MCB 135 K MidTerm I Review February 15

MCB 135 K Mid-Term I Review February 15, 2005 GSI: Laura Epstein

General Information • Mid-Term I – Friday – In Class Exam – 100 Points • 50 Points Multiple Choice and True / False • 50 Points Short Answer / Essay – Be prepared to place all personal belongings in aisle or at front of room before exam begins

Exam Material • • • Intro and Demography Comparative/Differential Aging Mitochondrial Decay Cellular Senescence Functional Assessment Epidemiology Telomeres Evolutionary Theory of Lifespan Oxidants, and Anti-Oxidants Neurodegeneration, Repair, and Plasticity Yeast as a model for cellular lifespan

Demography • Be familiar with: – – When does aging begin Role of genome and environment on aging What Demography is and how it is useful How life expectancy has changed in the last 200 years and what has contributed the most to this change – How the population distribution has changed and what impact this might have for the future – Centenarians and aging • What have we learned from these studies – How old was the oldest living documented person of each sex

Demography • Statistical study of human populations: – Size and density distribution • Vital Statistics: – Epidemiology: Births, deaths, diseases
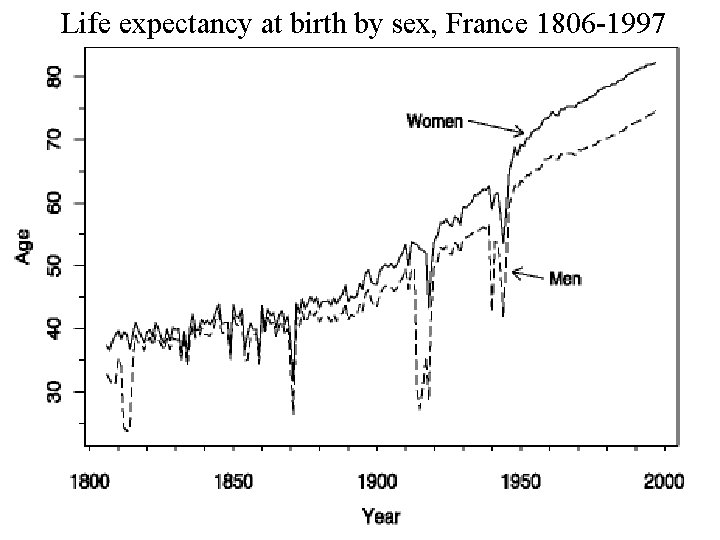
Life expectancy at birth by sex, France 1806 -1997
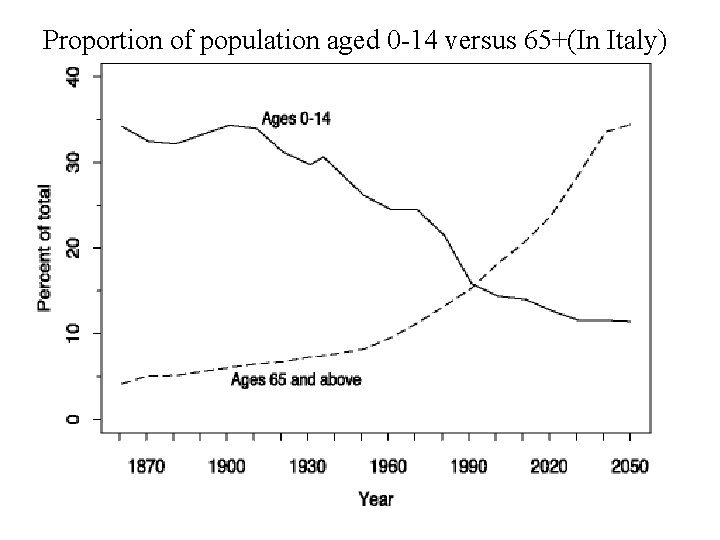
Proportion of population aged 0 -14 versus 65+(In Italy)

Centenarians • Generally good health – Escapers – Late onset of disease – Early disease that was overcome • SSC (Semi-Super) – 105+ • SC (Super) – 110+ • Possible role of IGF-1 Receptor • Oldest Female – 122 years – Jeanne Calment • Oldest Male – 115 years – Christian Mortensen

Women and Longevity • Probable Causes for increased longevity – Genetic – Environmental – Lesser Life Stress – Decreased Smoking – Protective Hormones – Better Protection Against Oxidative Damage

