MCB Exam 3 Review Stewart and Miner lectures

MCB Exam 3 Review Stewart and Miner lectures

In the 1930’s while carrying out classic mutagenesis experiments in flies, Hermann Muller (Nobel, 1938) first noted that chromosome ends had “distinct” properties and named them telomeres (telo meaning ”end” and mere meaning “part”).

Telomere Function distinguishes between the chromosome end a double strand break protects the chromosome from end-to-end fusions
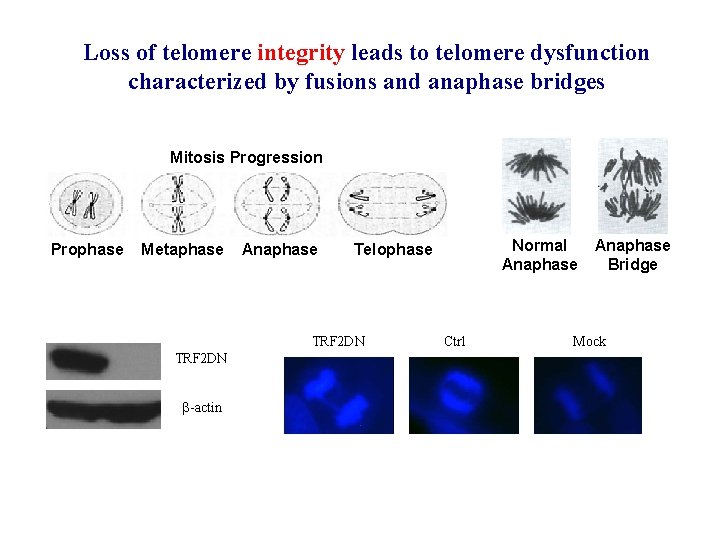
Loss of telomere integrity leads to telomere dysfunction characterized by fusions and anaphase bridges Mitosis Progression Prophase Metaphase Anaphase TRF 2 DN -actin Normal Anaphase Telophase Ctrl Anaphase Bridge Mock
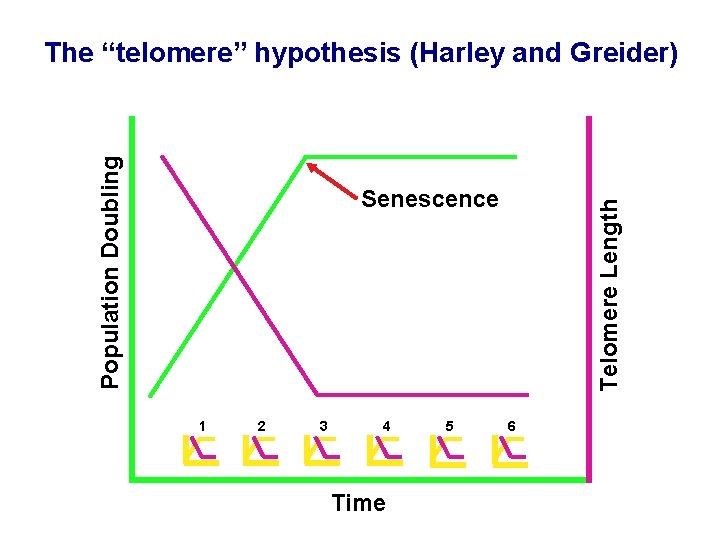
Population Doubling The “telomere” hypothesis (Harley and Greider) 1 2 3 4 Time 5 Telomere Length Senescence 6
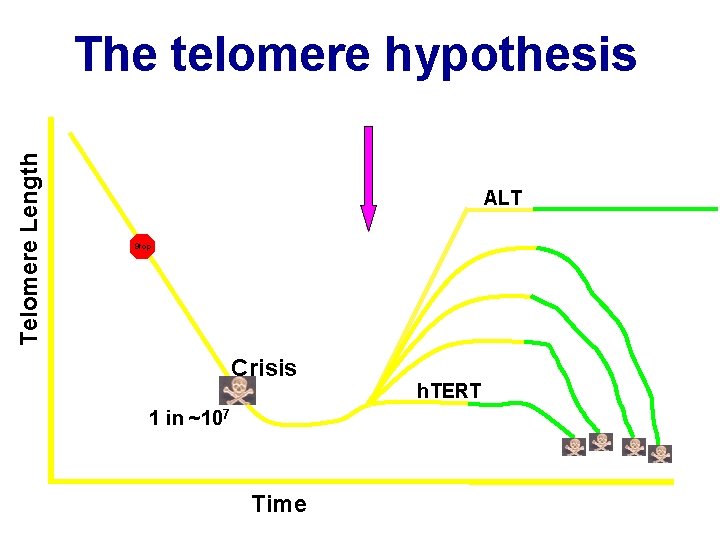
Telomere Length The telomere hypothesis ALT Stop Crisis 1 in ~107 Time h. TERT
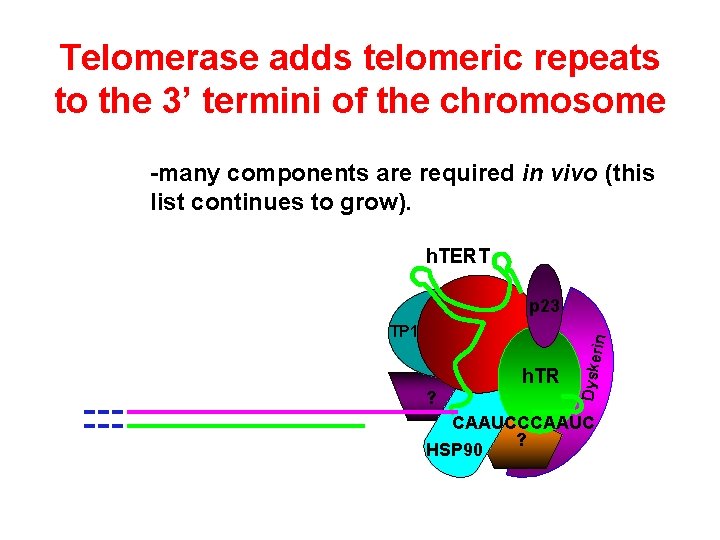
Telomerase adds telomeric repeats to the 3’ termini of the chromosome -many components are required in vivo (this list continues to grow). h. TERT p 23 h. TR ? Dyske ri n TP 1 CAAUCCCAAUC ? HSP 90

Proof that telomerase was important in immortality had to wait until the catalytic component was identified… 1997 -- the catalytic component of telomerase (h. TERT) is cloned -Weinberg Lab at the Whitehead Institute/MIT -Cech Lab at the University of Colorado-Boulder

