MCB Electrophysiology Fun MiniReview Measuring Concentration Solute concentration

MCB Electrophysiology Fun Mini-Review

Measuring Concentration • Solute concentration can be expressed in different ways: – Molarity = moles solute/liter solution – Molality = moles solute/kg solvent – Equivalency (meq/L = m. N) = moles x valence (5 mmoles Na = 5 meq ; 5 mmoles Ca 2+ = 10 meq) • 1 mole of anything = 6 x 1023 units (atoms, molecules, ions, electrons, dissolved particles, etc. ) • The total concentration of dissolved particles is expressed: – Osmolarity = total moles of dissolved particles / liter solution – Osmolality = total moles of dissolved particles / kg solvent • These solutions would have the same osmolarity: 300 m. M sucrose = 150 m. M Na. Cl = 100 m. M Na 2 SO 4

p. H: A Special Case • The concentration of hydrogen ions and hydroxide ions determine the p. H of a solution • • • p. H = -log[H+], where [H+] is measured in molarity [H+] = 10 -p. H > 7 is considered basic p. H = 7 is considered neutral p. H < 7 is considered acidic Normal plasma p. H = 7. 4 (40 x 10 -9 M; 40 n. M)
![Tonicity vs Osmolarity • Osmolarity = [dissolved particles] (physical chemistry) • Tonicity is the Tonicity vs Osmolarity • Osmolarity = [dissolved particles] (physical chemistry) • Tonicity is the](http://slidetodoc.com/presentation_image_h2/6243f5dae730f7cb709355aa1737f25a/image-4.jpg)
Tonicity vs Osmolarity • Osmolarity = [dissolved particles] (physical chemistry) • Tonicity is the effective osmotic pressure relative to blood plasma (cell physiology – depends on permeability) 150 m. M Na. Cl – isotonic, isosmotic 10 m. M Et. OH – hypo-osmotic and hypotonic Both together – isotonic, hyperosmotic

Approximate Compositions of Extracellular and Intracellular Compartments Extracellular Concentration (m. M) Intracellular Concentration (m. M) Cations Na+ K+ Mg 2+ Ca 2+ H+ 140 4 1 -2 1. 25 4 x 10 -5 (p. H 7. 4; 40 n. M) 14 120 0. 5 5 x 10 -5 8 x 10 -5 (p. H 7. 1; 80 n. M) Anions Cl. HCO 3 - 105 24 10 10 Because the cell is electrically neutral the large deficit in intracellular anions reflects the fact that most cellular constituents are negatively charged. The concentrations for Mg and Ca are given for free ions.
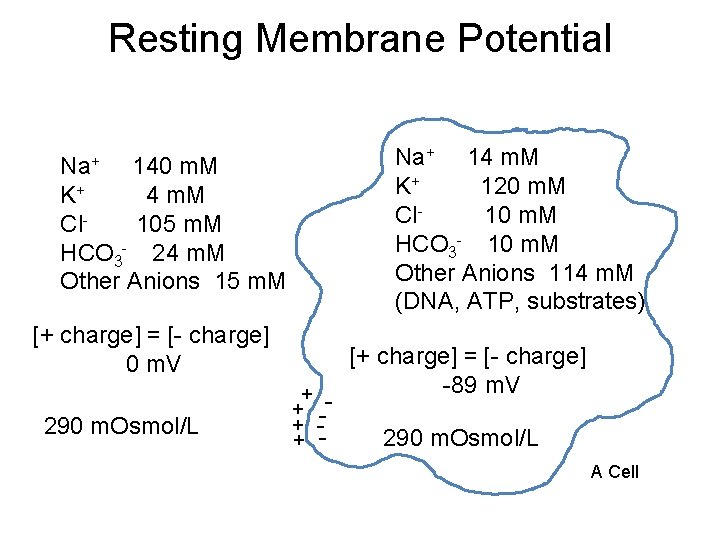
Resting Membrane Potential Na+ 14 m. M K+ 120 m. M Cl 10 m. M HCO 3 - 10 m. M Other Anions 114 m. M (DNA, ATP, substrates) Na+ 140 m. M K+ 4 m. M Cl 105 m. M HCO 3 - 24 m. M Other Anions 15 m. M [+ charge] = [- charge] 0 m. V 290 m. Osmol/L + + - [+ charge] = [- charge] -89 m. V 290 m. Osmol/L A Cell
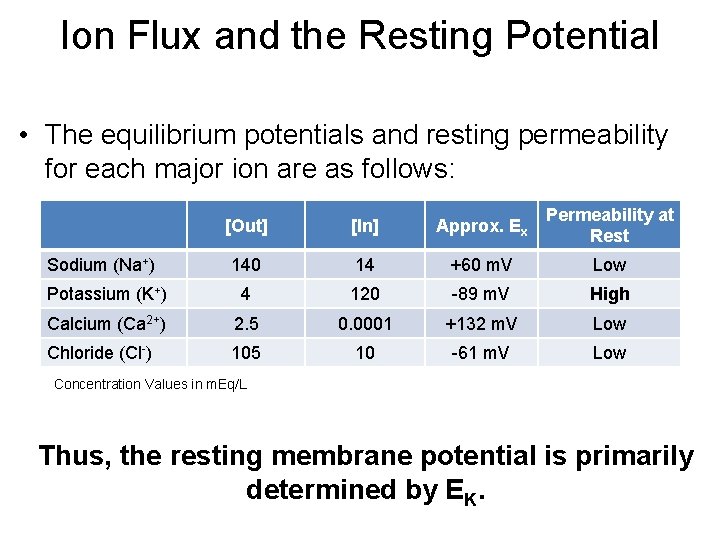
Ion Flux and the Resting Potential • The equilibrium potentials and resting permeability for each major ion are as follows: [Out] [In] Approx. Ex Permeability at Rest 140 14 +60 m. V Low Potassium (K+) 4 120 -89 m. V High Calcium (Ca 2+) 2. 5 0. 0001 +132 m. V Low Chloride (Cl-) 105 10 -61 m. V Low Sodium (Na+) Concentration Values in m. Eq/L Thus, the resting membrane potential is primarily determined by EK.

