MDS Classification and Advances in Therapy BTG 2013

MDS: Classification and Advances in Therapy BTG 2013 S. Varma PGIMER, Chandigarh India

MDS • Highly heterogeneous group of disorders ▫ Variable natural history ▫ Variable mortality rate ▫ Variable response to therapy • Commonest cause of death ▫ Progressive bone marrow failure ▫ Conversion to AML
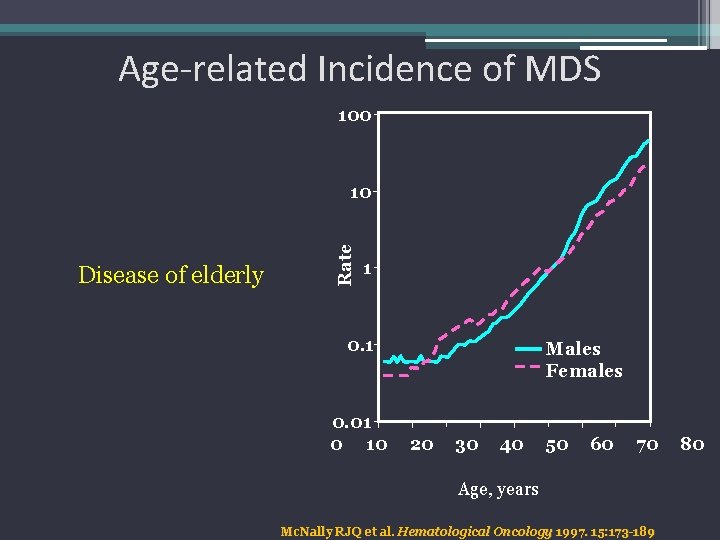
Age-related Incidence of MDS 100 Disease of elderly Rate 10 1 0. 01 0 10 Males Females 20 30 40 50 60 70 Age, years Mc. Nally RJQ et al. Hematological Oncology 1997. 15: 173 -189 80

Classification

Historical Perspective • Pseudo-aplastic anemia • Refractory Anemia • Pre-leukemia • Myelodysplastic syndrome Luzzatto AM. anemia pseudoaplastica Riv Ven 1907; 47: 193. Bomford RR, Rhodes CP. Refractory anemia. Q J Med 1941; 10: 175 -281.
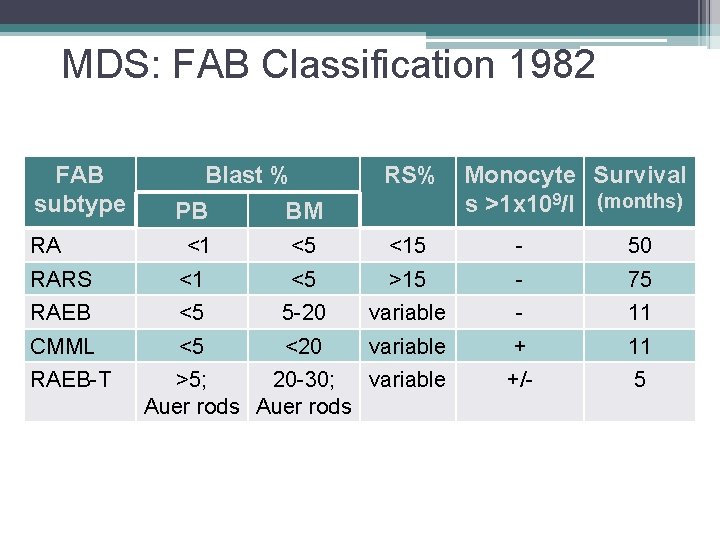
MDS: FAB Classification 1982 FAB subtype RA Blast % PB BM <1 RS% Monocyte Survival s >1 x 109/l (months) <5 <15 - 50 RARS <1 <5 >15 - 75 RAEB <5 5 -20 variable - 11 CMML <5 <20 variable + 11 +/- 5 RAEB-T >5; 20 -30; variable Auer rods

MDS: Limitations of FAB Classification • • Multilineage cytopenia with <5% BM blasts Rough prediction of prognosis Cytogenetics not given importance Ill defined entities: childhood MDS, T-MDS & other secondary MDS • Immunophenotyping and genetic techniques not included