Several groups demonstrated that telomerase could immortalize “some” cell types -Bodner, 1998 -Rufer, 1998 -Vaziri, 1998 h. TERT “Normal” Immortal Mortal “Normal”
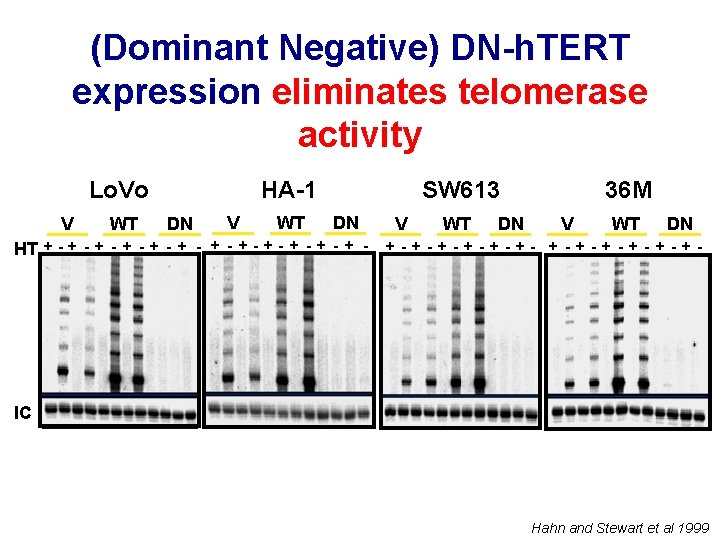
(Dominant Negative) DN-h. TERT expression eliminates telomerase activity Lo. Vo V WT HA-1 DN V WT SW 613 DN HT + - + - + - V WT DN 36 M V WT DN +-+-+-+-+-+- IC Hahn and Stewart et al 1999
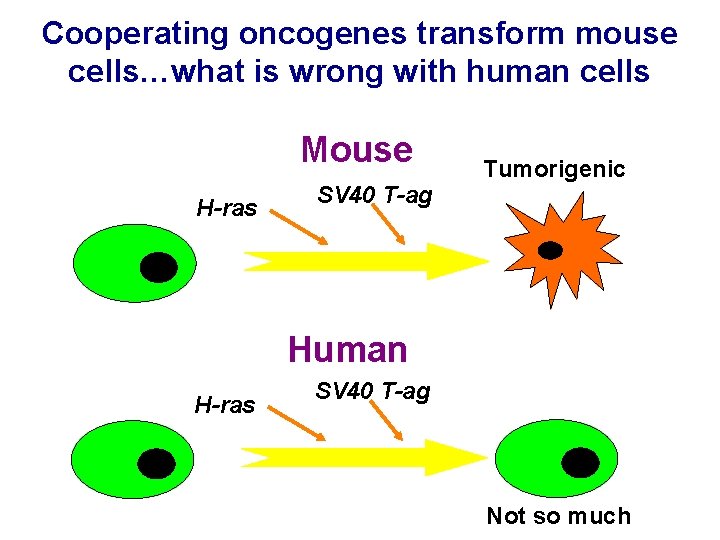
Cooperating oncogenes transform mouse cells…what is wrong with human cells Mouse H-ras SV 40 T-ag Tumorigenic Human H-ras SV 40 T-ag Not so much
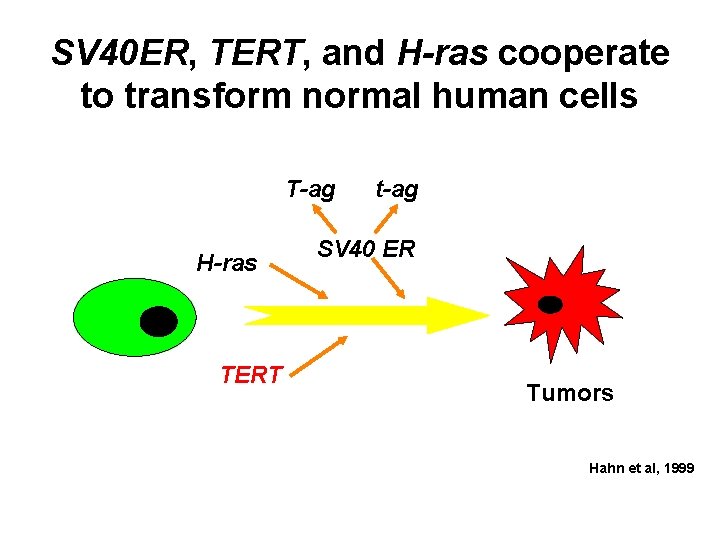
SV 40 ER, TERT, and H-ras cooperate to transform normal human cells T-ag H-ras TERT t-ag SV 40 ER Tumors Hahn et al, 1999
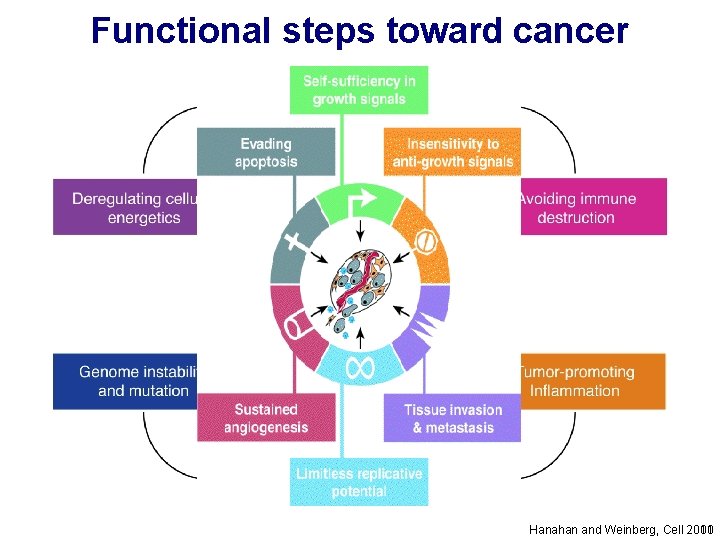
Functional steps toward cancer Hanahan and Weinberg, Cell 2000 2011

Telomerase activity in human carcinomas Tissue Head/neck and lung GI and pancreas Hepatic tissue Incidence 78 -100% 85 -100% 86% Breast 75 -88% Cervical/endometrial/vaginal/ovarian Prostate 91 -100% 90% Kidney/urinary 83 -100% Neural* 50 -100% Skin 83 -95% Hematological tissue 73 -100% *retinoblastoma, meningioma, neuroblastoma, Shay 1997
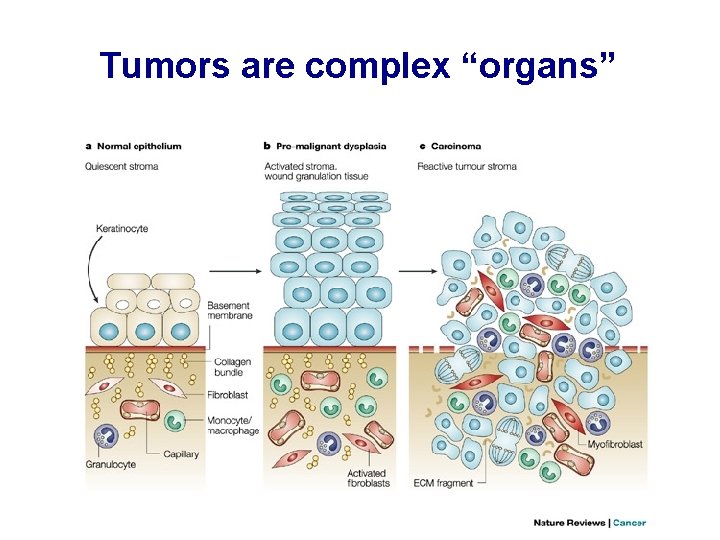
Tumors are complex “organs”
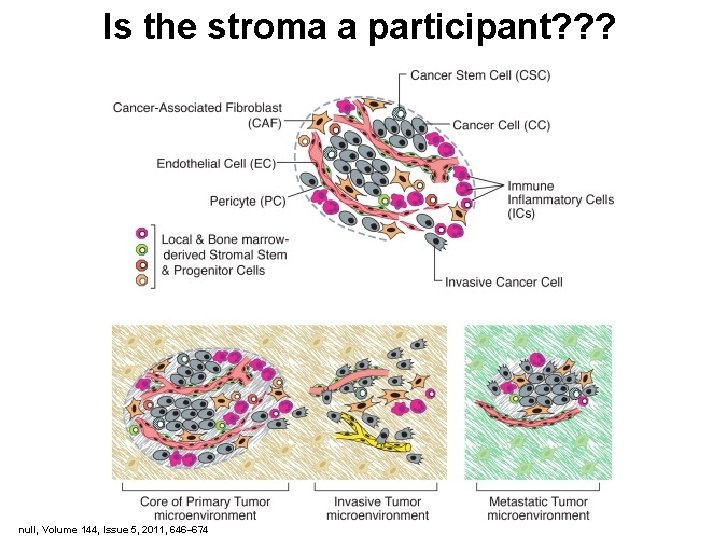
Is the stroma a participant? ? ? null, Volume 144, Issue 5, 2011, 646– 674
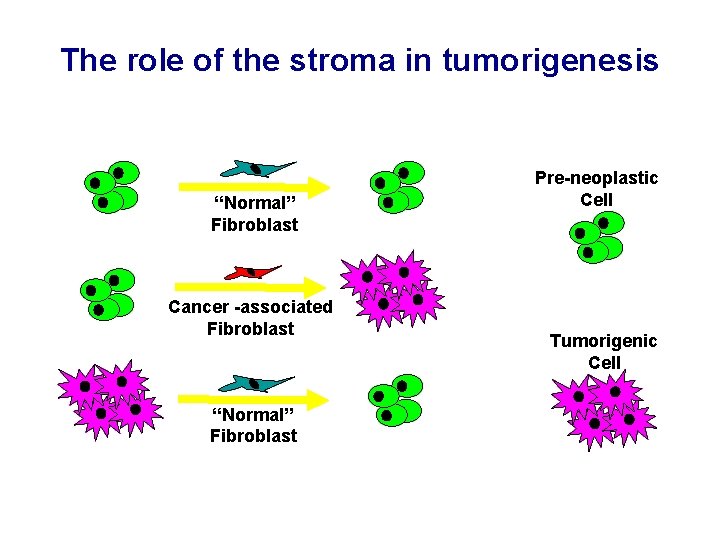
The role of the stroma in tumorigenesis “Normal” Fibroblast Cancer -associated Fibroblast “Normal” Fibroblast Pre-neoplastic Cell Tumorigenic Cell
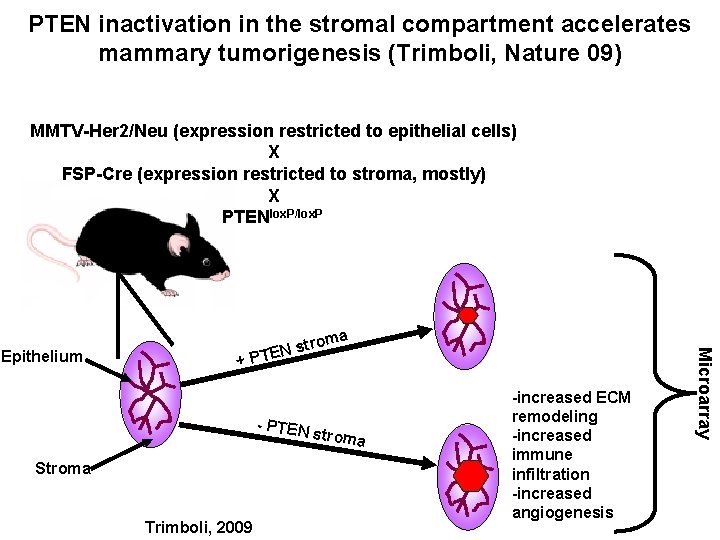
PTEN inactivation in the stromal compartment accelerates mammary tumorigenesis (Trimboli, Nature 09) MMTV-Her 2/Neu (expression restricted to epithelial cells) X FSP-Cre (expression restricted to stroma, mostly) X PTENlox. P/lox. P a + PT - PTEN Stroma Trimboli, 2009 stroma -increased ECM remodeling -increased immune infiltration -increased angiogenesis Microarray Epithelium trom EN s

