Measurement and Dimensional Analysis Vocabulary Element Compound Mixture

Measurement and Dimensional Analysis Vocabulary: Element Compound Mixture Chemical Symbol Atom Qualitative Quantitative Accuracy Precision Metric Units Meters Grams Liters Significant Figures Conversion Factors

Objectives 1. Differentiate between elements, compounds and mixtures. 2. Understand elements are represented by chemical symbols. 3. Define atom. 4. Count and use significant figures and scientific notation.

Chemistry All the “stuff” in the universe is made from building blocks formed in stars. These building blocks and everything made from them are called matter. Chemistry is the study of matter and the changes it undergoes.

Matter vs. Mass Matter is anything that has mass and takes up space. Examples: solids, liquids, gases, and any other states of matter Things that are NOT matter: light, heat, sound. . . Mass is the amount of matter in an object.

Classifying Substances Elements cannot be broken down or changed into smaller substances by chemical means. -The smallest particle of an element is an atom. Compounds are made up of 2 or more different elements chemically bonded together. Mixtures are a physical blend of two or more substances mixed together. ”

Chemical Symbols Chemical symbols are a shorthand for the elements. The chemical symbols are represented on the Periodic Table with their corresponding element. How the symbols can be read: A Chemical Symbol… -begins with a capitalized letter. The first letter MUST be capitalized to denote an element! -any additional letters are lowercase.

States of Matter & Kinetic Molecular Theory The physical form of matter is considered the “State of Matter. ” This describes the spacing and motion of a substance. The most common states of matter are solids, liquids, and gases. Many others exist (plasma, Bose-Einstein condensate, super-fluids, super-solids, etc. . ) but are not applicable to this class. Kinetic Molecular Theory states that all particles are in motion regardless of the state of matter.

States of Matter Solids are a form of matter that have their own definite shape and volume. Liquids are a form of matter that have a definite volume but take the shape of the container. Gases have no definite shape or volume. They expand to fill their container. Vapor refers to the gaseous state of a substance that is a solid or liquid at room temperature.
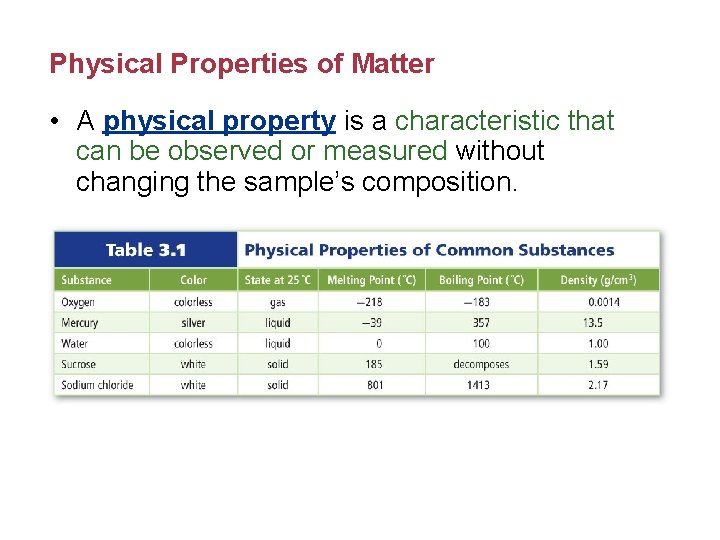
Physical Properties of Matter • A physical property is a characteristic that can be observed or measured without changing the sample’s composition.

Physical Properties of Matter (cont. ) • Extensive properties are dependent on the amount of substance present, such as mass, length, or volume. • Intensive properties are independent of the amount of substance present, such as density.

Chemical Properties of Matter • The ability of a substance to combine with or change into one or more other substances is called a chemical property. – Iron forming rust – Copper turning green in the air

Physical Changes A change that alters a substance without changing its composition is known as a physical change. A phase change is a transition of matter from one state to another. Examples: Boiling, freezing, melting, and condensing all describe phase changes in chemistry.
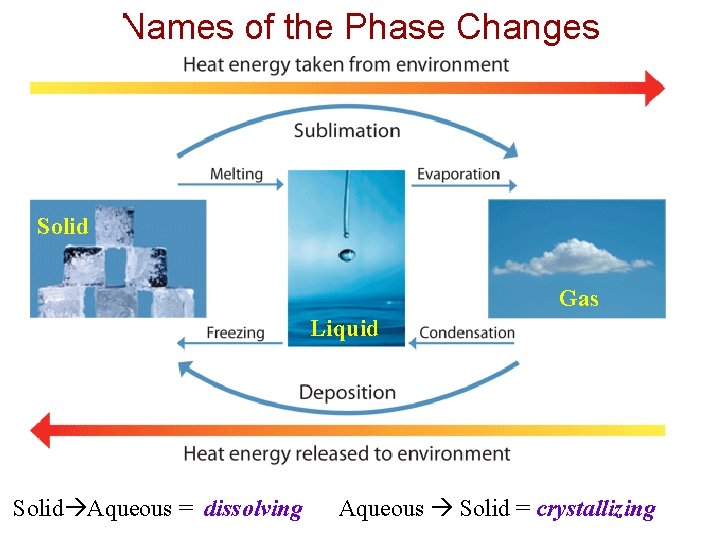
Names of the Phase Changes Solid Gas Liquid Solid Aqueous = dissolving Aqueous Solid = crystallizing