Comparison of MDS Classifications FAB classification WHO Classsification 2001 WHO Classification 2008 RA RA Refractory cytopenia with unilineage dyplasia • Refractory anemia • Refractory neutropenia • Refractory thrombocytopenia RARS Refractory anemia with ring sideroblasts (RARS) RCMD-RS RAEB I and 2 RAEB-T RAEB II/ AML CMML MDS-UC MDS associated with isolated del(5 q) Childhood myelodysplastic syndrome • Refractory cytopenia of childhood
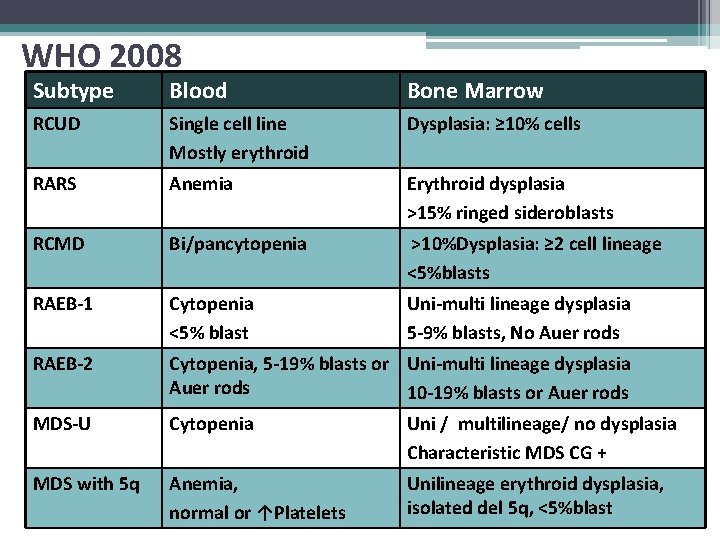
WHO 2008 Subtype Blood Bone Marrow RCUD Single cell line Mostly erythroid Dysplasia: ≥ 10% cells RARS Anemia Erythroid dysplasia >15% ringed sideroblasts RCMD Bi/pancytopenia >10%Dysplasia: ≥ 2 cell lineage <5%blasts RAEB-1 Cytopenia <5% blast Uni-multi lineage dysplasia 5 -9% blasts, No Auer rods RAEB-2 Cytopenia, 5 -19% blasts or Uni-multi lineage dysplasia Auer rods 10 -19% blasts or Auer rods MDS-U Cytopenia Uni / multilineage/ no dysplasia Characteristic MDS CG + MDS with 5 q Anemia, normal or ↑Platelets Unilineage erythroid dysplasia, isolated del 5 q, <5%blast
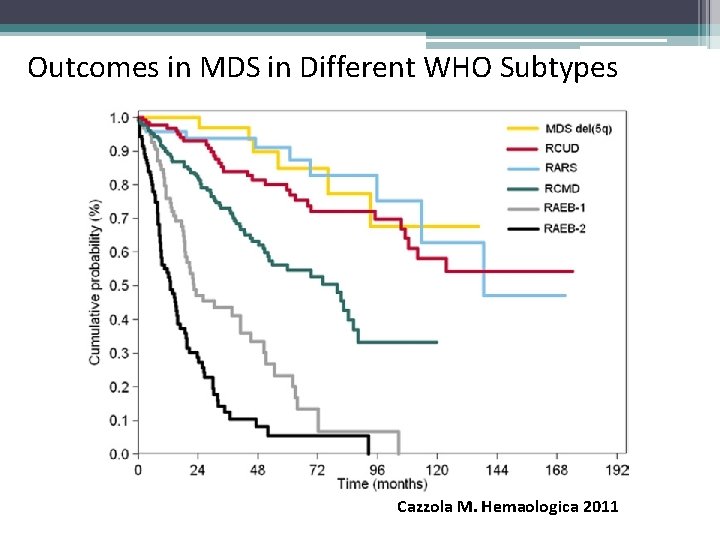
Outcomes in MDS in Different WHO Subtypes Cazzola M. Hemaologica 2011