Early studies demonstrated that tumors arose in areas prone to wounding…. . i. e. - Deelman found tumors arose at the margin of accidental wounds in the skin of mice treated with tar. -Bissell’s group found that RSV infected chickens developed tumors at the site of wounds. -BPV induces tumors at sites of wounds. -Tat and v-jun transgenic mice develop tumors at sites of wounds. -These are but a few examples… For review: Sieweke & Bissell
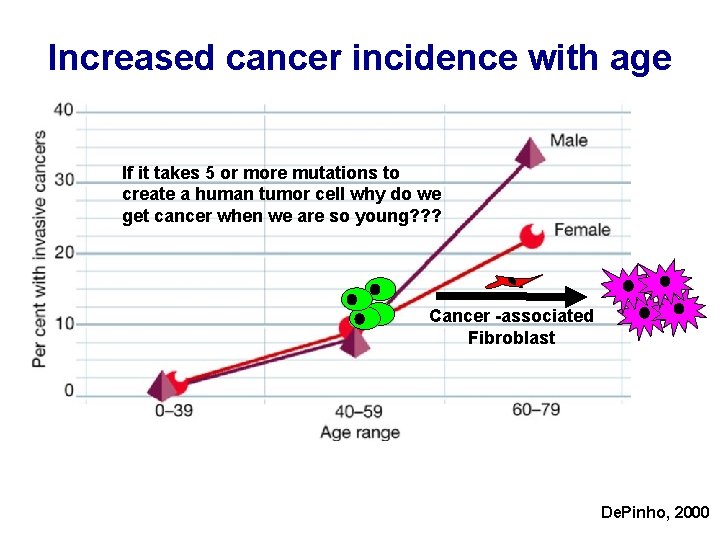
Increased cancer incidence with age If it takes 5 or more mutations to create a human tumor cell why do we get cancer when we are so young? ? ? Cancer -associated Fibroblast De. Pinho, 2000
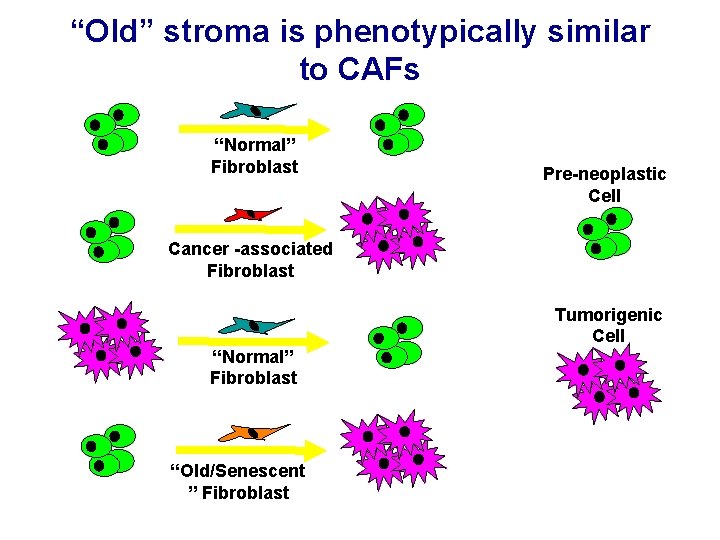
“Old” stroma is phenotypically similar to CAFs “Normal” Fibroblast Pre-neoplastic Cell Cancer -associated Fibroblast Tumorigenic Cell “Normal” Fibroblast “Old/Senescent ” Fibroblast

