MDEpi Net An Update Danica MarinacDabic MD Ph

MDEpi. Net : An Update Danica Marinac-Dabic, MD, Ph. D Director, Division of Epidemiology 11/30/2020

NEED FOR A MEDICAL DEVICE METHODOLOGY AND INFRASTUCTURE PORTFOLIO Data that informs post-market safety and effectiveness of medical devices are: 1. Hierarchically structured (patients nested within operators) 2. Safety and effectiveness signals are confounded with operator learning effects 3. Pre-market evidence based on smaller sample sizes (relative to drug trials) – 4. 5. 6. Subgroup effects harder to discern! Require innovative linking techniques to identify specific device (lack of UDI) Include information to inform about device improvements – Requires innovative harvesting of particular features to promote the TPLC Global information: how to capitalize on world-wide information to inform safety and effectiveness?

2010: FDA MDEpi. Net Initiative Goal: To develop national /international infrastructure and innovative methodological approaches for conducting robust studies and surveillance to improve medical device safety and effectiveness understanding throughout the device life cycle through Public Private Partnership with academia and other stakeholders. 11/30/2020
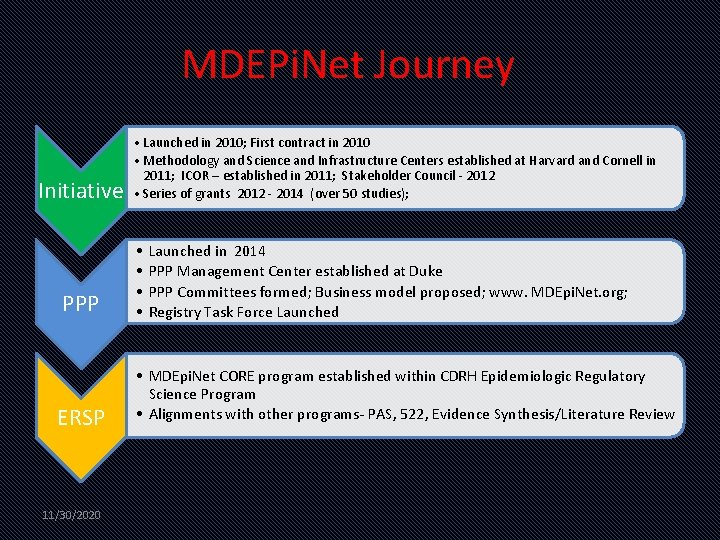
MDEPi. Net Journey Initiative PPP ERSP 11/30/2020 • Launched in 2010; First contract in 2010 • Methodology and Science and Infrastructure Centers established at Harvard and Cornell in 2011; ICOR – established in 2011; Stakeholder Council - 2012 • Series of grants 2012 - 2014 (over 50 studies); • • Launched in 2014 PPP Management Center established at Duke PPP Committees formed; Business model proposed; www. MDEpi. Net. org; Registry Task Force Launched • MDEpi. Net CORE program established within CDRH Epidemiologic Regulatory Science Program • Alignments with other programs- PAS, 522, Evidence Synthesis/Literature Review
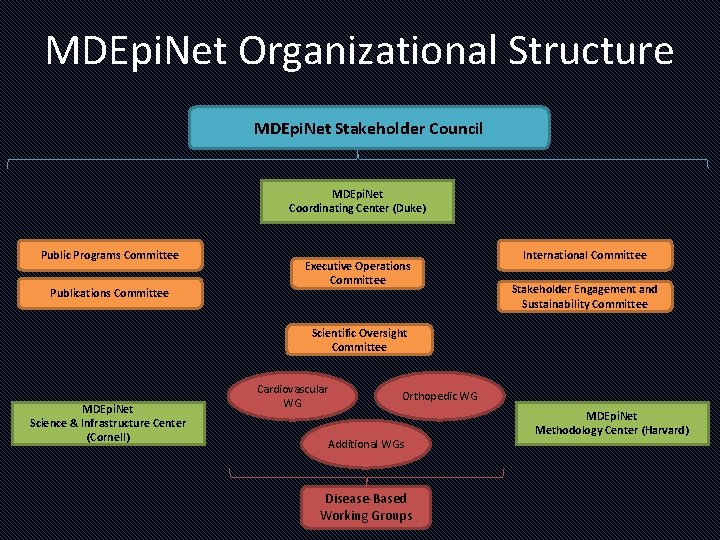
MDEpi. Net Organizational Structure MDEpi. Net Stakeholder Council MDEpi. Net Coordinating Center (Duke) Public Programs Committee Publications Committee Executive Operations Committee International Committee Stakeholder Engagement and Sustainability Committee Scientific Oversight Committee MDEpi. Net Science & Infrastructure Center (Cornell) Cardiovascular WG Orthopedic WG Additional WGs Disease-Based Working Groups MDEpi. Net Methodology Center (Harvard)


Creating the Infrastructure Portfolio Example: International Consortia Development § International Consortium of Orthopedic Registries (ICOR) § Global partnership – 29 registries – 8 contributing data – 4 million implants; CER and surveillance (27 papers) § Methods for combining de-identified data § International Consortium of Cardiac Registries (ICCR) § International Consortium of Vascular Registries (ICVR) § Building on two multi-stakeholder national registry efforts (valve and vascular space) § Plan for conversion of studies to novel surveillance § Will be basis for real - time active surveillance; Allow considerations of expanding indications; Contribute to assessment of pre-post balance; Lead to less costly PAS; UDI links to clinically meaningful attributes