Advances in Management Improved prognostic scores Disease related variables Host factors Appropriate clinical decision Disease eradication/ control Prolonging overall survival Managing complications of disease and therapy Improving quality of life
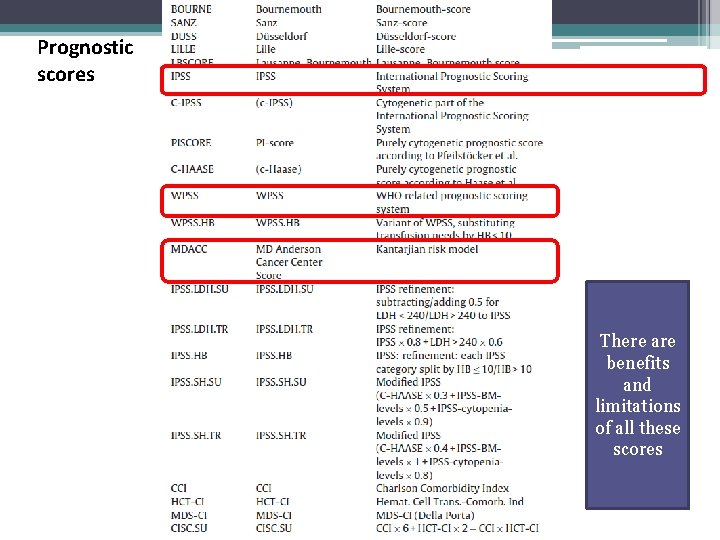
Prognostic scores Most widely used There are benefits and limitations of all these scores
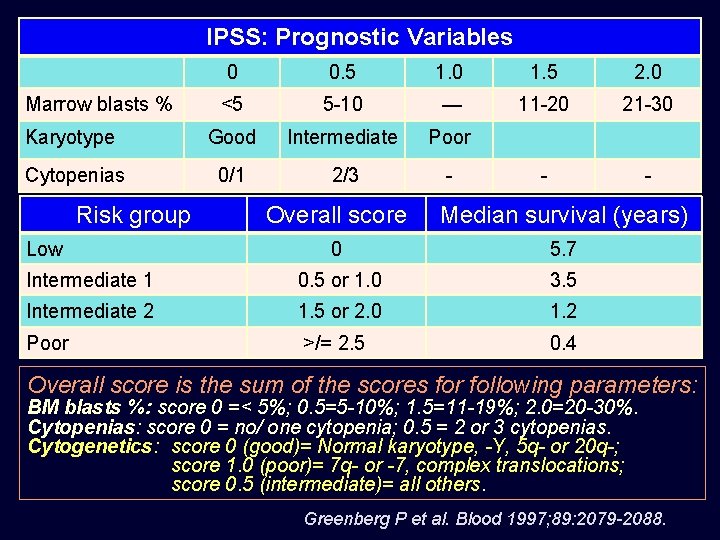
IPSS: Prognostic Variables 0 0. 5 1. 0 1. 5 2. 0 <5 5 -10 — 11 -20 21 -30 Karyotype Good Intermediate Poor Cytopenias 0/1 2/3 - - - Marrow blasts % Risk group Overall score Median survival (years) 0 5. 7 Intermediate 1 0. 5 or 1. 0 3. 5 Intermediate 2 1. 5 or 2. 0 1. 2 Poor >/= 2. 5 0. 4 Low Overall score is the sum of the scores for following parameters: BM blasts %: score 0 =< 5%; 0. 5=5 -10%; 1. 5=11 -19%; 2. 0=20 -30%. Cytopenias: score 0 = no/ one cytopenia; 0. 5 = 2 or 3 cytopenias. Cytogenetics: score 0 (good)= Normal karyotype, -Y, 5 q- or 20 q-; score 1. 0 (poor)= 7 q- or -7, complex translocations; score 0. 5 (intermediate)= all others. Greenberg P et al. Blood 1997; 89: 2079 -2088.
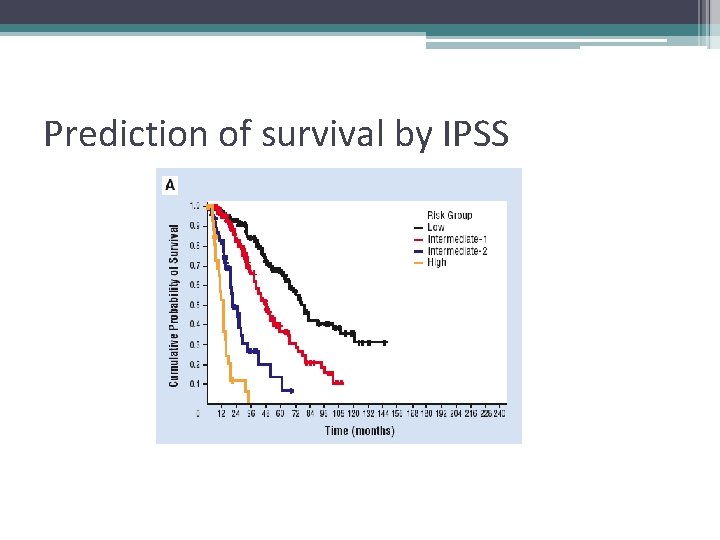
Prediction of survival by IPSS

IPSS Pros • Simplicity: ▫ Use of only 3 variables • Applicable at centers with limited lab support • Widely used in clinical practice and research ▫ Bulk of scientific data on MDS is based of IPSS Cons • Includes patients with ▫ 20 -30% blasts ▫ CMML • Does not consider severity of cytopenias ▫ Strong predictor of outcome • Can not be applied in pre-treated patients

WHO Prognostic Scoring System *BM fibrosis grade 2 -3 shifts risk group by one step