Other characteristics of senescent cells • • • Absence of proliferation markers Flattened cell morphology Increased cell volume Senescence-associated β-galactosidase p 16 INK 4 a tumor suppressor expression Senescence-associated heterochromatin foci • Altered secretory profile (SASP)
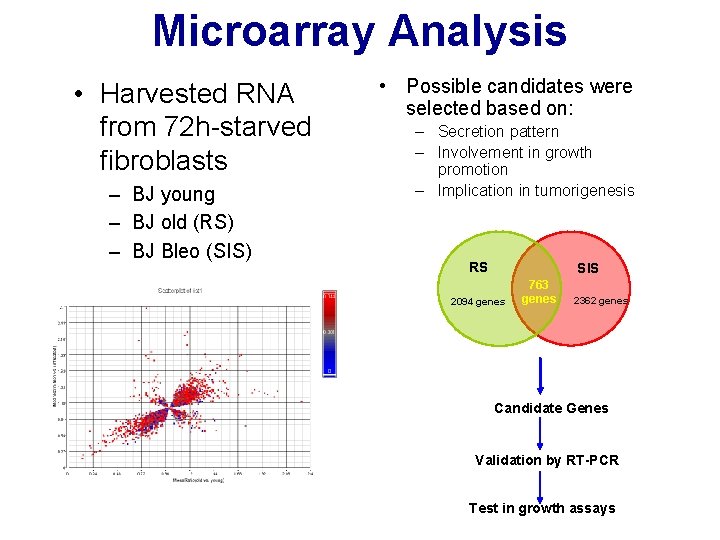
Microarray Analysis • Harvested RNA from 72 h-starved fibroblasts – BJ young – BJ old (RS) – BJ Bleo (SIS) • Possible candidates were selected based on: – Secretion pattern – Involvement in growth promotion – Implication in tumorigenesis RS SIS 2094 genes 763 genes 2362 genes Candidate Genes Validation by RT-PCR Test in growth assays
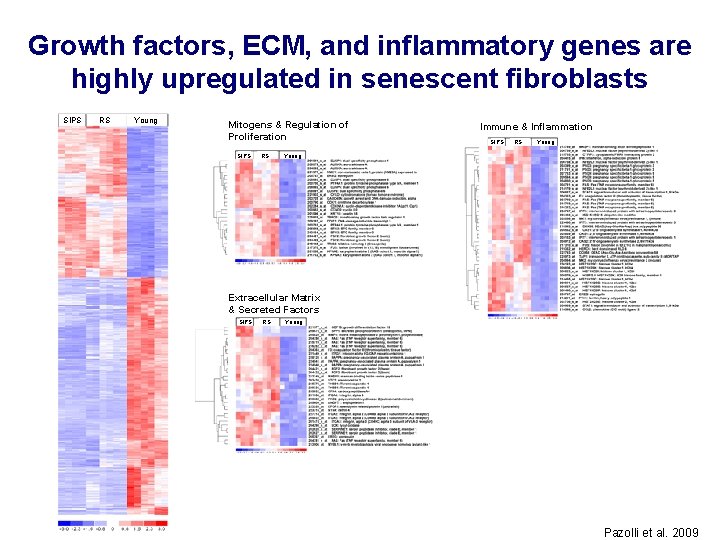
Growth factors, ECM, and inflammatory genes are highly upregulated in senescent fibroblasts SIPS RS Young Mitogens & Regulation of Proliferation SIPS RS Immune & Inflammation SIPS RS Young Extracellular Matrix & Secreted Factors SIPS RS Young Pazolli et al. 2009
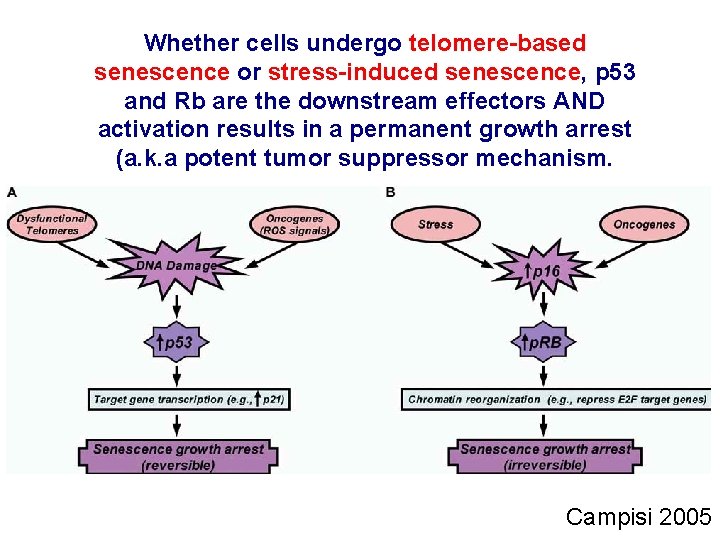
Whether cells undergo telomere-based senescence or stress-induced senescence, p 53 and Rb are the downstream effectors AND activation results in a permanent growth arrest (a. k. a potent tumor suppressor mechanism. Campisi 2005
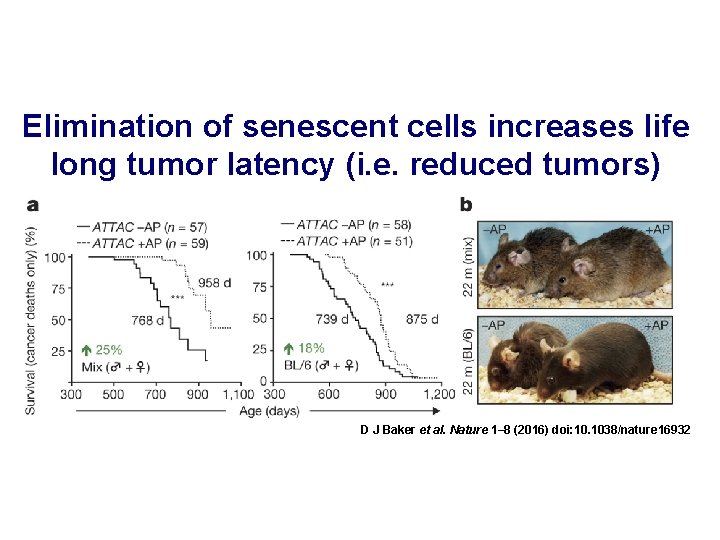
Elimination of senescent cells increases life long tumor latency (i. e. reduced tumors) D J Baker et al. Nature 1– 8 (2016) doi: 10. 1038/nature 16932
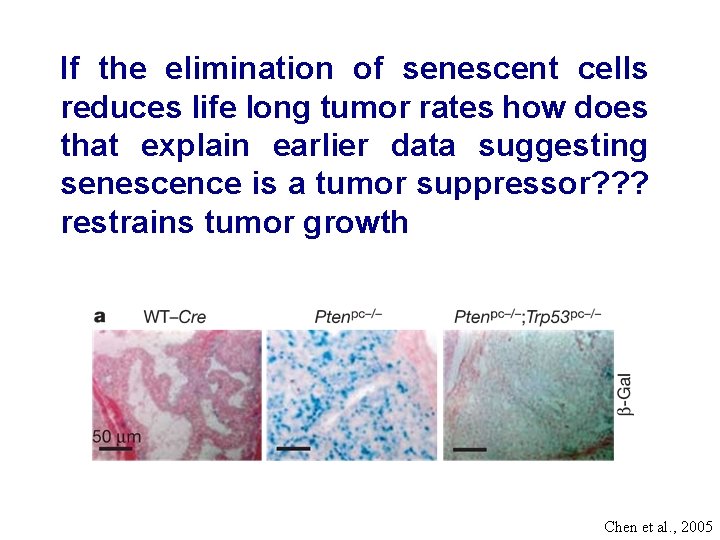
If the elimination of senescent cells reduces life long tumor rates how does that explain earlier data suggesting senescence is a tumor suppressor? ? ? restrains tumor growth Chen et al. , 2005
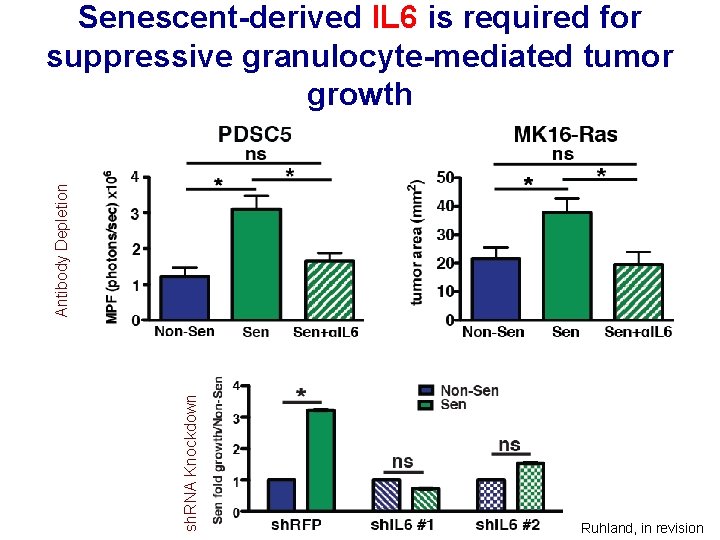
sh. RNA Knockdown Antibody Depletion Senescent-derived IL 6 is required for suppressive granulocyte-mediated tumor growth Ruhland, in revision
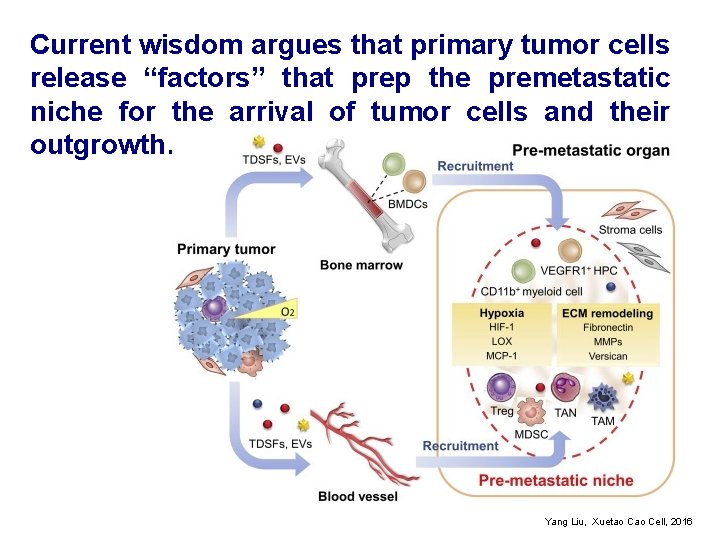
Current wisdom argues that primary tumor cells release “factors” that prep the premetastatic niche for the arrival of tumor cells and their outgrowth. Yang Liu, Xuetao Cell, 2016

The Extracellular Matrix November 29, 2016 Jeff Miner, Ph. D. Renal Division 7717 Wohl Clinic 362 -8235 minerj@wustl. edu

Why do all multicellular animals have ECM? • Acts as structural support to maintain cell organization and integrity (endothelial tubes of the cardiovascular system; mucosal lining of gut; skeletal muscle fiber integrity) • Compartmentalizes tissues (pancreas: islets vs. exocrine component; skin: epidermis vs. dermis) • Provides hardness to bone and teeth (collagen fibrils become mineralized/calcified) • Presents information to adjacent cells: – Inherent signals (e. g. , RGD motif in fibronectin) – Bound signals (BMP 7, TGFβ, FGF, SHH, etc. ) • Serves as a highway for cell migration during development (neural crest migration), in normal tissue maintenance (intestinal mucosa), and in injury or disease (wound healing and cancer)

Types of ECMs • Basement membrane (basal lamina) – Epithelia, endothelia, muscle, fat, nerves • Elastic fibers – Skin, lung, large blood vessels • Stromal or interstitial matrix • Bone, tooth, and cartilage • Tendon and ligament

Cells Need Receptors to Recognize and Respond to ECM • • • Integrins Dystroglycan Syndecans Muscle-Specific kinase (Mu. SK) Discoidin domain receptors (DDRs) 1 and 2 Others

Types of ECM Components • Collagens • Proteoglycans – Perlecan, aggrecan, agrin, collagen XVIII • Hyaluronan (no protein core) • Large Modular Glycoproteins – Laminins, nidogens, fibronectin, vitronectin • Fibrillins, elastin, LTBPs, MAGPs, fibulins • “Matricellular” Proteins – SPARC, Thrombospondins, Osteopontin, tenascins