Comparative / Differential Aging • Be familiar with: – What are the models for study – What does physiological assessment require – What are the physiological correlates with longevity and how do they correlate

Figure 3. 1: Comparative Maximum Life Spans **Detailed discussion of figure in the legend, pg. 26

Mitochondrial Decay • Be familiar with: – – – Mitochondria function/damage Acetyl Carnitine (ALCAR) R-a-Lipoic Acid (LA) Behavior of old rats fed ALCAR and LA Micronutrient under-nutrition in Americans

Mitochondria • O 2 ® O 2 - ® H 2 O 2 ® · OH ® H 2 O, oxidative metabolism • - Reaction intermediates are similar to products formed by radiation exposure. • -We are always making lesions in our DNA and fixing it • -With infection, our immune cells fight the infection and release free radicals • - 1 -2% leakage of reactive oxygen species (ROS)

Oxidative Damage Products from Base Excision Repair (BER) and Nucleotide Excision Repair (NER) are secreted in the urine. - Frequency of oxidative events increases in older individuals Rat liver cells – Young ~24, 000 oxidative lesions/cell Old ~67, 000/cell
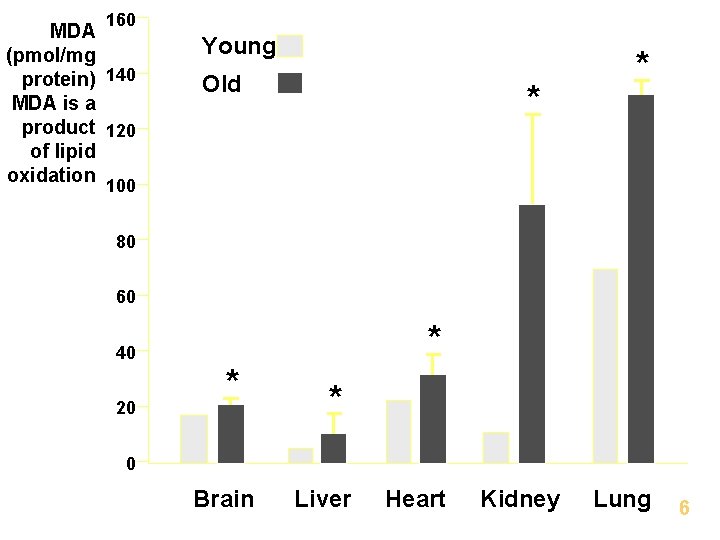
160 MDA (pmol/mg protein) 140 MDA is a product 120 of lipid oxidation Young Old * * 100 80 60 40 20 * * * 0 Brain Liver Heart Kidney Lung 6

Mitochondria from old rats compared to those from young rats: 1) Lower Cardiolipin—a lipid used in the mitochondrial membrane 2) Lower Membrane Potential 3) Lower Oxygen Utilization 4) Increased Oxidant Leakage

L-Carnitine/Acetyl-L-Carnitine (ALCAR) • Transports long-chain fatty acids into mitochondria • Removes short- and medium-chain fatty acids that accumulate • Feeding ALCAR to aging rats is able to suppress or ameliorate many age-related changes in mitochondrial function and oxidant stress. • restores membrane potential • although oxidant leakage remains • • Mediates the ratio of acetyl-Co. A/Co. A Decreases with age in plasma and in brain Improves cognitive function in rats ALCAR is something beneficial!! And we lose it with age! 12
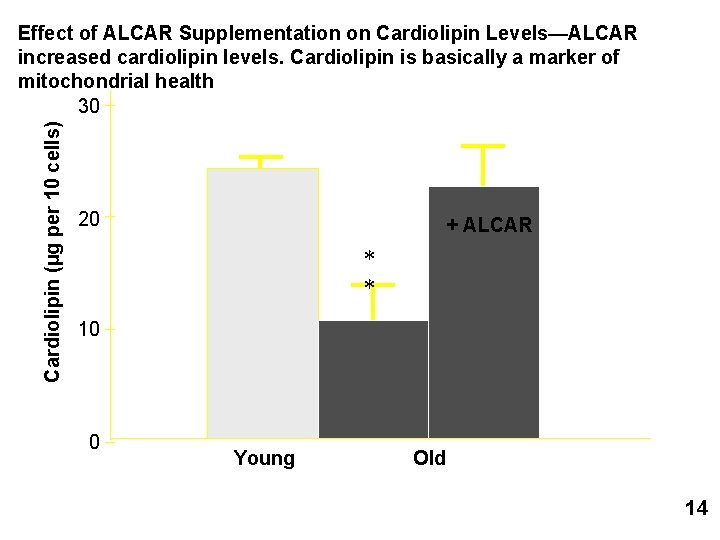
Cardiolipin (µg per 10 cells) Effect of ALCAR Supplementation on Cardiolipin Levels—ALCAR increased cardiolipin levels. Cardiolipin is basically a marker of mitochondrial health 30 20 + ALCAR * * 10 0 Young Old 14