WPSS Pros Cons • Simplicity: use of only 3 variables • Accurate prediction of survival and risk of leukemic evolution at any time during the course of their disease • Useful in predicting post transplant outcome • Not applicable for secondary MDS
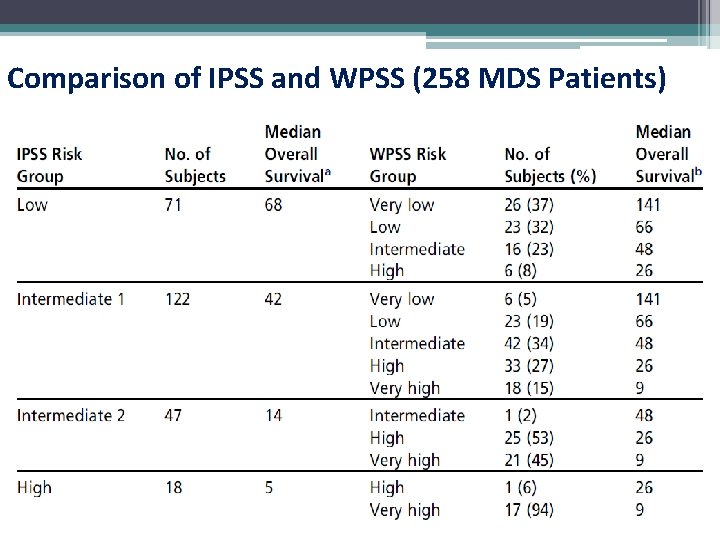
Comparison of IPSS and WPSS (258 MDS Patients)
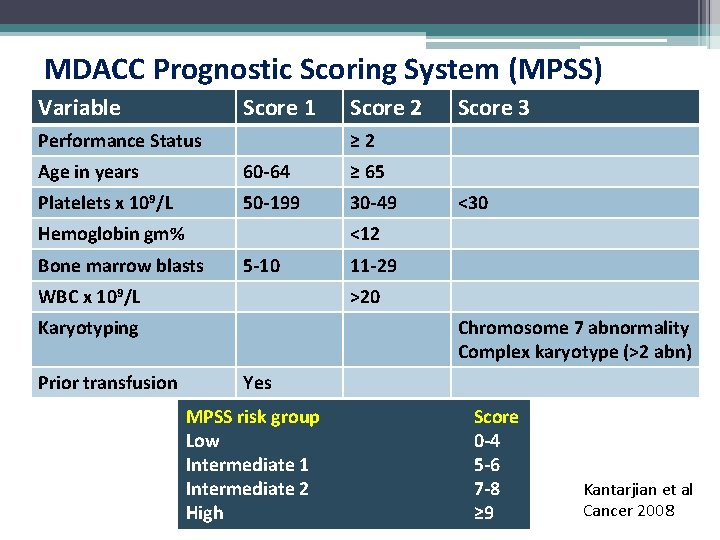
MDACC Prognostic Scoring System (MPSS) Variable Score 1 Performance Status Score 2 ≥ 2 Age in years 60 -64 ≥ 65 Platelets x 109/L 50 -199 30 -49 Hemoglobin gm% <30 <12 Bone marrow blasts 5 -10 WBC x 109/L 11 -29 >20 Karyotyping Prior transfusion Score 3 Chromosome 7 abnormality Complex karyotype (>2 abn) Yes MPSS risk group Low Intermediate 1 Intermediate 2 High Score 0 -4 5 -6 7 -8 ≥ 9 Kantarjian et al Cancer 2008

2012 Revised IPSS Fine tune the prognostic impact of • Cytogenetic abnormalities • Depth of cytopenia Prognostic Subgroup Cytogenetic Abnormality Median OS, Mos Median Time to AML, Mos del(11 q), -Y 60. 8 NR Good Normal, del(20 q), del(5 q) alone or double, del(12 p) 48. 6 NR Intermediate +8, del(7 q), i(17 q), +19, any other single or double, independent clones 26. 0 78. 0 inv(3)/t(3 q)/del(eq), -7, double including del(7 q), complex (3) 15. 8 21. 0 complex (≥ 3) 5. 9 8. 2 Very good Poor Very poor Schanz J, et al. J Clin Oncol. 2012; 30: 820 -829. Greenberg PL, et al. Blood. 2012; 120: 2454 -2465.
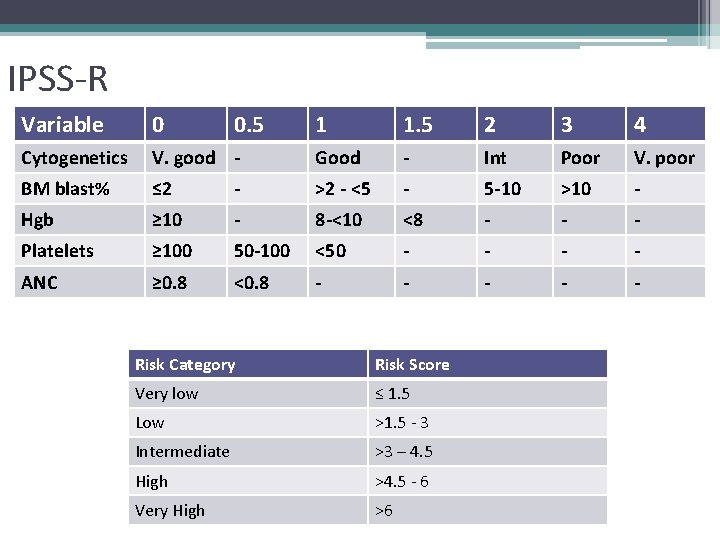
IPSS-R Variable 0 Cytogenetics 0. 5 1 1. 5 2 3 4 V. good - Good - Int Poor V. poor BM blast% ≤ 2 - >2 - <5 - 5 -10 >10 - Hgb ≥ 10 - 8 -<10 <8 - - - Platelets ≥ 100 50 -100 <50 - - ANC ≥ 0. 8 <0. 8 - - - Risk Category Risk Score Very low ≤ 1. 5 Low >1. 5 - 3 Intermediate >3 – 4. 5 High >4. 5 - 6 Very High >6