Basement Membranes • Specialized layers of extracellular matrix surrounding or adjacent to all epithelia, endothelia, peripheral nerves, muscle cells, and fat cells • In general, basement membranes appear very similar to each other by EM. • But all are not alike! • There is a wealth of molecular and functional heterogeneity due primarily to isoform variations of BM components.
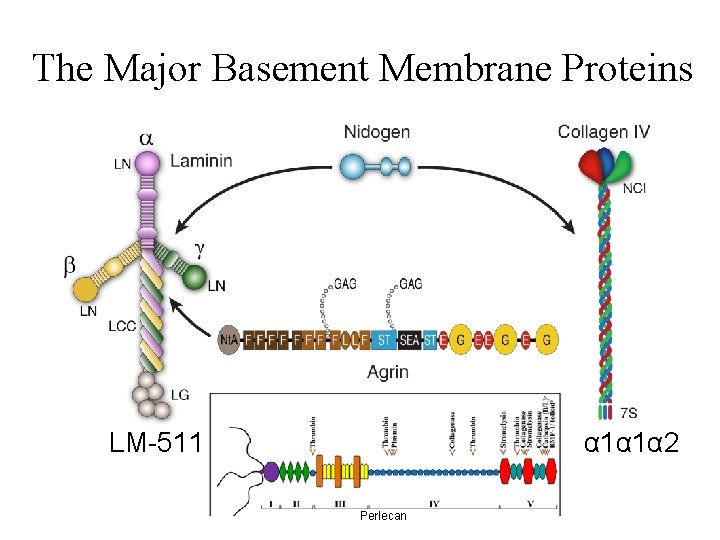
The Major Basement Membrane Proteins LM-511 α 1α 1α 2 Perlecan
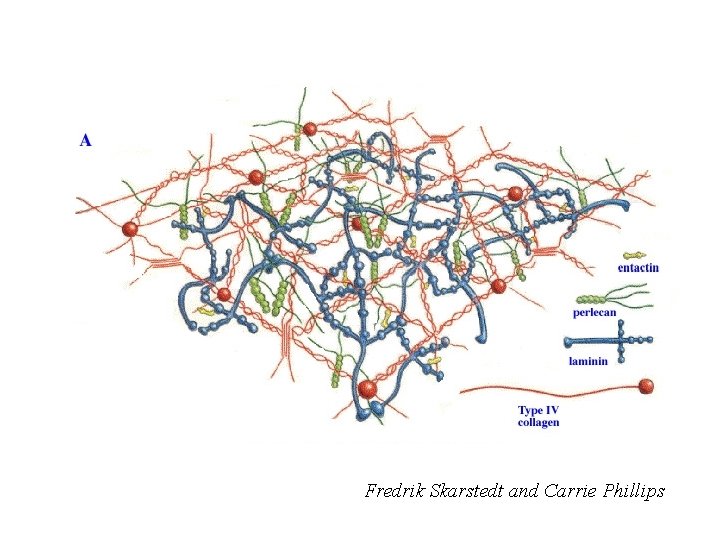
Fredrik Skarstedt and Carrie Phillips

Basement Membranes are Involved in a Multitude of Biological Processes • Cell proliferation, differentiation, and migration • Cell polarization and organization, as well as maintenance of tissue structure • Separation of epithelia from the underlying stroma/mesenchyme/interstitium, which contains a non-basement membrane matrix • Kidney glomerular filtration (barrier between the bloodstream and the urinary space)

Primary Components of All Basement Membranes • • Collagen IV 6 chains form α chain heterotrimers Laminin 12 chains form several α-β-γ heterotrimers Entactin/Nidogen 2 isoforms Sulfated proteoglycans Perlecan and Agrin are the major ones; Collagen XVIII is another History: The Engelbreth-Holm-Swarm (EHS) tumor: A blessing with a caveat.
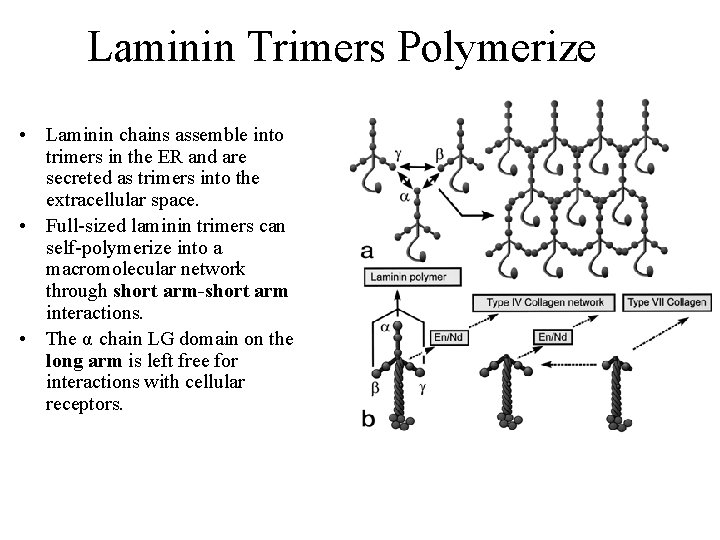
Laminin Trimers Polymerize • Laminin chains assemble into trimers in the ER and are secreted as trimers into the extracellular space. • Full-sized laminin trimers can self-polymerize into a macromolecular network through short arm-short arm interactions. • The α chain LG domain on the long arm is left free for interactions with cellular receptors.
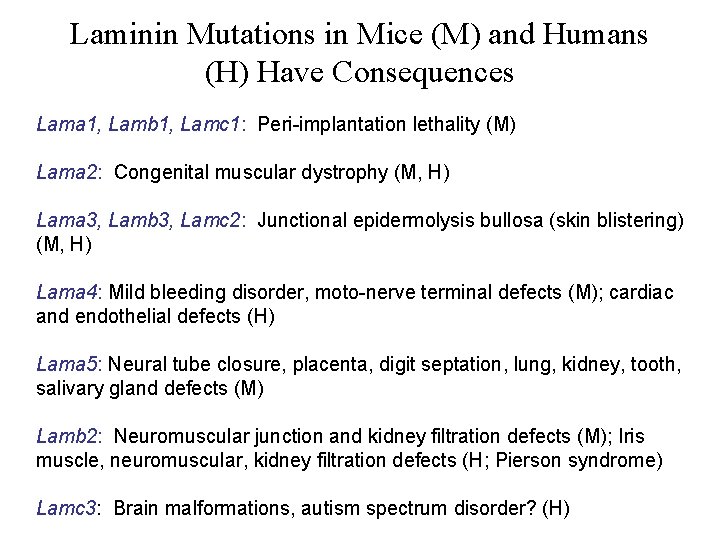
Laminin Mutations in Mice (M) and Humans (H) Have Consequences Lama 1, Lamb 1, Lamc 1: Peri-implantation lethality (M) Lama 2: Congenital muscular dystrophy (M, H) Lama 3, Lamb 3, Lamc 2: Junctional epidermolysis bullosa (skin blistering) (M, H) Lama 4: Mild bleeding disorder, moto-nerve terminal defects (M); cardiac and endothelial defects (H) Lama 5: Neural tube closure, placenta, digit septation, lung, kidney, tooth, salivary gland defects (M) Lamb 2: Neuromuscular junction and kidney filtration defects (M); Iris muscle, neuromuscular, kidney filtration defects (H; Pierson syndrome) Lamc 3: Brain malformations, autism spectrum disorder? (H)

Perlecan • Found widely in basement membranes and in cartilage. • Contains domains similar to LDL receptor, laminin, and NCAM • Binds to Collagen IV and to Entactin/Nidogen
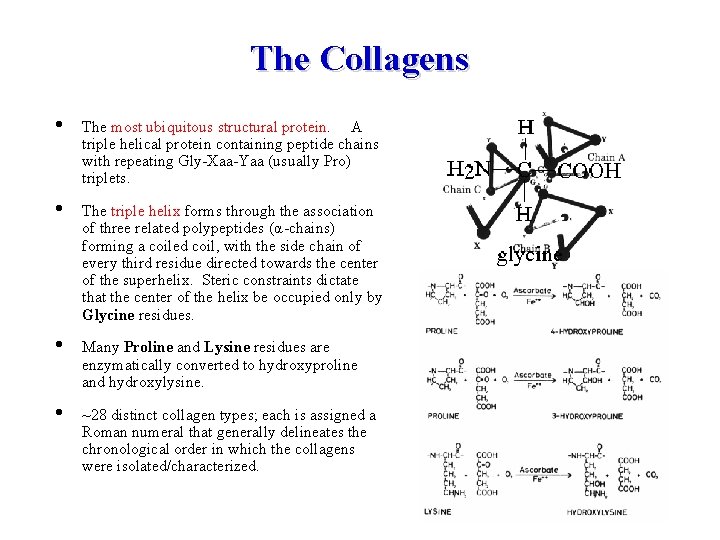
The Collagens • The most ubiquitous structural protein. A triple helical protein containing peptide chains with repeating Gly-Xaa-Yaa (usually Pro) triplets. • The triple helix forms through the association of three related polypeptides (α-chains) forming a coiled coil, with the side chain of every third residue directed towards the center of the superhelix. Steric constraints dictate that the center of the helix be occupied only by Glycine residues. • Many Proline and Lysine residues are enzymatically converted to hydroxyproline and hydroxylysine. • ~28 distinct collagen types; each is assigned a Roman numeral that generally delineates the chronological order in which the collagens were isolated/characterized.
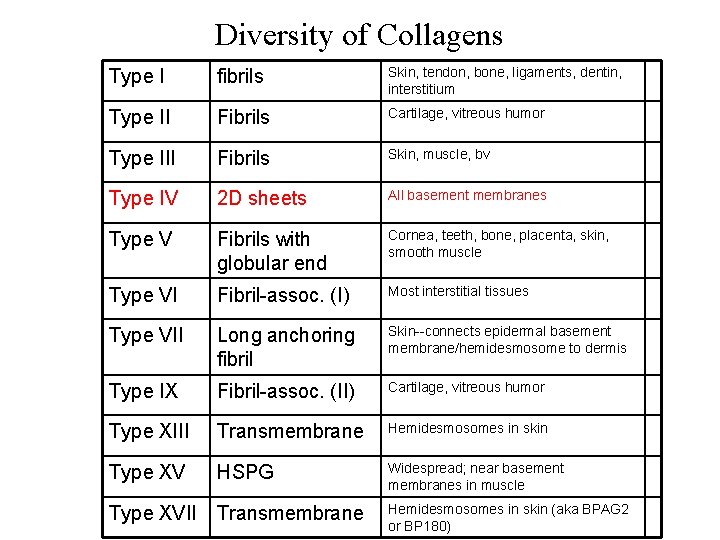
Diversity of Collagens Type I fibrils Skin, tendon, bone, ligaments, dentin, interstitium Type II Fibrils Cartilage, vitreous humor Type III Fibrils Skin, muscle, bv Type IV 2 D sheets All basement membranes Type V Fibrils with globular end Cornea, teeth, bone, placenta, skin, smooth muscle Type VI Fibril-assoc. (I) Most interstitial tissues Type VII Long anchoring fibril Skin--connects epidermal basement membrane/hemidesmosome to dermis Type IX Fibril-assoc. (II) Cartilage, vitreous humor Type XIII Transmembrane Hemidesmosomes in skin Type XV HSPG Widespread; near basement membranes in muscle Type XVII Transmembrane Hemidesmosomes in skin (aka BPAG 2 or BP 180)