Chemical Changes A change that involves one or more substances turning into new substances is called a chemical change. Examples: Decomposing, rusting, exploding, burning, or oxidizing are all terms that describe chemical changes. Indicators of a chemical change: • • • Light Heat Electricity Gas production Precipitate is produced
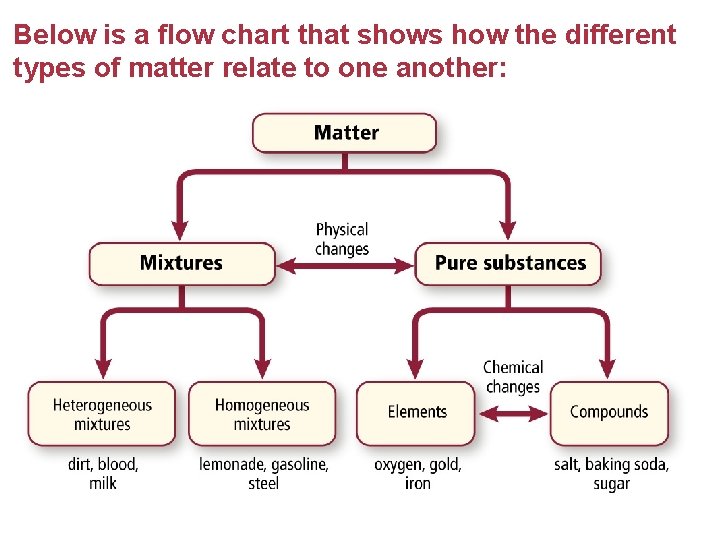
Below is a flow chart that shows how the different types of matter relate to one another:

Energy is the ability to do work or produce heat. Two forms of energy exist, potential and kinetic. • Potential energy is due to composition or position. • Kinetic energy is energy of motion. The law of conservation of energy says in any chemical reaction or physical process, energy can be converted from one form to another, it is neither created nor destroyed
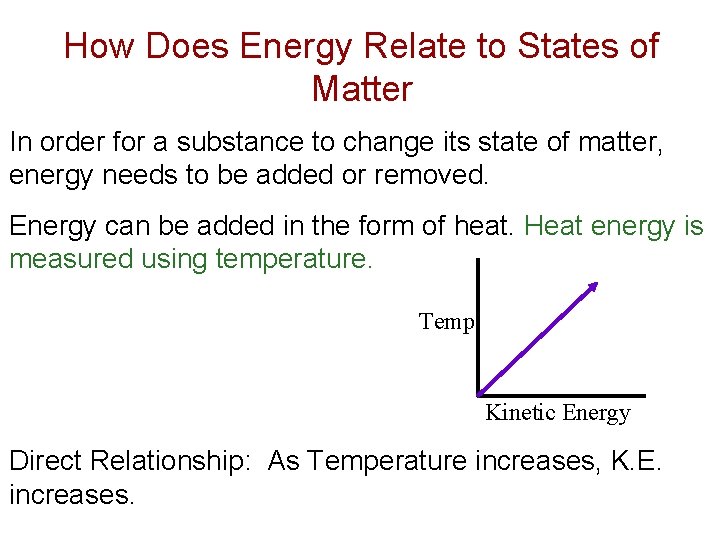
How Does Energy Relate to States of Matter In order for a substance to change its state of matter, energy needs to be added or removed. Energy can be added in the form of heat. Heat energy is measured using temperature. Temp. Kinetic Energy Direct Relationship: As Temperature increases, K. E. increases.