Advances in therapy of MDS

Treatment considerations • Myelodysplasia are incurable without HSCT • Highly variable natural history • Treatment considerations must take into account many factors, including the ▫ ▫ Pathologic diagnosis The prognosis based on the IPSS or WPSS Cell line /s affected Feasibility of performing a clinical trial

Tools to treat MDS • • • Observation Supportive therapy (Transfusions) Hematopoietic growth factors Iron chelation Lenalidomide (Revlimid 2005) Hypomethylating agents ▫ Azacitidine (Vidaza 2004) ▫ Decitabine (Dacogen 2006) • Immunosuppression • Allogeneic stem cell transplantation • Newer agents

To Trick or Treat • Treatment should be reserved and potentially diagnosis to be transmitted to the patient and family, only if there are symptoms resulting from anemia or other cytopenias or perhaps pre-symptomatic anemia or severe thrombocytopenia. • Old and frail patients or those who have equivocal diagnostic features, benefit from a period of observation. • Neutropenia without infection is a poor justification for initiation of therapy. Stone RM. Blood 2009
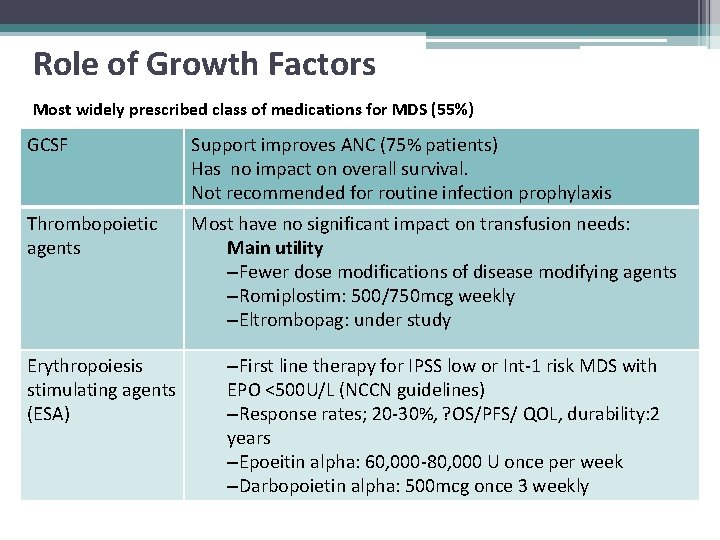
Role of Growth Factors Most widely prescribed class of medications for MDS (55%) GCSF Support improves ANC (75% patients) Has no impact on overall survival. Not recommended for routine infection prophylaxis Thrombopoietic agents Most have no significant impact on transfusion needs: Main utility –Fewer dose modifications of disease modifying agents –Romiplostim: 500/750 mcg weekly –Eltrombopag: under study Erythropoiesis stimulating agents (ESA) –First line therapy for IPSS low or Int-1 risk MDS with EPO <500 U/L (NCCN guidelines) –Response rates; 20 -30%, ? OS/PFS/ QOL, durability: 2 years –Epoeitin alpha: 60, 000 -80, 000 U once per week –Darbopoietin alpha: 500 mcg once 3 weekly

Newer approaches. Immunosuppressants Immunologic suppression of normal BM function, similar to the situation in aplastic anemia, has been postulated to account for cytopenias in some MDS patients Specific candidates- Refractory anemia with relatively hypoplastic marrow

Predictor of Response to Immunosuppressant • HLA-DR-15 -positivity • RA (<5% blasts) • IPSS Low/Int-1 • Age <60 years • Brief transfusion history • Trisomy 8 abnormality • Normal cytogenetics • Marrow cellularity <30%

ATG • Phase II study (N=35) on MDS-RA • Both equine and rabbit ATG were found to be active • Response to ▫ Equine ATG: 29% (34/115) ▫ Rabbit ATG: RR 42%. ▫ 75% responders durable response (median 31. 5 months). Stadler M, Leukemia 2004; 18: 460 -5 Jonasova A, Br J Haematol. 1998; 100: 304 -309. Molldrem JJ, Br J Haematol. 1997; 99: 699 -705.

Chromosomal Abnormality: del 13 q 22 patients with bone marrow failure MDS U Only del (13 q ) Del (13 q) plus other abnormalities number 16 6 GPI def clone 16 3 Response to IST 100% (14/14) 40% (2/5) 10 yr OSR 83% 63% Progression to AML None 2 • MDS-U with del (13 q) is a benign disorder with good response to IST • Del (13 q) should not be considered intermediate risk abnormality Hosokawa et al, Haematologica 2012; 97: 1845

