Measurement and the Metric System Quantitative observations Measurement

Measurement and the Metric System Quantitative observations

Measurement Fundamental Direct measurement Indirect Derived measurement quantities Calculations Density Heat quantities energy

Rules for Significant Measurements One place past the smallest calibration Indicates where uncertainty is Communicate instrument precision of the

Protocols for deriving quantities Multiplication Addition and division and subtraction Rounding values

Metric System Developed in France in 1795 Revised and called International System of Measurements (SI) in 1960 Based on units of 10
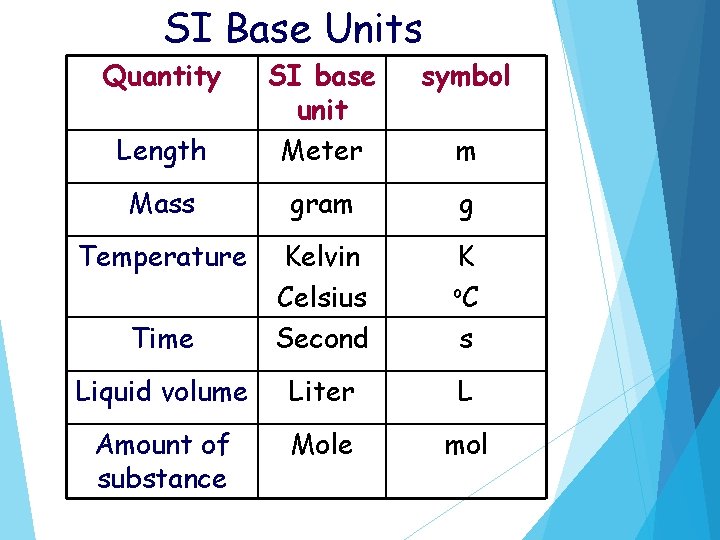
SI Base Units Quantity symbol Length SI base unit Meter Mass gram g Temperature Time Kelvin Celsius Second K o. C s Liquid volume Liter L Amount of substance Mole mol m

Memorize these !!!!! * “unit” refers to “base unit” 1 Kilo = 1 x 103 units 1 Hecto = 1 x 102 units 1 Mega(M) = 1 x 106 units 1 Deka = 1 x 101 units 1 x 106 Micro(µ) = 1 unit Basic unit = 1 1 x 109 Nano(n) = 1 unit 1 x 101 deci (d) = 1 unit 1 x 1012 Pico(p) = 1 unit 1 x 102 centi 1 x 103 milli(m) = 1 unit (c)= 1 unit 1 Giga(G) = 1 x 109 units

Conversions Practice

Density Amount of matter per unit volume Water: Iron: Lead 1. 000 g/ ml at 25 C 7. 87 g/cm 3 11. 3 g/cm 3 Density is calculated by: D = mass volume

Density is the amount of matter in a given space (volume) D = mass volume
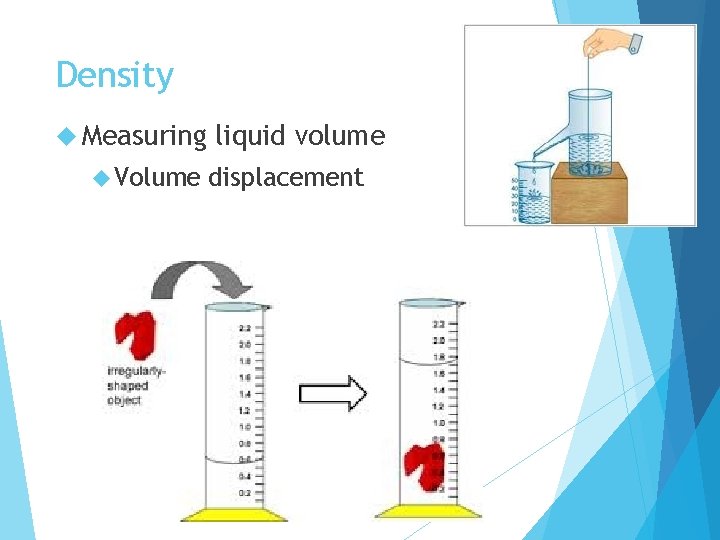
Density Measuring Volume liquid volume displacement
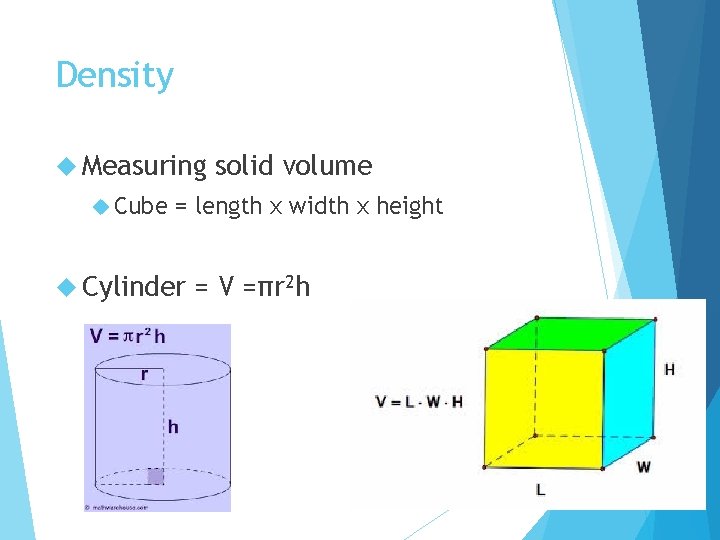
Density Measuring Cube solid volume = length x width x height Cylinder = V =πr 2 h

Density Ex: A solid displaces 45. 67 ml of water and has a mass of 98. 5 grams. Calculate its density. Is it a block of lead?

Density practice

Specific gravity Comparison of the ratio of the density of a substance to the density of liquid water at 4 C (1. 00 g/ml)

Temperature Scales Temperature is the measure of the average kinetic energy of a system. Implies molecular motion Celsius temperature Kelvin temperature

Temperature Scales Fahrenheit Celsius Kelvin Water Boils Water Freezes Molecular Motion Stops 212 o. F 100 o. C 373 K 32 o. F 0 o C 273 K -460 o. F -273 o. C 0 K (absolute zero)

Converting between Temperature Scales Fahrenheit and Celsius: o. C = 5/9 (o. F-32) o. F = (9/5 x o. C)+32 Ex. What is the Celsius value for 60. 0 o. F? o. C = 5/9(60 -32) o. C = 5/9(28) o. C = 15. 55 o. C =15. 6

From Celsius to Kelvin: o. C K = + 273 Ex. 80. 0 o. C would be what value on the Kelvin scale? K = 80 + K = 353 273

Heat and Specific Heat is the flow of thermal energy in a system Heat Ice Heat flows from hotter to cooler cubes! can flow into or out of a system

Heat and Specific Heat transfer that causes temperature changes simply change in KINETIC ENERGY Heat transfer that causes phase changes imply change in POTENTIAL ENERGY

The Joule Metric 4. 18 unit of energy joules raises 1. 00 g of water 1 C. This is the SPECIFIC HEAT OF WATER!
Percent Composition

When you see %, think…… Part X 100 = % Whole

Percent Composition percent by mass % composition = Mass of element x 100% mass of compound


Terms to review Fundamental and derived quantities Precision and accuracy Matter Mass Volume Density Significant Figures % composition Scientific notation Metric system Dimensional analysis
- Slides: 27