Temperature In science, the units of temperature used are degrees Celsius and Kelvin. Fahrenheit is only used in 3 countries and isn’t practical for scientific use. To switch from Fahrenheit to Celsius use the following equation: 9/5 ºC +32 = ºF More commonly we will need to change Celsius to Kelvin. ºC +273 = Kelvin

Practice Problems: 1. What Kelvin temperature is the freezing point of water, 0˚C? 2. What is 90 K in degrees Celsius. 3. Which phase of water has the highest K. E. ? Lowest K. E? ___
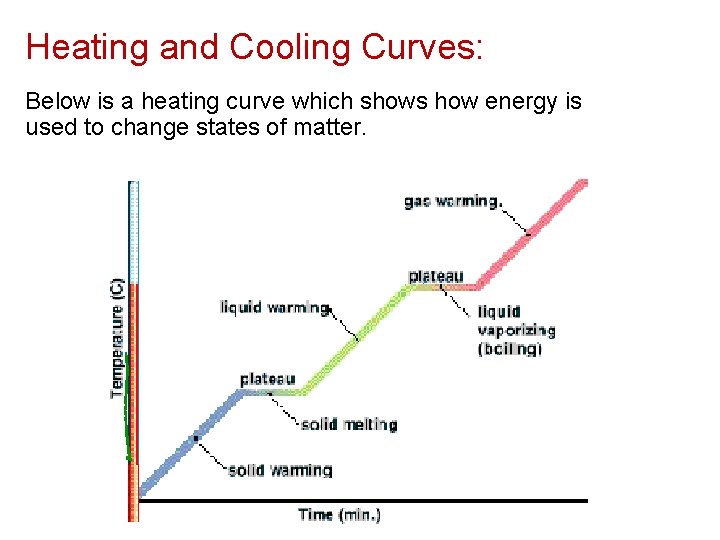
Heating and Cooling Curves: Below is a heating curve which shows how energy is used to change states of matter.

Qualitative vs. Quantitative Qualitative measurements give results in a descriptive nonnumeric form. (The result of a measurement is an adjective describing the object. ) Examples: short , heavy , long, cold, etc… Quantitative measurements give results in numeric form. (The results of a measurement contain a number. ) Examples: 4’ 6”, 600 lbs, 22 meters, 5 ᵒC, etc…
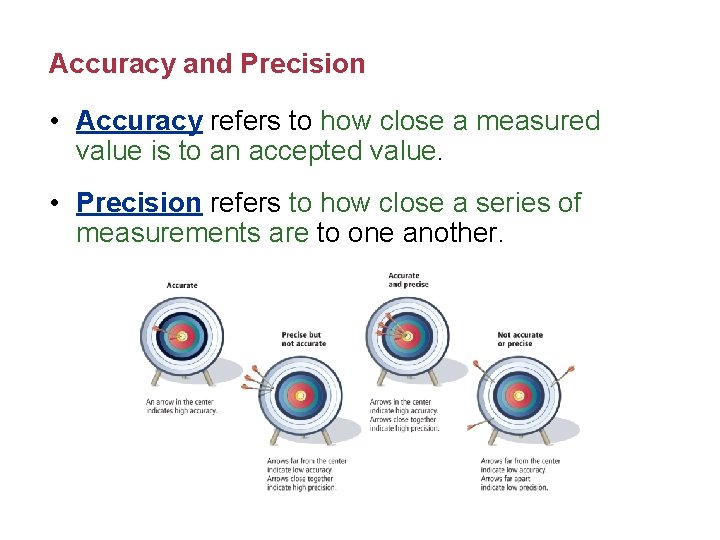
Accuracy and Precision • Accuracy refers to how close a measured value is to an accepted value. • Precision refers to how close a series of measurements are to one another.
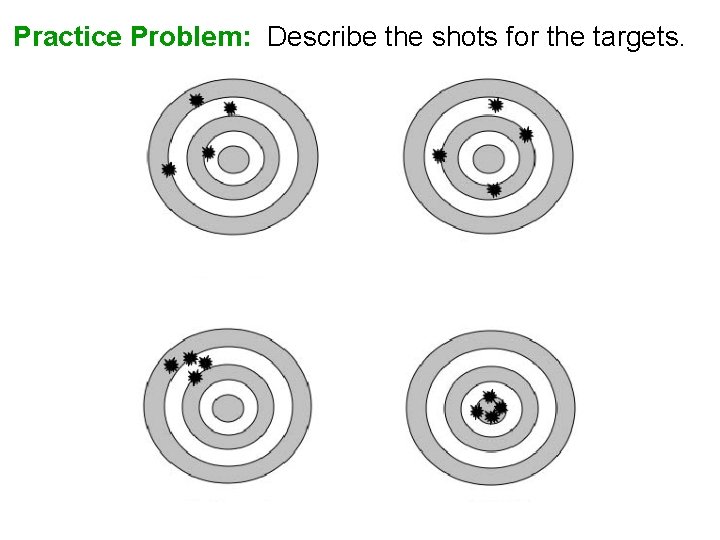
Practice Problem: Describe the shots for the targets.
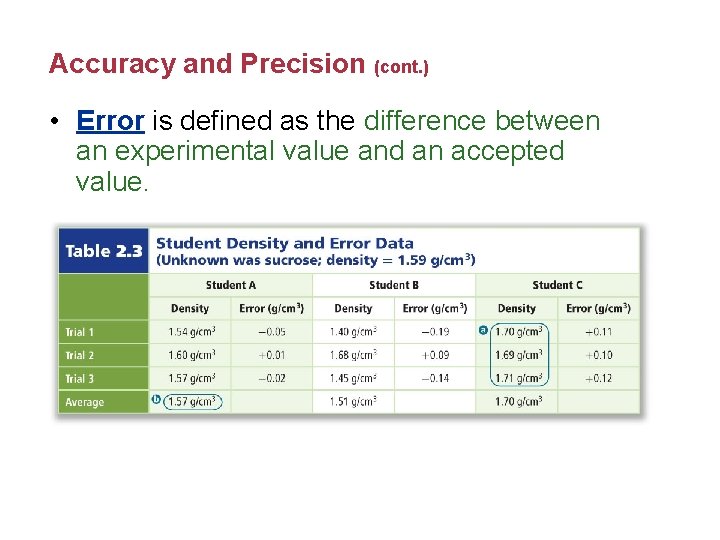
Accuracy and Precision (cont. ) • Error is defined as the difference between an experimental value and an accepted value.