Biological response modifiers special case of Del 5 q syndrome Eligibility: • del(5 q) • IPSS low or Int-1 • platelets > 50 K/mm 3 • neutrophils > 500/mm 3 • transfusion dependent
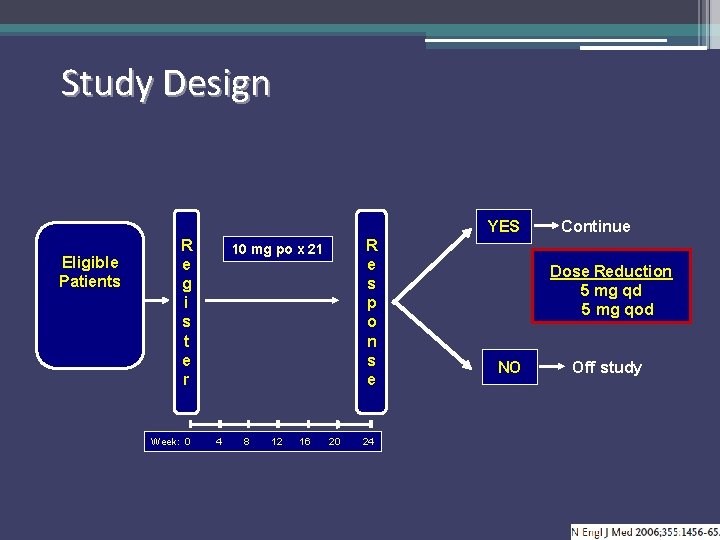
Study Design YES Eligible Patients R e g i s t e r Week: 0 R e s p o n s e 10 mg po x 21 4 8 12 16 20 24 Continue Dose Reduction 5 mg qd 5 mg qod NO Off study

Results Frequency of Cytogenetic Response According to Karyotype Complexity
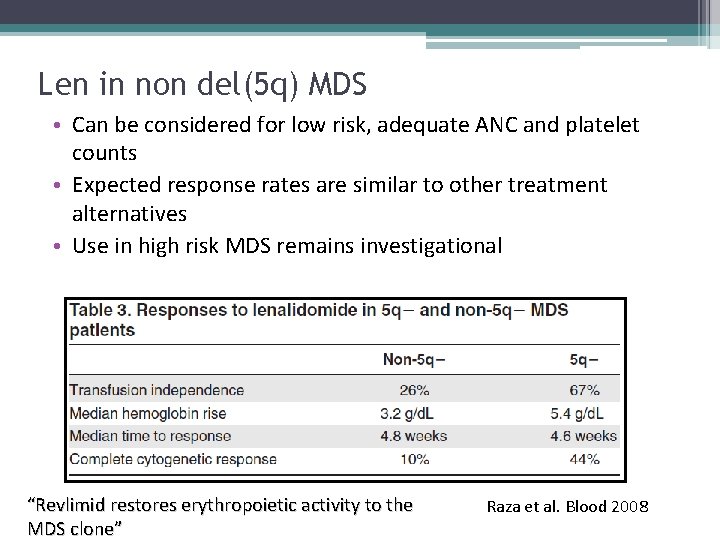
Len in non del(5 q) MDS • Can be considered for low risk, adequate ANC and platelet counts • Expected response rates are similar to other treatment alternatives • Use in high risk MDS remains investigational “Revlimid restores erythropoietic activity to the MDS clone” Raza et al. Blood 2008