Collagen IV Network Trimers (aka protomers) associate with each other, four at the N-terminus and two at the C-terminus (hexamer), to form a chicken wire-like network that provides strength and flexibility to the basement membrane.
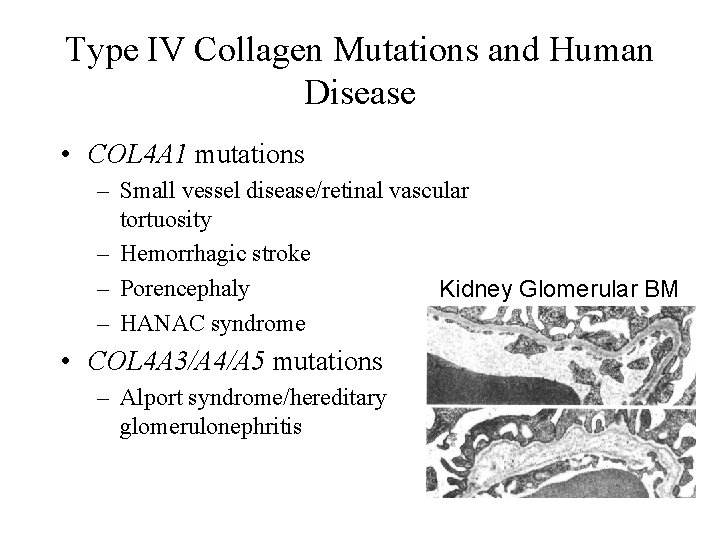
Type IV Collagen Mutations and Human Disease • COL 4 A 1 mutations – Small vessel disease/retinal vascular tortuosity – Hemorrhagic stroke – Porencephaly Kidney Glomerular BM – HANAC syndrome • COL 4 A 3/A 4/A 5 mutations – Alport syndrome/hereditary glomerulonephritis
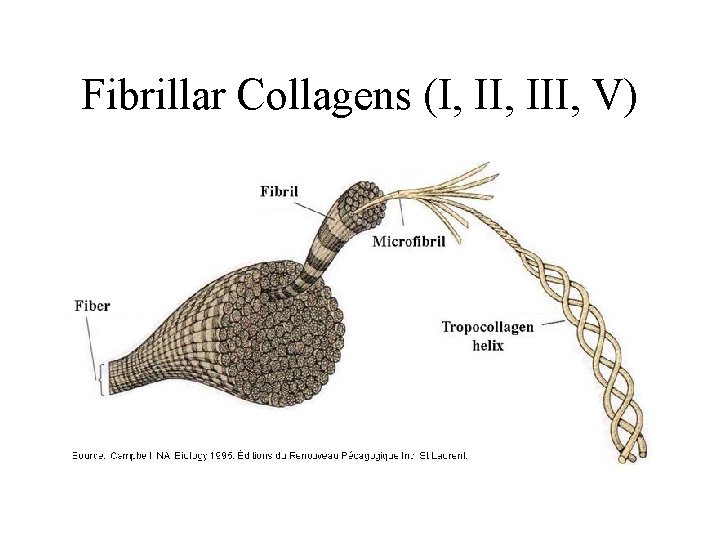
Fibrillar Collagens (I, III, V)
Fibrillar Collagens (I, III, V) • Connective tissue proteins that provide tensile strength • Triple helix, composed of three α chains • Glycine at every third position (Gly-X-Y) • High proline content – Hydroxylation required for proper folding and secretion • Found in bone, skin, tendons, cartilage, arteries
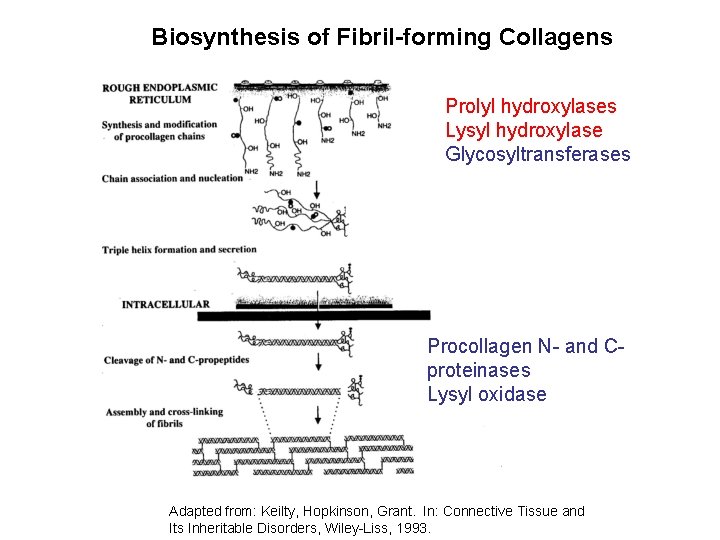
Biosynthesis of Fibril-forming Collagens Prolyl hydroxylases Lysyl hydroxylase Glycosyltransferases Procollagen N- and Cproteinases Lysyl oxidase Adapted from: Keilty, Hopkinson, Grant. In: Connective Tissue and Its Inheritable Disorders, Wiley-Liss, 1993.

Collagen Crosslinking • If crosslinking is inhibited (Lysyl hydroxylase mutations; vitamin C deficiency), collagenous tissues become fragile, and structures such as skin, tendons, and blood vessels tend to tear. There also many bone manifestations of under-crosslinked collagen. • Hydroxylation of specific lysines governs the nature of the cross-link formed, which affects the biomechanical properties of the tissue. Collagen is especially highly crosslinked in the Achilles tendon, where tensile strength is crucial.

Scurvy • Liver spots on skin, spongy gums, bleeding from mucous membranes, immobility, depression • Caused by Vitamin C deficiency • Ascorbate is required for prolyl hydroxylase and lysyl hydroxylase activities • Acquired disease of fibrillar collagen Illustration from Man-of-War by Stephen Biesty (Dorling-Kindersley, NY, 1993)

Osteogenesis Imperfecta (brittle bone disease) Clinical: Ranges in severity from mild to perinatal lethal bone fragility, short stature, bone deformities, teeth abnormalities, gray -blue sclerae, hearing loss Biochemical: Reduced and/or abnormal Type I collagen Molecular Genetics: Mutations in either Type I collagen gene, COL 1 A 1 or COL 1 A 2, resulting in haploinsufficiency or disruption of the triple helical domain (dominant negative: glycine substitutions most common)