Accuracy and Precision (cont. ) • The error equation is error = experimental value – accepted value • The experimental value is the one determined by actual measurements, whereas the accepted value is the one found in the book or reference. • Percent error expresses error as a percentage of the accepted value.
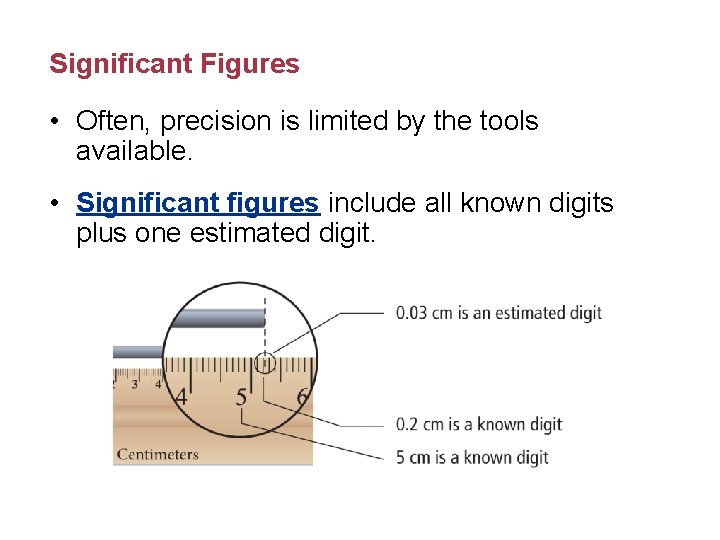
Significant Figures • Often, precision is limited by the tools available. • Significant figures include all known digits plus one estimated digit.