Hypomethylating agents
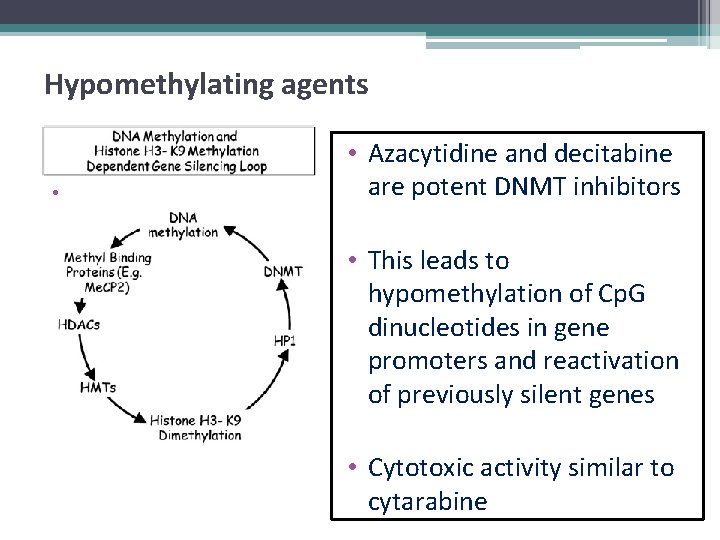
Hypomethylating agents • • Azacytidine and decitabine are potent DNMT inhibitors • This leads to hypomethylation of Cp. G dinucleotides in gene promoters and reactivation of previously silent genes • Cytotoxic activity similar to cytarabine
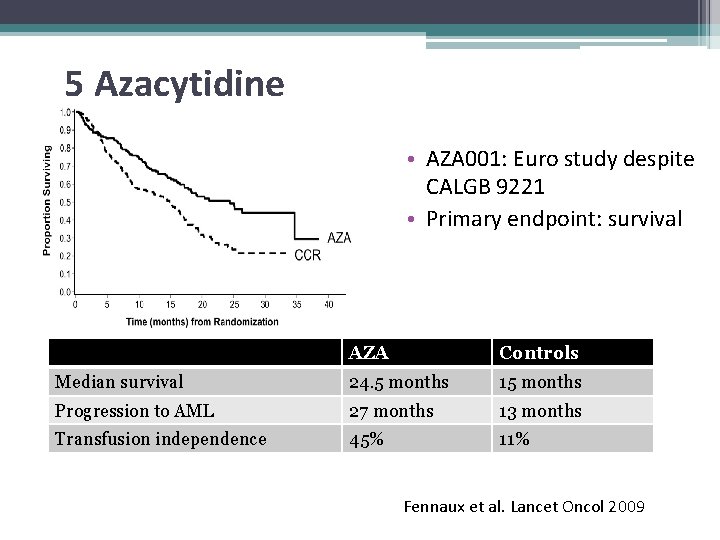
5 Azacytidine • AZA 001: Euro study despite CALGB 9221 • Primary endpoint: survival AZA Controls Median survival 24. 5 months 15 months Progression to AML 27 months 13 months Transfusion independence 45% 11% Fennaux et al. Lancet Oncol 2009
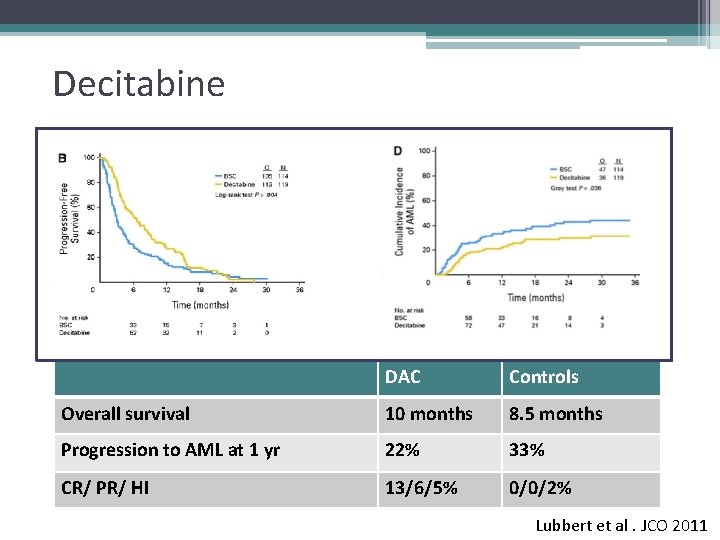
Decitabine DAC Controls Overall survival 10 months 8. 5 months Progression to AML at 1 yr 22% 33% CR/ PR/ HI 13/6/5% 0/0/2% Lubbert et al. JCO 2011

Hypomethylating agents When to start – Int/ high risk MDS (IPSS) – Transfusion dependent/ EPO failure – Not yet known if early treatment is better than late treatment in MDS Which drug – NCCN recommends Azacitidine preference over Decitabine – EORTC study failed to show survival benefit. – MDACC regimen (5 day 20 mg/m 2/d) highest CR – Aza vs Decitabine head to head trial results awaited Optimal dose, schedule, route – Azacitidine: – 7 day 75 mg/m 2/d sc q 28 days (5 -2 -2 or 50 mg/m 2 5 -2 -5 schedule) – Decitabine: – 3 day 15 mg/m 2/dose IV 8 hrly (total dose 135 mg/m 2) inpatient – 5 day 20 mg/m 2 /d over 1 hr (total dose 100 mg/m 2) outpatient Duration – Optimal duration- not known – To treat responding pts till disease progression, as long as tolerated – At least 4 cycles recommended for adequate response Steensma et al. Hematol Oncol clin N Am 2010

Predictive Factors for Achieving Response to Hypomethylating Agents Positive • Negative • Mol/ Cyto: ▫ Mutated TET 2 ▫ Mutated EZH 2 ▫ Phosphoinositase – Phospholipase C beta 1 hypomethylation • Clinical Variable ▫ Doubling of Platelets • Mutated P 53 • Abnormal/ complex karyotype • • BM Blasts >15% Previous therapy Transfusion dependency BM fibrosis grade 3 Santini V, ASH 2012
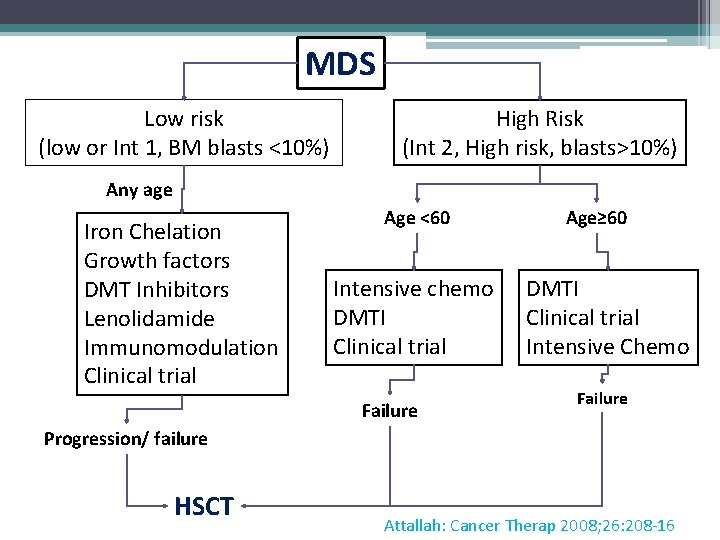
MDS Low risk (low or Int 1, BM blasts <10%) High Risk (Int 2, High risk, blasts>10%) Any age Iron Chelation Growth factors DMT Inhibitors Lenolidamide Immunomodulation Clinical trial Age <60 Intensive chemo DMTI Clinical trial Failure Age≥ 60 DMTI Clinical trial Intensive Chemo Failure Progression/ failure HSCT Attallah: Cancer Therap 2008; 26: 208 -16