Nernst Equation Work in thermodynamics and his formulation of the Nernst heat theorem helped pave the way for the third law of thermodynamics. For this work he won the 1920 Nobel Prize in Chemistry. Nernst helped establish the modern field of physical chemistry and contributed to electrochemistry, thermodynamics and solid-state physics. Walther Hermann Nernst 25 June 1864 – 18 November 1941 Revered by students everywhere for developing the Nernst equation (1887).
![Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in) Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in)](http://slidetodoc.com/presentation_image_h2/6243f5dae730f7cb709355aa1737f25a/image-9.jpg)
Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in) @ 30° Eion = Ion Measured Z = charge [ion]out = concentration of ion outside of cell [ion]in = concentration of ion inside of cell
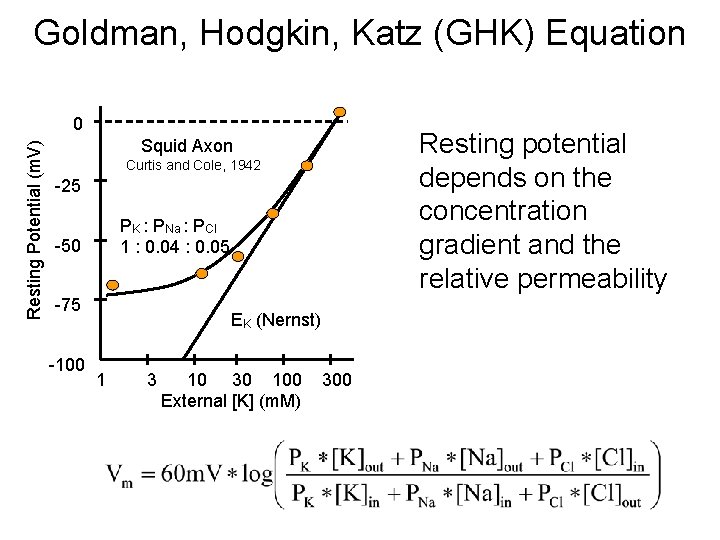
Goldman, Hodgkin, Katz (GHK) Equation Resting Potential (m. V) 0 Resting potential depends on the concentration gradient and the relative permeability Squid Axon Curtis and Cole, 1942 -25 PK : PNa : PCl 1 : 0. 04 : 0. 05 -50 -75 -100 EK (Nernst) 1 3 10 30 100 External [K] (m. M) 300
![Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in) Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in)](http://slidetodoc.com/presentation_image_h2/6243f5dae730f7cb709355aa1737f25a/image-11.jpg)
Nernst Equation Eion = (60 m. V / z) * log ([ion]out / [ion]in) @ 30° ENa = (60 m. V / 1) * log ([140 m. M]out / [14 m. M]in) = +60 m. V + EK = (60 m. V / 1) * log ([4 m. M]out / [120 m. M]in) = -89 m. V + ECa = (60 m. V/2) * log ([1. 25 m. M]out /[5 x 10 -5 m. M]in) = +132 m. V 2+ ECl- = (60 m. V / -1) * log ([105 m. M]out / [10 m. M]in) = -61 m. V
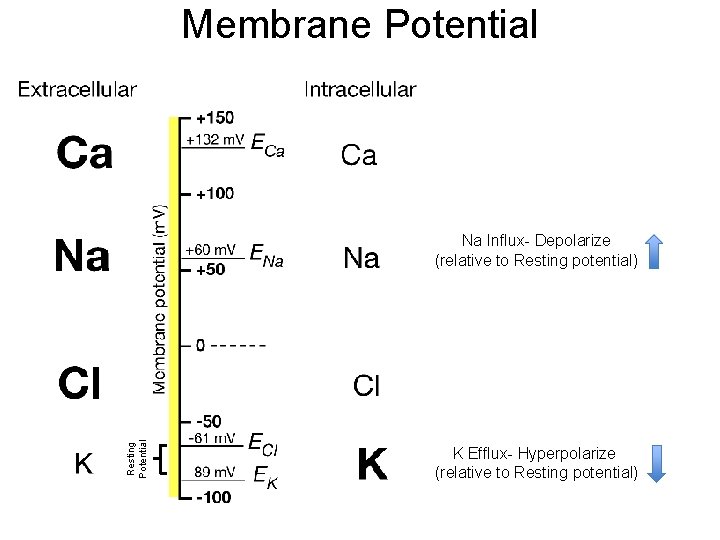
Membrane Potential Resting Potential Na Influx- Depolarize (relative to Resting potential) K Efflux- Hyperpolarize (relative to Resting potential)
- Slides: 12