Significant Figures in class In Lab: Whenever a value is measured, (such as the length of an object with a ruler), it must have correct sig figs this means all numbers that are known Record one last digit for the measurement that is estimated. (This means that you will reading in-between the marks of the device and make a thoughtful approximation of what the next number is. )
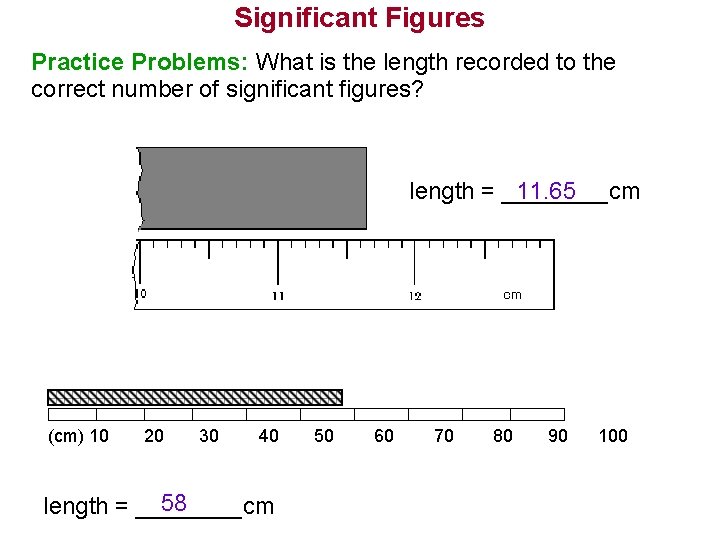
Significant Figures Practice Problems: What is the length recorded to the correct number of significant figures? length = ____cm 11. 65 (cm) 10 20 30 40 58 length = ____cm 50 60 70 80 90 100
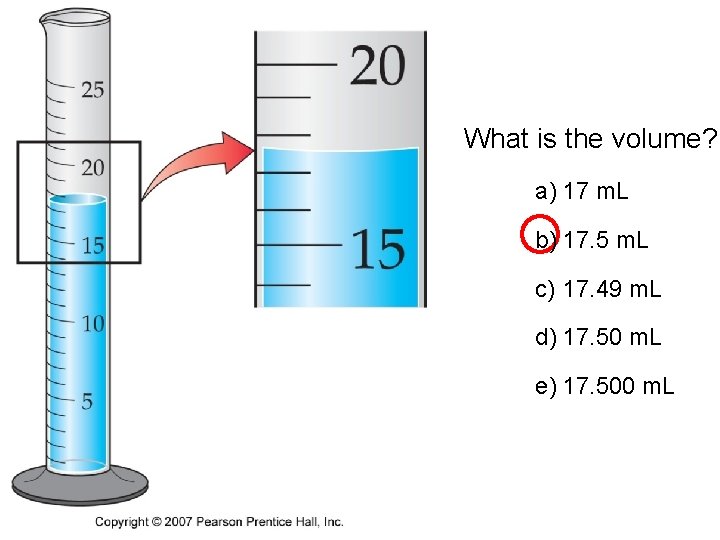
• What is the volume? a) 17 m. L b) 17. 5 m. L c) 17. 49 m. L d) 17. 50 m. L e) 17. 500 m. L
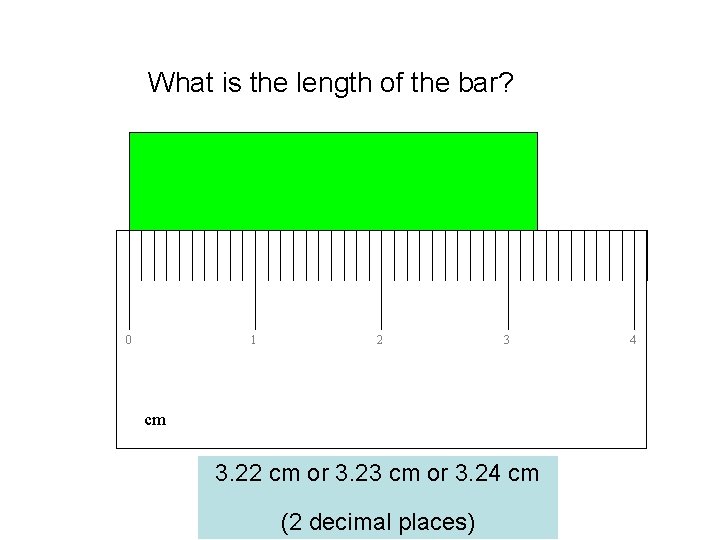
What is the length of the bar? 0 1 2 3 cm 3. 22 cm or 3. 23 cm or 3. 24 cm (2 decimal places) 4

The Box-and-Dot Method: A Simple Strategy for Counting Significant Figures Suppose we want to determine the number of significant digits in the following measurement. In this method, a “dot” is the decimal point. 0. 0020530 meters Step 1 -Draw a box around all nonzero digits, beginning with the leftmost nonzero digit and ending with the rightmost nonzero digit in the number, including any “sandwiched” zeros. 0. 0020530 meters Step 2 - If a dot is present, draw a box around any trailing zeros. 0. 0020530 meters Step 3 - All the boxed digits are significant. (5 sig. figs. )

More Counting Sig. Figs. Practice Problems State the number of significant figures in each measurement. 1) 2804 m 2) 2. 84 km 3) 5. 029 cm 4) 0. 003068 miles 5) 4. 6 x 105 gallons 6) 4. 06 x 10 -5 inches 7) 750 liters 8) 0. 000750 feet 9) 75, 000 seconds 10) 75. 00 meters 11) 75, 000. 0 grams 12) 10 cm

Scientific Notation Scientific notation can be used to express any number as a number between 1 and 10 (the coefficient) multiplied by 10 raised to a power (the exponent). Examples: 5, 203, 000, 000 miles = 5. 203 x 1012 miles 0. 000 042 mm = 4. 2 x 10− 8 mm

Steps for Writing Numbers in Scientific Notation 1. Write down all of the sig. figs. 2. Put the decimal point between the first and second digit. 3. Write “x 10” 4. Count how many places the decimal point has moved from its original location. This will be the exponent. . . either + or −. 5. If the original # was >1, the exponent is (+), and if the original # was <1, the exponent is (-). . (In other words, large numbers have (+) exponents, and small numbers have (-) exponents.

Scientific Notation Practice Problems: Write the following measurements in scientific notation or back to their expanded form. 477, 000 miles = 0. 000 910 m = 6. 30 x 109 miles = 3. 88 x 10− 6 kg =

Rounding Numbers • Calculators are not aware of significant figures. • Answers should not have more significant figures than the original data with the fewest figures, and should be rounded. • Sometimes, scientific notation is needed to get the correct number of significant figures.

Rounding Numbers (cont. ) • Addition and subtraction – Round numbers so all numbers have the same number of digits to the right of the decimal. • Multiplication and division – Round the answer to the same number of significant figures as the original measurement with the fewest significant figures.

