W) water (1. 0 g/m. L) 1) V W K 2) W K V 3) K V W

LEARNING CHECK If blood has a density of 1. 05 g/m. L, how many liters of blood are donated if 575 g of blood are given? 1) 2) 3) 0. 548 L 1. 25 L 1. 83 L

LEARNING CHECK A group of students collected 125 empty aluminum cans to take to the recycling center. If 21 cans make 1. 0 pound of aluminum, how many liters of aluminum (D=2. 70 g/cm 3) are obtained from the cans? 1) 1. 0 L 2) 2. 0 L 3) 4. 0 L