• R-a -Lipoic Acid (LA) – – Potent Anti-oxidant Lowers oxidants in old rats Restores ascorbic acid levels (vitamin C) Restores glutathione levels (another antioxidant) – Has been shown to be beneficial to rats in combination with ALCAR

• Behavior of old rats fed ALCAR and LA – ALCAR/LA can reverse age-associated losses in cognitive function – Increased ambulatory activity
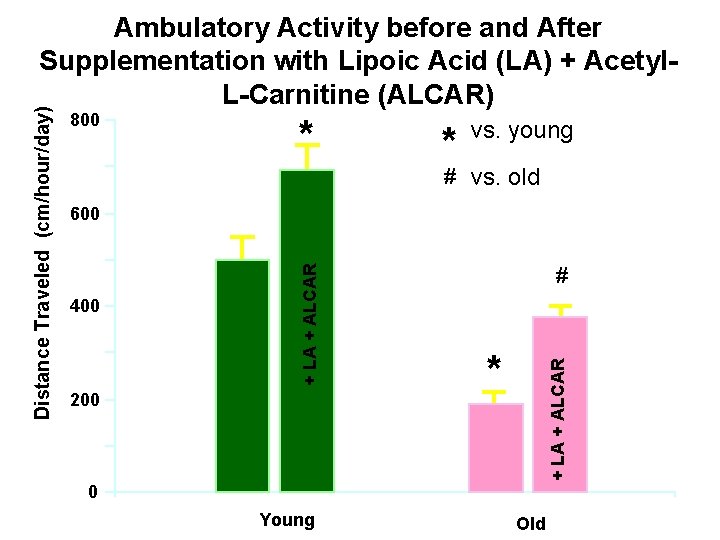
800 * * vs. young # vs. old 600 200 # * + LA + ALCAR 400 + LA + ALCAR Distance Traveled (cm/hour/day) Ambulatory Activity before and After Supplementation with Lipoic Acid (LA) + Acetyl. L-Carnitine (ALCAR) 0 Young Old

• As organisms age… • - mitochondrial potential decreases. • - oxidant leakage increases.

• Do americans get enough micronutrients? NO • Iron deficiency, results in loss of complex 4 (part of the electron transport chain in the mitochonrial membrane). – Complex 4 loss results in increased oxidative stress. – Iron deficiency mimics neurodegeneration. • Zinc deficiency – Increased DNA damage in zinc deficient cells • Biotin deficiency – Accelerates cell senescence • Magnesium deficiency

Other types of oxidative damage increase with age • Protein Carbonylation (Stadtman) • - Lipid oxidation (aldehydes "stuck" to proteins/enzymes) • - age pigment -fluorescent lipid peroxides accumulate with age, can see fluorescence in tissues of older individuals (humans and rats) • Mitochondria may deteriorate with time, old or damaged mitochondria may be destroyed by lysosomes in the cytoplasm.

Senescence • Be familiar with: – What senescence is – What the types of senescence are – What causes senescence – Why senescence is important to aging and disease