Calculations Using Sig. Figs. • There are different rules for rounding off answers to the correct number of sig. figs, but all I need you to do is round off your answers to 3 sig. figs. Examples: 4. 74 cm 4. 739 cm ≈_____ 98. 3 m 2 98. 28 m 2 ≈ _____ 5. 86 g/m. L 5. 855855 g/m. L ≈ ____ = 1. 00 x 104 9, 999 years ≈ 10, 000 ________ years

Rounding Numbers (cont. ) • Rules for rounding – Rule 1: If the digit to the right of the last significant figure is less than 5, do not change the last significant figure. – Rule 2: If the digit to the right of the last significant figure is greater than 5, round up to the last significant figure.
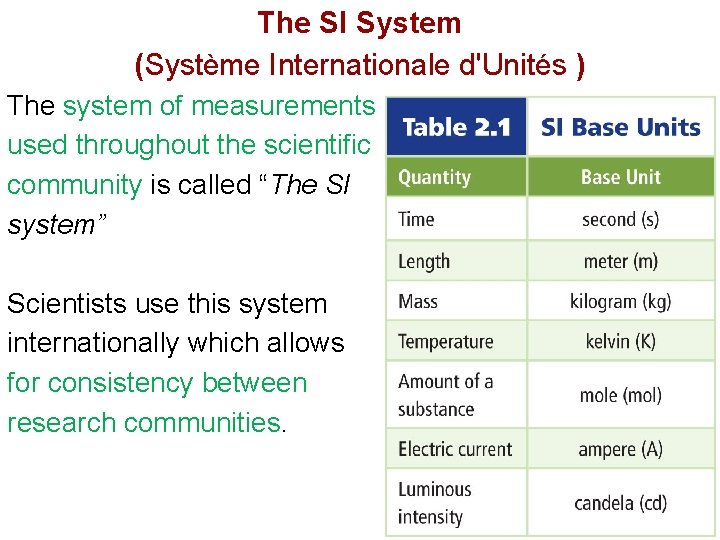
The SI System (Système Internationale d'Unités ) The system of measurements used throughout the scientific community is called “The SI system” Scientists use this system internationally which allows for consistency between research communities.
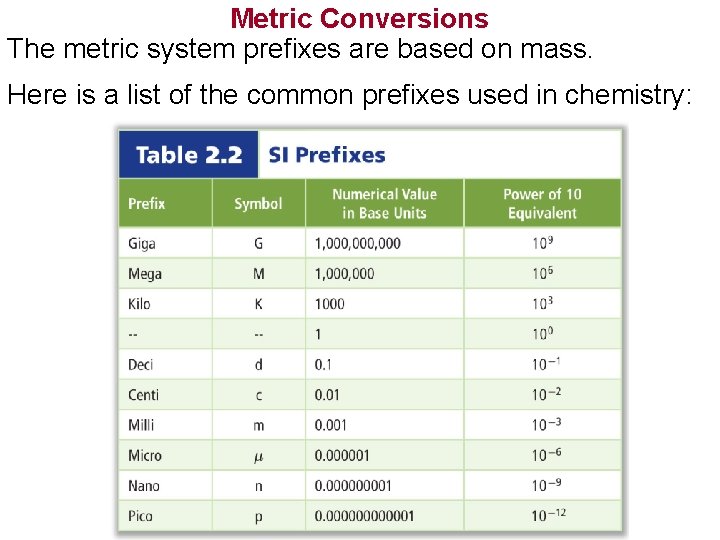
Metric Conversions The metric system prefixes are based on mass. Here is a list of the common prefixes used in chemistry:
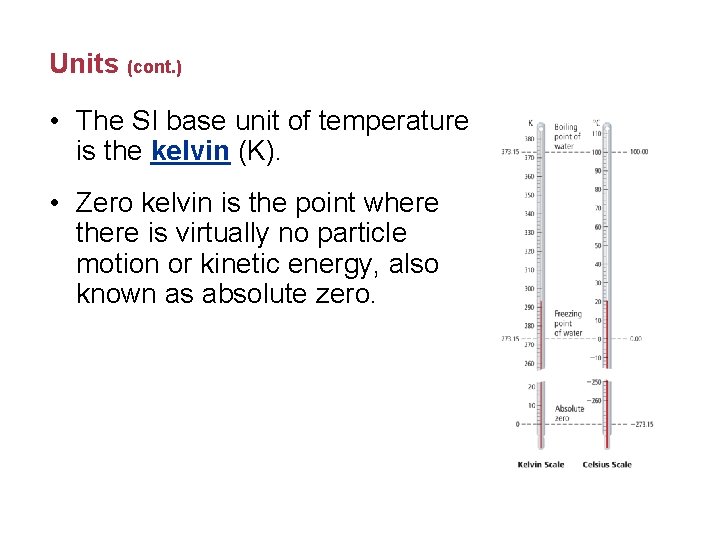
Units (cont. ) • The SI base unit of temperature is the kelvin (K). • Zero kelvin is the point where there is virtually no particle motion or kinetic energy, also known as absolute zero.

