ME 083 Thermodynamic Aside Gibbs Free Energy Professor

ME 083 Thermodynamic Aside: Gibbs Free Energy Professor David M. Stepp Mechanical Engineering and Materials Science 189 Hudson Annex david. stepp@duke. edu 549 -4329 or 660 -5325 24 February 2003

From Last Time…. Gibbs Free Energy: G = H – T*S Recall our Arrhenius relationship for the equilibrium number of vacancies (defects): Frenkel defects (vacancy plus interstitial defect)

THERMODYNAMIC ASIDE: GIBBS FREE ENERGY Energy of a System: A State Function (Depends only on the current condition of the system) Three Categories of Energy: Kinetic (motion), Potential (position), and INTERNAL (molecular motions) State Function Internal Energy: – How can we change a material’s (or system’s) internal energy? ∆U = Q + W’

State Function Internal Energy (U): ∆U = Q + W’ (The First Law of Thermodynamics) Q: Heat flow (into system) W: P-V work (on system) W’: Other work (on system) Energy Conservation Processes in nature have a natural direction of change SPONTANEOUS DOES NOT OCCUR

State Function Entropy (S) ∆SP ≥ 0 The Entropy of a system may increase or decrease during a process. The Entropy of the universe, taken as a system plus surroundings, can only increase. (The Second Law of Thermodynamics) “Entropy is Time’s Arrow” Note: The laws of thermodynamics are empirical, based on considerable experimental evidence.

One Can Show That: For Reversible Processes QREV = Td. S – QREV denotes the maximum heat absorbed – Note the cyclic path integral (reversibility) With this, the combined Notation for the First and Second Law can be expressed: d. U =Td. S + d. WREV + d. W’ BUT d. WREV = F*dx = F*(A/A)*dx = F/A*(-d. V) = -Pd. V d. U = Td. S – Pd. V + d. W’ Combined statement of First and Second Laws

The Third Law of Thermodynamics: The Entropy of all substances is the same at 0 K Both Entropy and Temperature Have Absolute Values (Both have an empirically observed zero point) State Function Enthalpy (H) H = U + P*V So: d. H = d. U + Pd. V + Vd. P Consider the special case where d. P = 0 and d. W’ = 0: d. HP = Td. SP = d. QREV, P

State Function Gibbs Free Energy G = H – TS = U + PV - TS d. G = d. U + Pd. V + Vd. P – Td. S – Sd. T = (Td. S – Pd. V + d. W’) + Pd. V + Vd. P – Td. S – Sd. T = Vd. P – Sd. T + d. W’ Special Case of d. T = 0 and d. P = 0: d. GT, P = d. W’T, P Or, if W’ = 0: d. GP = -Sd. T At constant Temperature and Pressure, the (change in) Gibbs Free Energy reflects all non-mechanical work done on the system.

Back to Crystals… Consider Frenkel Defects (vacancy + interstitial) The Free Energy of the Crystal can be written as – the Free Energy of the perfect crystal (G 0) – plus the free energy change necessary to create n interstitials and vacancies (n*∆g) – minus the entropy increase that derives from the different possible ways in which defects can be arranged (∆SC) ∆G = G-G 0 = n*∆g - T∆SC n = the number of defects ∆g = ∆h - T∆S (the energy to create defects)

The Configurational Entropy, ∆SC, is proportional to the number of ways in which the defects can be arranged (W). ∆SC = k. B* ln(W) (The Boltzmann Equation) Note: this constitutes a connection between atomistic and phenomenological descriptions (thru statistics). Perfect Crystal: N atoms which are indistinguishable can only be placed in one way on the N lattice sites: ∆SC = k*ln(N!/N!) = k*ln(1) = 0

Crystal with Vacancies and Interstitials With N normal lattice sites (and equal interstitial sites), ni (number of interstitial atoms) can be arranged in: distinct ways Similarly, the vacant sites can be arranged in: distinct ways

Recalling that the equilibrium number of vacancies obeys an Arrhenius relationship: Remember: ∆g = ∆h - T∆s Therefore: Assuming configurational entropy in creating defects is negligible Now compare this with our equation for Frenkel Defects:

The Configurational Entropy of these non-interacting defects is: Probability theory for statistically independent events: PAB = PA * PB Stirling’s Approximation for Large Numbers: ln(x!) ≈ x * ln(x) - x Recalling that ni = nv = n (for Frenkel defects):
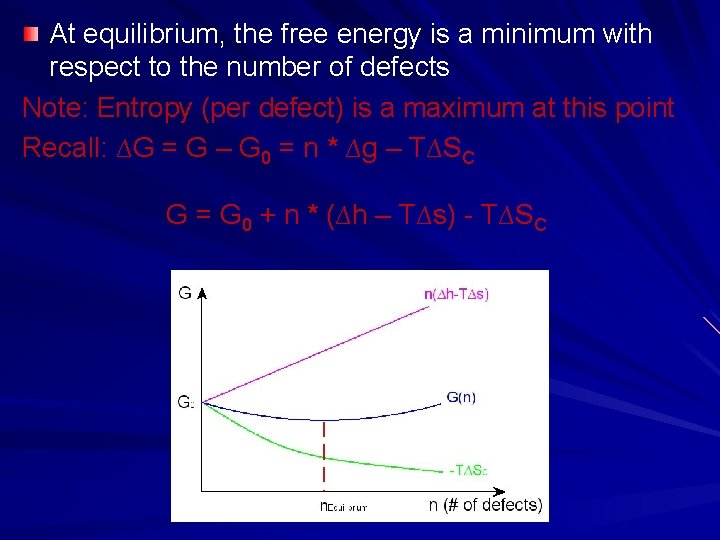
At equilibrium, the free energy is a minimum with respect to the number of defects Note: Entropy (per defect) is a maximum at this point Recall: ∆G = G – G 0 = n * ∆g – T∆SC G = G 0 + n * (∆h – T∆s) - T∆SC

Next Time… The Statistical Interpretation of Entropy ∆SC = k * ln(W) Configurational Entropy is proportional to the number of ways in which defects can be arranged
- Slides: 15