Creating the Methodology Portfolio Themes – High dimensional data – Shrinkage to borrow information – Missing data – Mixed data – Operator effect hierarchically structured data 11/30/2020 • Causal inference approaches with registry data • Methodology to bridge premarket and post-market evidence of device safety and effectiveness. • Methodology for research synthesis to compare medical device effectiveness • Development of a probabilistic risk assessment framework • Automated safety surveillance • UDI Demonstration • Big data

Leveraging Transcathether Valve Therapies ( TVT) and Vascular Quality Initiative (VQI) Registries § Building on two multi-stakeholder national registry efforts (valve and vascular space) § Conversion of traditional studies to novel surveillance § International cardiovascular consortia launched April 2013 (ICCR) and November 2014 (ICVR) Benefits: § Decreases PAS burden and increases real-world capture § Allows for studies to expand indications § Will be basis for real - time active surveillance 11/30/2020

International Consortium of Orthopedic Registries (ICOR) § Global partnership – 29 registries – 8 contributing data – 4 million implants; § Advanced comparative effectiveness /safety research and surveillance (27 papers) § Methods for combining de-identified data sets from registries § ICOR-USA – common data model Benefits: § Contributed to assessment of pre/post balance § Infrastructure used for more efficient, less costly PAS § Expands UDI capabilities with links to clinically meaningful attributes 11/30/2020

PASSION CV Registries Predictable And Su. Stainable Implementation Of National Cardio. Vascular Registries § § § Launched October 2014 Specific proposals in coronary, valve, EP and vascular fields - preparation underway PASSION CV Registries Steering Group will be convened in 2015 Benefits: § Focus on developing embedded IDE studies

National Device Surveillance System: 2014 Implementation Steps • Establish a Multi-stakeholder Medical Device Postmarket Surveillance Planning Board – Identify governance structure, practices/policies/procedures, methods, and business model(s) necessary to facilitate creation of a sustainable, integrated system • Establish a Medical Device Registry Task Force of Key Stakeholders under MDEpi. Net – Identify existing registries that may contribute and leverage on-going registry efforts (e. g. quality assurance, reimbursement) – Identify priority medical devices in need of registries – Define governance and data quality practices to meet multi-stakeholder needs – Develop strategies for the use of registries to support premarket approval and clearance

IMDRF: Integrating Device Registries, UDI and Innovative Tools for Medical Device Evaluation • Evaluate, compare & contrast current approaches to international data models in different device areas: • Generate essential principles document(s) for international collaboration & data sharing related to: – Data access, security, governance, informatics and related issues – Analytic methodologies for safety signal detection, device effectiveness & reliability • Complete proposal in two stages: – Essential principles of data linkage for regulatory convergence (Stage 1) – Essential principles of analytic methodologies for device evaluation (Stage 2)
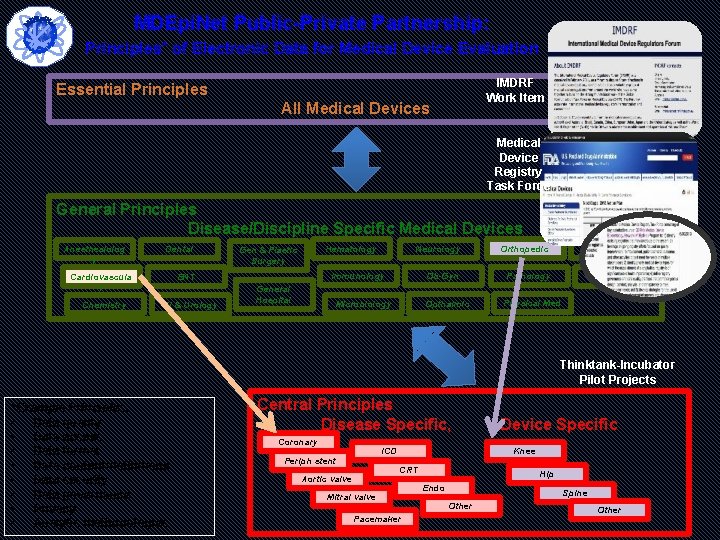
MDEpi. Net Public-Private Partnership: Principles* of Electronic Data for Medical Device Evaluation IMDRF Work Item Essential Principles All Medical Devices Medical Device Registry Task Force General Principles Disease/Discipline Specific Medical Devices Anesthesiolog y Dental Cardiovascula r Chemistry Gen & Plastic Surgery Neurology Orthopedic Ob-Gyn Pathology Immunology ENT GI & Urology Hematology General Hospital Opthalmic Microbiology Radiology Toxicology Physical Med Thinktank-Incubator Pilot Projects *Example Principles: • Data quality • Data access • Data format • Data content/definitions • Data security • Data governance • Privacy • Analytic methodologies Central Principles Disease Specific, Coronary stent Periph stent ICD Knee CRT Aortic valve Device Specific Mitral valve Pacemaker Hip Endo Spine Other
Thank you! 11/30/2020
- Slides: 15