Derived Units • Not all quantities can be measured with SI base units. • A unit that is defined by a combination of base units is called a derived unit.
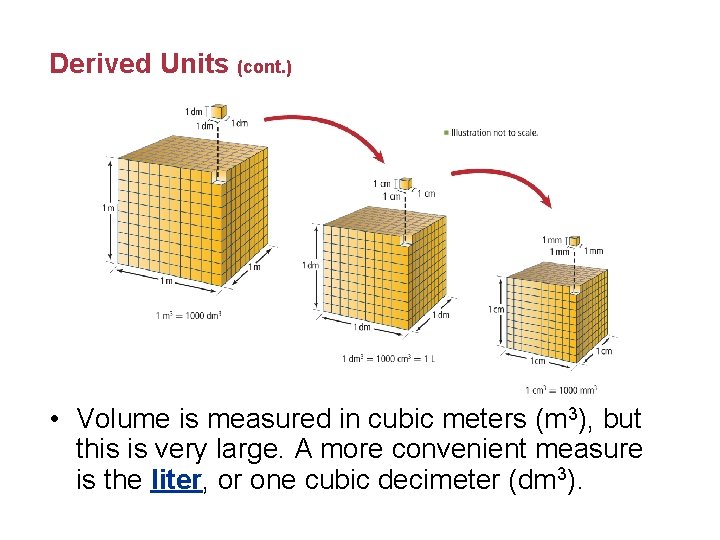
Derived Units (cont. ) • Volume is measured in cubic meters (m 3), but this is very large. A more convenient measure is the liter, or one cubic decimeter (dm 3).

Derived Units (cont. ) • Density is a derived unit, g/cm 3, the amount of mass per unit volume. This is the ratio of mass to volume. • Density is not dependent on the sample size but what the sample is made of. • The density of an object will determine if it will float or sink in another phase. If an object floats, it is less dense than the other substance. If it sinks, it is more dense.

Density Practice Problems 1) What is the density of a piece of wood that has a mass of 25. 0 grams and a volume of 29. 4 cm 3? 2) What is the volume of a piece of aluminum that has a mass of 33. 0 grams and a density of 2. 70 g/m. L?
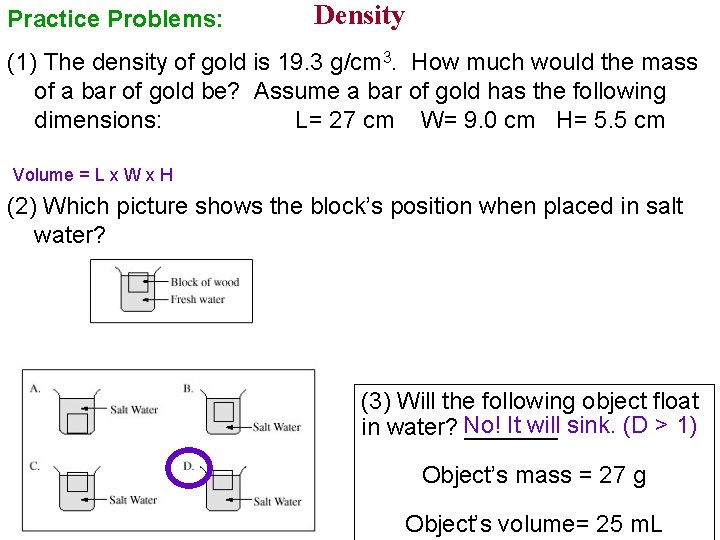
Practice Problems: Density (1) The density of gold is 19. 3 g/cm 3. How much would the mass of a bar of gold be? Assume a bar of gold has the following dimensions: L= 27 cm W= 9. 0 cm H= 5. 5 cm Volume = L x W x H (2) Which picture shows the block’s position when placed in salt water? (3) Will the following object float It will sink. (D > 1) in water? No! _______ Object’s mass = 27 g Object’s volume= 25 m. L
Density Video Clips • Diet Soda: https: //www. youtube. com/watch? v=Mzs. ORE 0 ae 10 • 7 layers: https: //www. youtube. com/watch? v=-CDk. Juo_LYs • Bowling balls: https: //www. youtube. com/watch? v=Lwc 6 Qkl. QGFw • Helium vs. SF 6: https: //www. youtube. com/watch? v=u 19 Qf. JWI 1 o. Q https: //www. youtube. com/watch? v=win. ZR 7 Jqx. Bs

Section 2. 1 Assessment What is the relationship between mass and volume called? A. density B. space C. matter D. weight A. B. C. D. A B C D

Notes Check: What is the difference between accuracy and precision? What are significant figures and why do we use them? What are SI Units? Why do we use scientific notation?