Cell-Matrix Interactions December 1, 2016 Jeff Miner, Ph. D. Division of Nephrology 7717 Wohl Clinic 362 -8235 minerj@wustl. edu Twitter: @Jeff. Miner. Ph. D
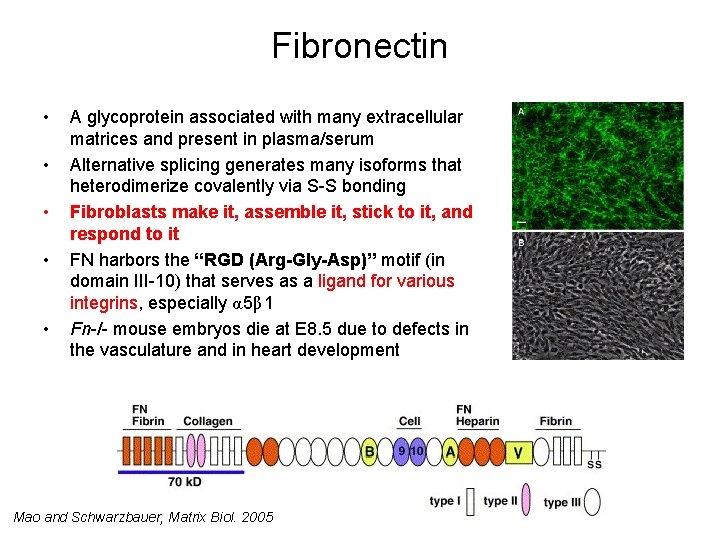
Fibronectin • • • A glycoprotein associated with many extracellular matrices and present in plasma/serum Alternative splicing generates many isoforms that heterodimerize covalently via S-S bonding Fibroblasts make it, assemble it, stick to it, and respond to it FN harbors the “RGD (Arg-Gly-Asp)” motif (in domain III-10) that serves as a ligand for various integrins, especially α 5β 1 Fn-/- mouse embryos die at E 8. 5 due to defects in the vasculature and in heart development Mao and Schwarzbauer, Matrix Biol. 2005
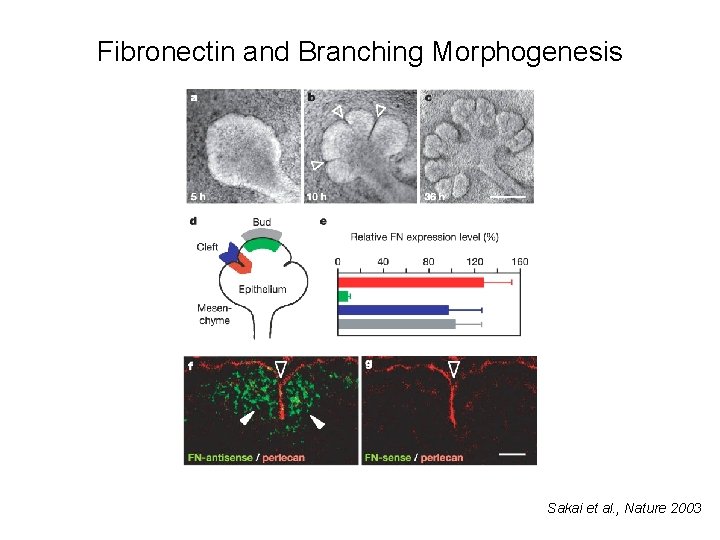
Fibronectin and Branching Morphogenesis Sakai et al. , Nature 2003
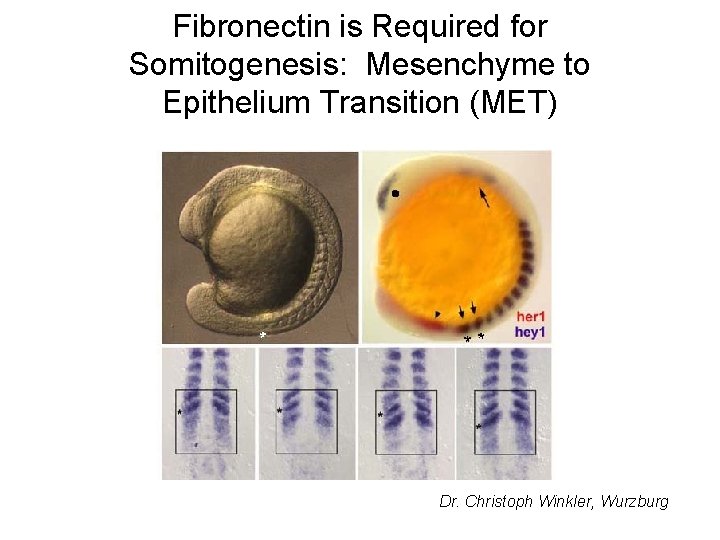
Fibronectin is Required for Somitogenesis: Mesenchyme to Epithelium Transition (MET) Dr. Christoph Winkler, Wurzburg
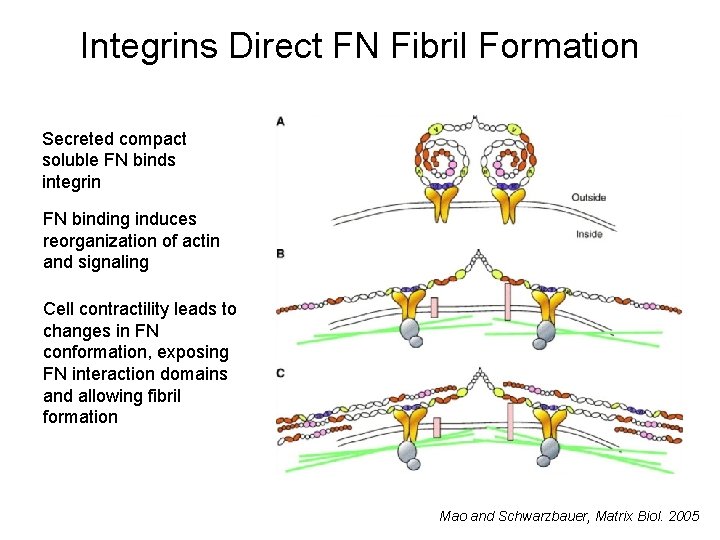
Integrins Direct FN Fibril Formation Secreted compact soluble FN binds integrin FN binding induces reorganization of actin and signaling Cell contractility leads to changes in FN conformation, exposing FN interaction domains and allowing fibril formation Mao and Schwarzbauer, Matrix Biol. 2005
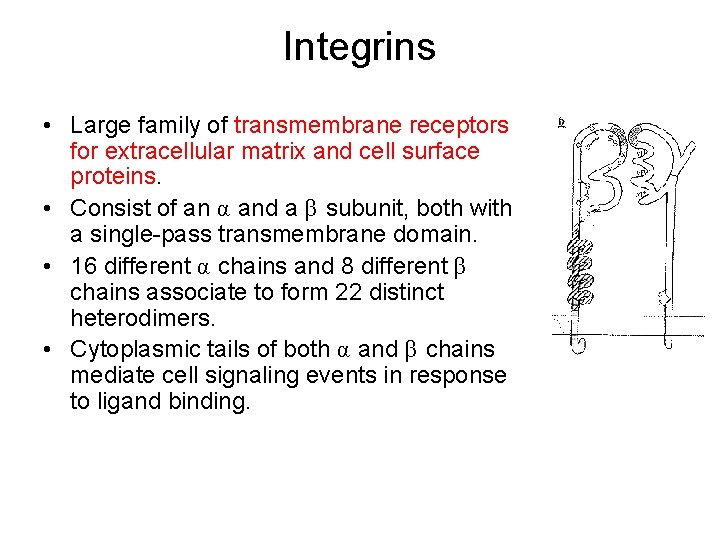
Integrins • Large family of transmembrane receptors for extracellular matrix and cell surface proteins. • Consist of an α and a β subunit, both with a single-pass transmembrane domain. • 16 different α chains and 8 different β chains associate to form 22 distinct heterodimers. • Cytoplasmic tails of both α and β chains mediate cell signaling events in response to ligand binding.

Integrins • Some integrins bind to a specific site on matrix proteins, such as Arg-Gly-Asp (RGD), which is found in fibronectin, vitronectin, tenascin, et al. • Ligand binding absolutely requires divalent cation** • As mechanotransducers, integrins link the extracellular matrix to the force generating actin-myosin cytoskeleton. This both allows the cell to influence the nature of the extracellular matrix, and allows the ECM to influence cellular architecture and behavior.
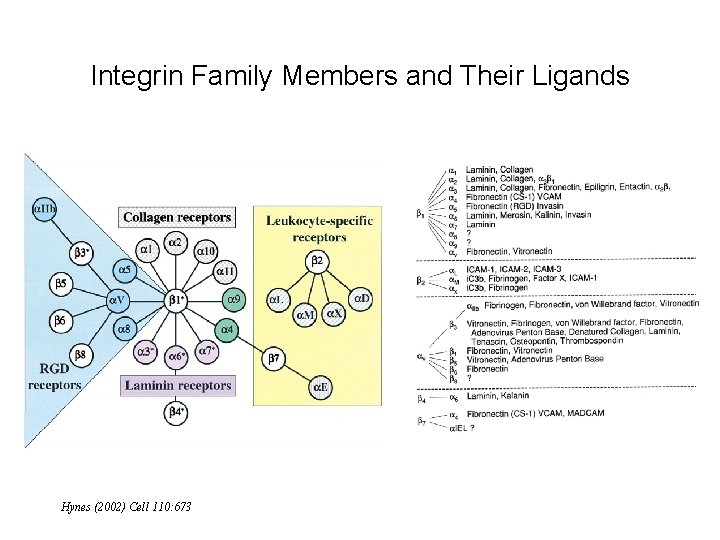
Integrin Family Members and Their Ligands Hynes (2002) Cell 110: 673