Unconventional and upcoming agents

What’s on the Horizon? • In the quest of effective therapy, currently there approximately 200 clinical trials are ongoing and numerous agents are at various stages of drug development • The need for a novel agent is particularly noted in patients failing hypomethylating agents who are ineligible for stem cell transplant Kulasekararaj AG, Semin Hematol , 2012; 49: 350 -60
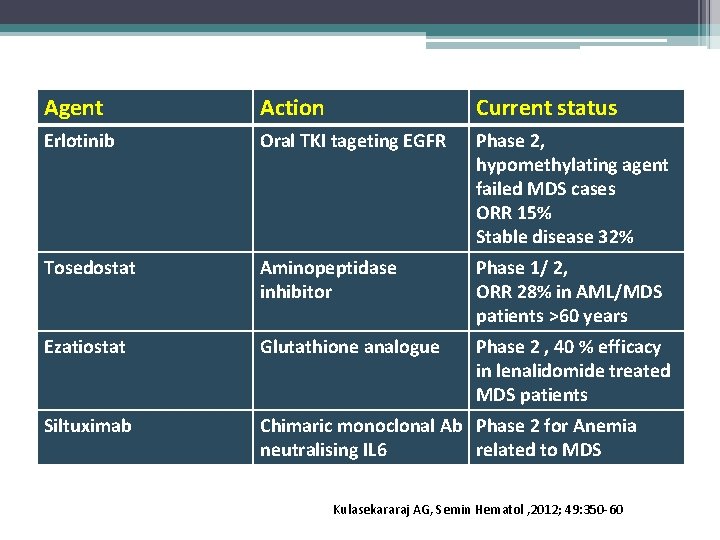
Agent Action Current status Erlotinib Oral TKI tageting EGFR Phase 2, hypomethylating agent failed MDS cases ORR 15% Stable disease 32% Tosedostat Aminopeptidase inhibitor Phase 1/ 2, ORR 28% in AML/MDS patients >60 years Ezatiostat Glutathione analogue Phase 2 , 40 % efficacy in lenalidomide treated MDS patients Siltuximab Chimaric monoclonal Ab Phase 2 for Anemia neutralising IL 6 related to MDS Kulasekararaj AG, Semin Hematol , 2012; 49: 350 -60
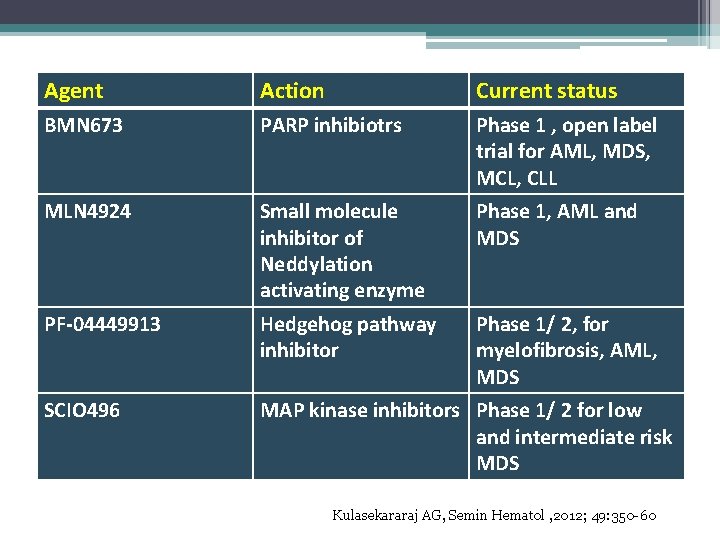
Agent Action Current status BMN 673 PARP inhibiotrs Phase 1 , open label trial for AML, MDS, MCL, CLL MLN 4924 Small molecule inhibitor of Neddylation activating enzyme Phase 1, AML and MDS PF-04449913 Hedgehog pathway inhibitor Phase 1/ 2, for myelofibrosis, AML, MDS SCIO 496 MAP kinase inhibitors Phase 1/ 2 for low and intermediate risk MDS Kulasekararaj AG, Semin Hematol , 2012; 49: 350 -60

Take Home Message • Myelo-dysplastic syndromes are heterogeneous disorders • Prognostic scores are evolving with use of cytogenetics and molecular markers • Treatment depends upon the prognostic and host factors • MTI and IMIDs are being increasingly used • HSCT is the only curative treatment • Treatment paradigms are evolving
- Slides: 46