Dimensional Analysis • Dimensional analysis is a systematic approach to problem solving that uses conversion factors to move, or convert, from one unit to another. • A conversion factor is a ratio of equivalent values having different units. Examples: 1 min/ 60 sec (or 60 sec/ 1 min) 7 days/ 1 week (or 1 week/ 7 days) 1000 m/ 1 km (or 1 km/ 1000 m)

Dimensional Analysis (cont. ) • Using conversion factors – A conversion factor must cancel one unit and introduce a new one.

Dimensional Analysis How to Solve Conversion Problems Step 1: Find what you know! Draw a long line and place what you know at the beginning. At the end of the line put an equals sign. Step 2: Identify what you are trying to get. Put the units after the equals sign. Step 3: Identify the conversion factors needed to get to the final units. Step 4: Let’s do a practice problem to figure out the answer. (Make sure your units cancel out) Step 5: Multiply everything on top and divide by everything on the bottom.

Practice Problems: 1. How many seconds are there in 8. 25 hours? 2. How many yards are there in 504 inches? 3. How many days are there in 26, 748 seconds?

Converting “Complex” Units Complex Units are any unit with something in the numerator and denominator (top and bottom of a fraction) example: When doing a problem with complex units: -follow the same steps as before -put the denominator unit beneath the line. -pick one unit to change at a time.

Practice Problems: (1) The speed of sound is about 330 meters/sec. What is the speed of sound in units of miles/hour? (2) The density of water is 1. 0 g/m. L. What is the density of water in units of lbs/gallon? (2. 2 lbs = 1 kg) (3. 78 L = 1 gal) (3) Convert 33, 500 in 2 to m 2 (5280 ft = 1609 m) (12 inches = 1 foot)

Section 2. 2 Assessment What is a systematic approach to problem solving that converts from one unit to another? A. conversion ratio B. conversion factor C. scientific notation D. dimensional analysis A. B. C. D. A B C D

Section 2. 2 Assessment Which of the following expresses 9, 640, 000 in the correct scientific notation? A. 9. 64 104 B. 9. 64 105 C. 9. 64 × 106 D. 9. 64 610 A. B. C. D. A B C D

Section 2. 3 Assessment Determine the number of significant figures in the following: 8, 200, 723. 0, and 0. 01. A. 4, 4, and 3 B. 4, 3, and 3 C. 2, 3, and 1 D. 2, 4, and 1 A. B. C. D. A B C D

Divide 6. 0 109 by 1. 5 103. A. 4. 0 106 B. 4. 5 103 C. 4. 0 103 D. 4. 5 106 A. B. C. D. A B C D

How many significant figures are there in 0. 0000245010 meters? A. 4 B. 5 C. 6 D. 11 A. B. C. D. A B C D

Which is NOT a quantitative measurement of a liquid? A. color B. volume C. mass D. density A. B. C. D. A B C D

Section 2. 1 Units and Measurements Key Concepts • SI measurement units allow scientists to report data to other scientists. • Adding prefixes to SI units extends the range of possible measurements. • To convert to Kelvin temperature, add 273 to the Celsius temperature. K = °C + 273 • Volume and density have derived units. Density, which is a ratio of mass to volume, can be used to identify an unknown sample of matter.

Section 2. 2 Scientific Notation and Dimensional Analysis Key Concepts • A number expressed in scientific notation is written as a coefficient between 1 and 10 multiplied by 10 raised to a power. • To add or subtract numbers in scientific notation, the numbers must have the same exponent. • To multiply or divide numbers in scientific notation, multiply or divide the coefficients and then add or subtract the exponents, respectively. • Dimensional analysis uses conversion factors to solve problems.

Section 2. 3 Uncertainty in Data Key Concepts • An accurate measurement is close to the accepted value. A set of precise measurements shows little variation. • The measurement device determines the degree of precision possible. • Error is the difference between the measured value and the accepted value. Percent error gives the percent deviation from the accepted value. error = experimental value – accepted value

Section 2. 3 Uncertainty in Data (cont. ) Key Concepts • The number of significant figures reflects the precision of reported data. • Calculations should be rounded to the correct number of significant figures.

Section 2. 4 Representing Data Key Concepts • Circle graphs show parts of a whole. Bar graphs show a factor varies with time, location, or temperature. • Independent (x-axis) variables and dependent (y-axis) variables can be related in a linear or a nonlinear manner. The slope of a straight line is defined as rise/run, or ∆y/∆x. • Because line graph data are considered continuous, you can interpolate between data points or extrapolate beyond them.
- Slides: 67