Integrins Need to be Activated • Integrin adhesiveness can be dynamically regulated through a process termed inside-out signaling. • Ligand binding transduces signals from the cellular environment to the interior of the cell through outside-in signaling. • Protein structure analyses have provided insights into the mechanisms whereby integrins become activated to bind ligand how ligand binding translates to changes in intracellular signaling. Adair and Yeager, Meth. Enzymol. 2007
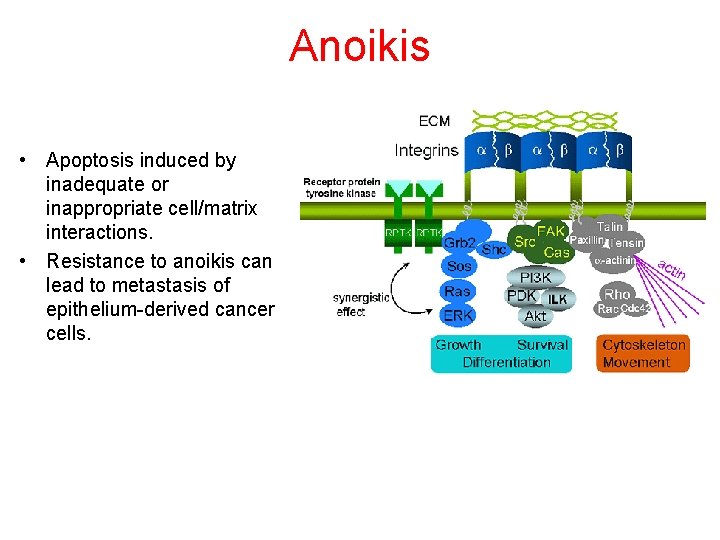
Anoikis • Apoptosis induced by inadequate or inappropriate cell/matrix interactions. • Resistance to anoikis can lead to metastasis of epithelium-derived cancer cells.
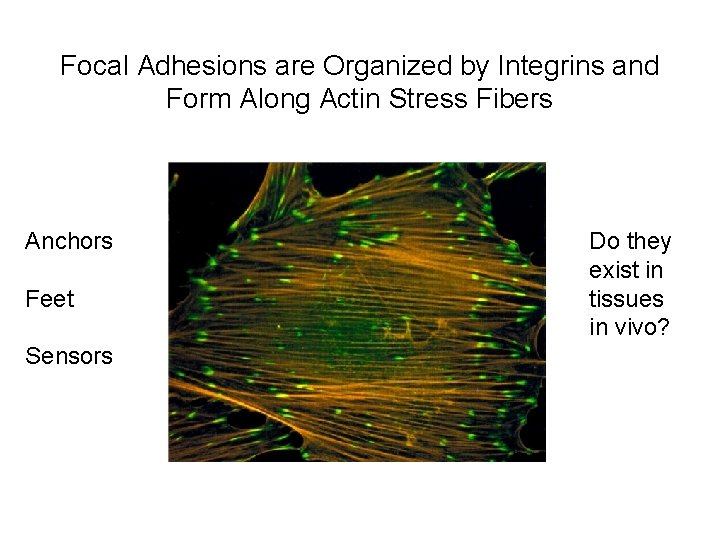
Focal Adhesions are Organized by Integrins and Form Along Actin Stress Fibers Anchors Feet Sensors Do they exist in tissues in vivo?

Receptors for the Basement Membrane • Cells are thought to recognize the basement membrane through receptors that interact with specific basement membrane components, primarily with laminin. – Integrins – Dystroglycan • Binding of receptors to the basement membrane can result in signal transduction and alterations in cell behavior.
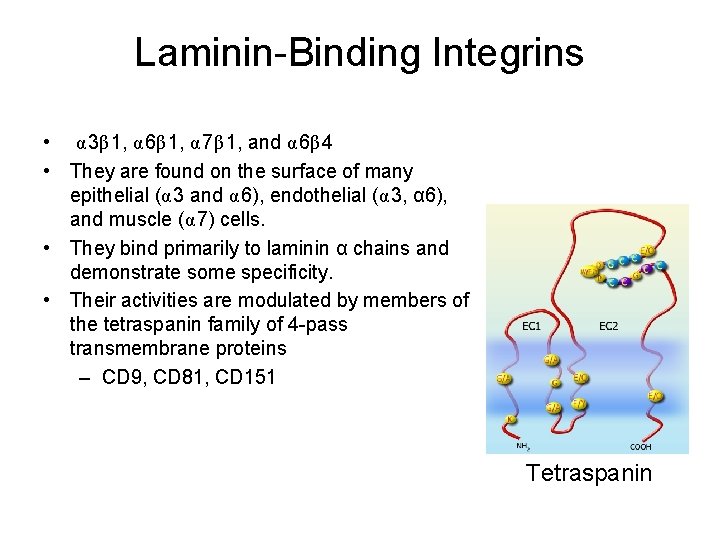
Laminin-Binding Integrins • α 3β 1, α 6β 1, α 7β 1, and α 6β 4 • They are found on the surface of many epithelial (α 3 and α 6), endothelial (α 3, α 6), and muscle (α 7) cells. • They bind primarily to laminin α chains and demonstrate some specificity. • Their activities are modulated by members of the tetraspanin family of 4 -pass transmembrane proteins – CD 9, CD 81, CD 151 Tetraspanin
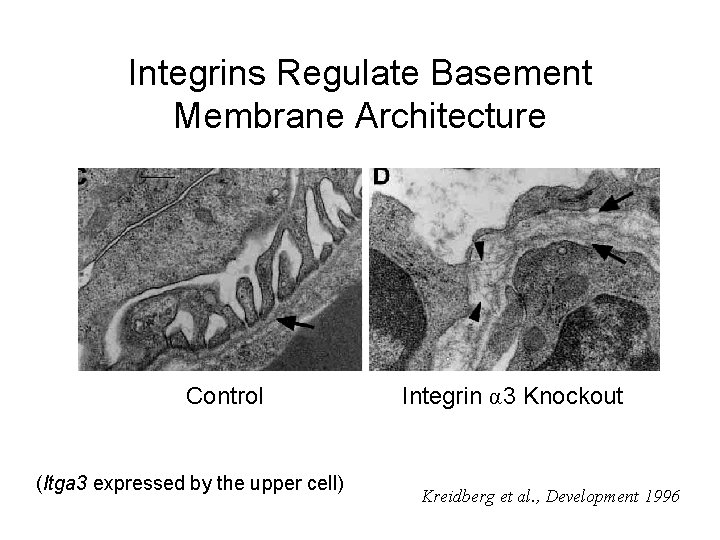
Integrins Regulate Basement Membrane Architecture Control (Itga 3 expressed by the upper cell) Integrin α 3 Knockout Kreidberg et al. , Development 1996
The Dermal-Epidermal Junction
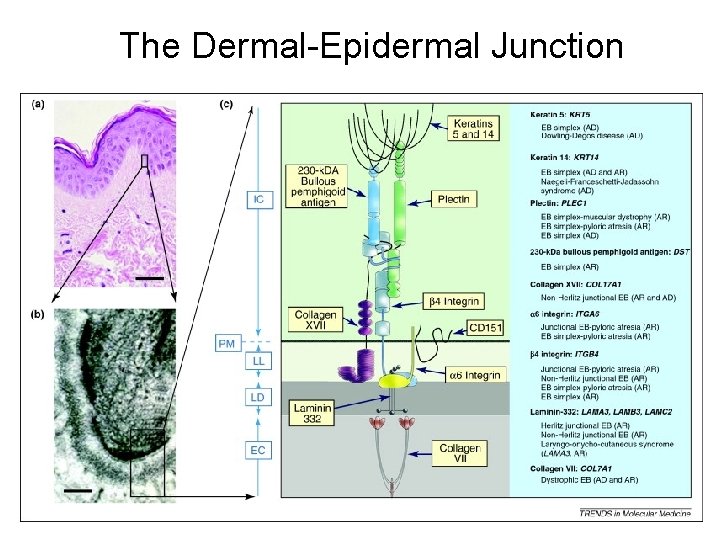
The Dermal-Epidermal Junction

Collagen-Binding Integrins • Integrin α 1β 1 and α 2β 1 bind to type IV, as well as to other collagens. – Itga 1 and Itga 2 knockouts have mild phenotypes, but they show that α 1β 1 downregulates collagen IV synthesis, whereas α 2β 1 upregulates it, at least in the setting of injury and fibrosis.
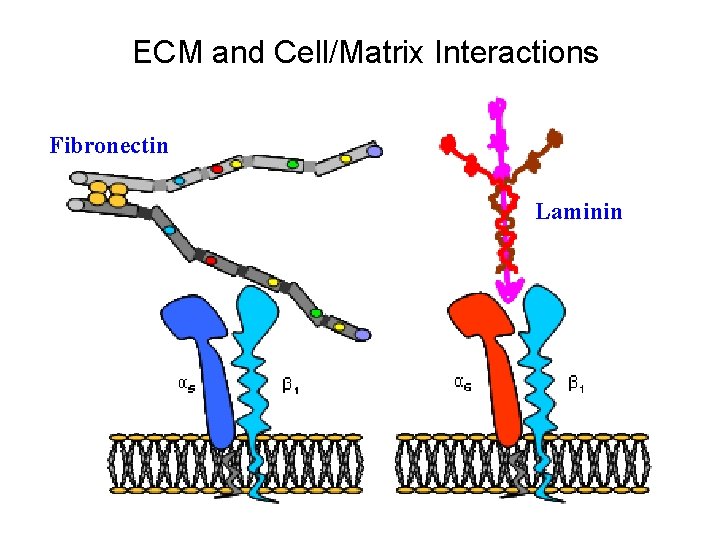
ECM and Cell/Matrix Interactions Fibronectin Laminin
- Slides: 70