Measurable Quantities T P V Thermodynamic Balances S


Measurable Quantities: T, P, V Thermodynamic Balances: S, H, U, G (Gibbs Free Energy), A (Helmholz Free Energy) Example: DH = Cp DT Cp = (d. H/d. T)p Relate measurable quantities to thermodynamic quantities for balances through differential calculus (materials constants like Cp, Cv, m. JT, ap, k. T and P, V, T).

Simple System: -No Gradients -Reversible -No fields or walls U(S, V) H(S, P)

Gibbs and Helmholtz Free Energies isochoric isobaric
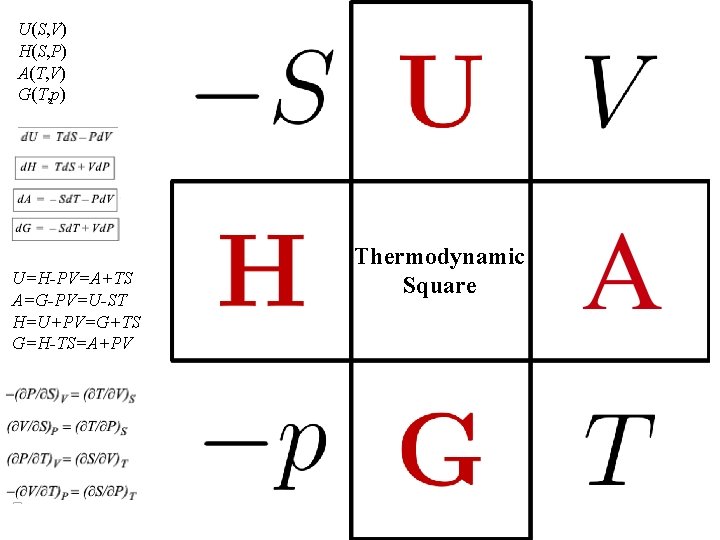
U(S, V) H(S, P) A(T, V) G(T, p) U=H-PV=A+TS A=G-PV=U-ST H=U+PV=G+TS G=H-TS=A+PV Thermodynamic Square


Legendre Transformations From the energy balance on the first slide U depends on S and V: d. U = Td. S - Pd. V (d. U/d. V)S = -P is not useful since we can’t hold S constant very easily it would be more useful to have a different energy expression that depends on V and T rather than V and S. To obtain this we find the desired variable, T = (d. U/d. S)V. T is the conjugate variable of S in the d. U equation. A = U – TS : : d. A = d. U – Td. S –Sd. T : : d. A = -Sd. T - Pd. V The Legendre transform of the d. U equation is d. A = -Sd. T - Pd. V. (d. A/d. V)T = -P

Legendre Transformations Start with: d. A = -Sd. T – Pd. V Depends on V and T P is the conjugate to V. G = A + PV : : d. G = d. A + Pd. V + Vd. P : : d. G = -Sd. T + Vd. P G = PV - ST

Legendre Transformations https: //www. aapt. org/docdirectory/meetingpresentations/SM 14/Mungan. Poster. pdf Accessed 3/2/15




Standard Tabulated Partial Differential Terms for Thermodynamics:

Differential Equalities Needed to Solve Thermodynamic Problems: Expand in two variables, x and y
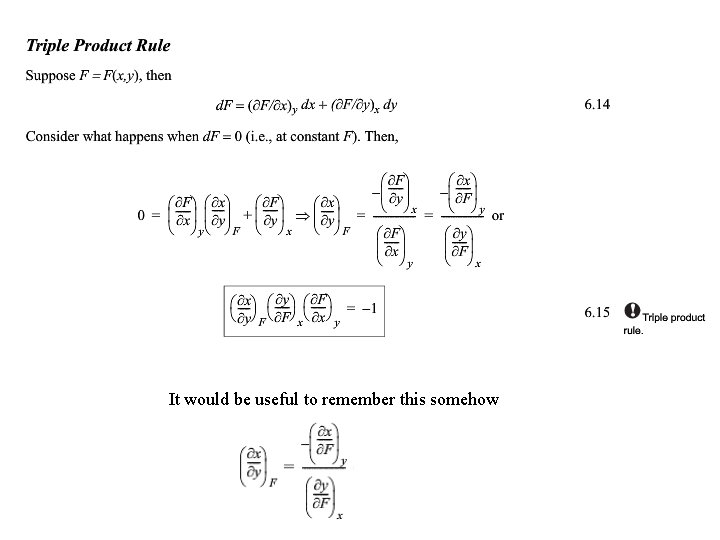
It would be useful to remember this somehow



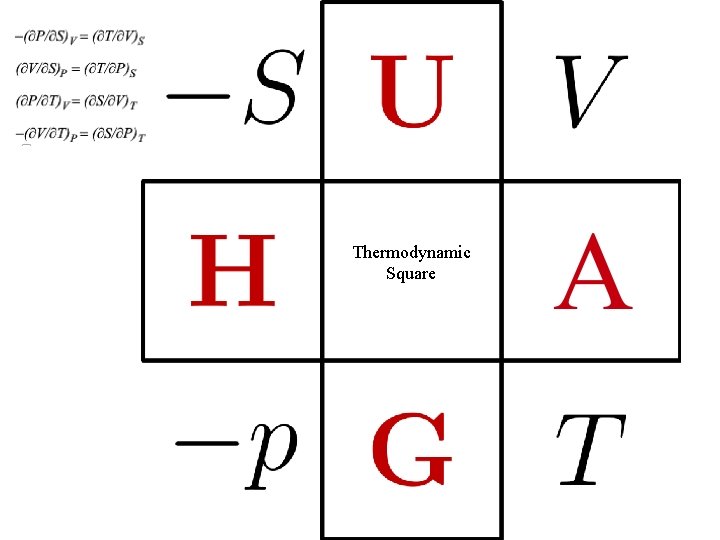
Thermodynamic Square



Example 6. 3 S(T, P) d. S(T, P) = (d. S/d. T)Pd. T + (d. S/d. P)Td. P (d. S/d. T)P=Cp/T And by Maxwell (d. S/d. P)T = -(d. V/d. T)P = -Va. P d. S(T, P) = CPd. T/T - Va. P d. P

From the definitions of Cp and Cv and the chain rule changes in the state functions can be defined in terms of measurable parameters:


-S U V H A -P G T














- Slides: 39