Cellular Senescence What is it? Response of normal cells to potentially cancer-causing events
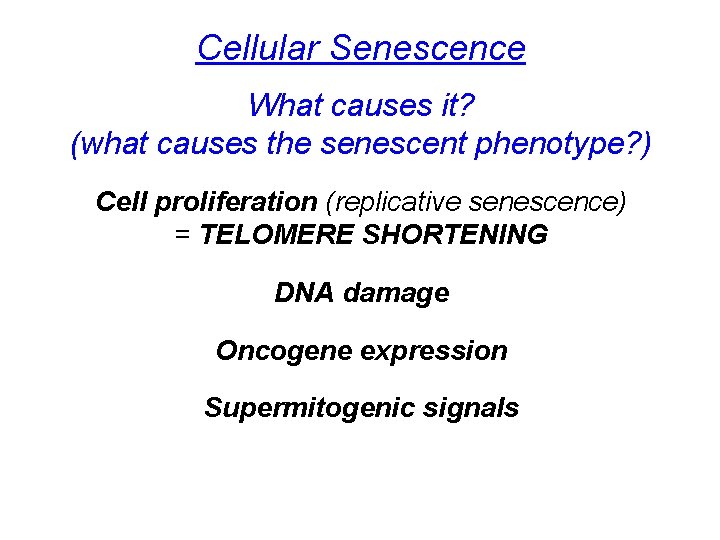
Cellular Senescence What causes it? (what causes the senescent phenotype? ) Cell proliferation (replicative senescence) = TELOMERE SHORTENING DNA damage Oncogene expression Supermitogenic signals
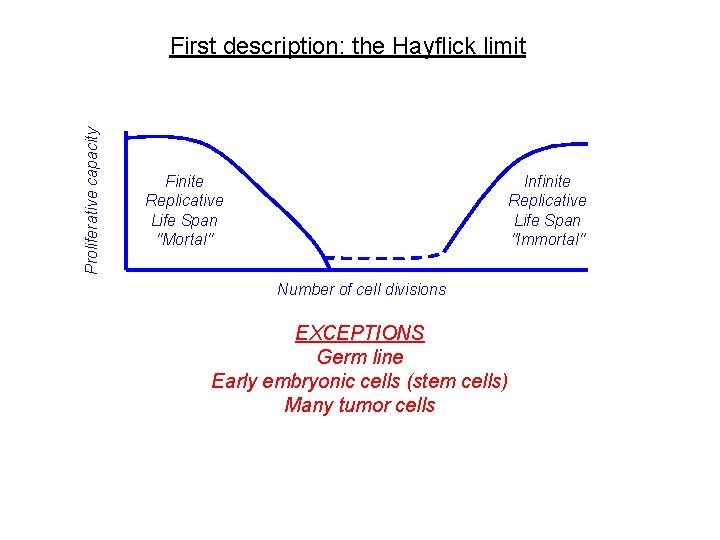
Proliferative capacity First description: the Hayflick limit Finite Replicative Life Span "Mortal" Infinite Replicative Life Span "Immortal" Number of cell divisions EXCEPTIONS Germ line Early embryonic cells (stem cells) Many tumor cells

What happens when cells exhaust their replicative life span REPLICATIVE SENESCENCE • Irreversible arrest of cell proliferation (universal) • Resistance to apoptosis (stem cells) • Altered function (universal but cell type specific) SENESCENT PHENOTYPE
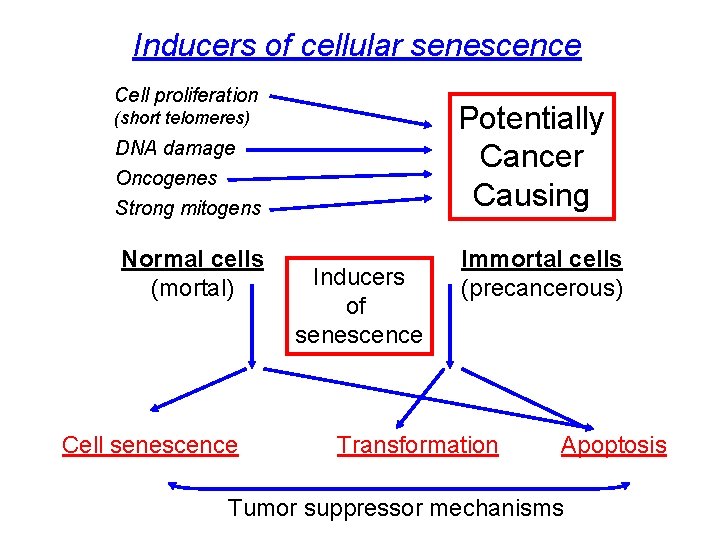
Inducers of cellular senescence Cell proliferation Potentially Cancer Causing (short telomeres) DNA damage Oncogenes Strong mitogens Normal cells (mortal) Cell senescence Inducers of senescence Immortal cells (precancerous) Transformation Apoptosis Tumor suppressor mechanisms

p 53 and p. RB proteins • Nuclear proteins controlled by complex pathways (upstream regulators and downstream effectors) • Control expression of other genes • Halt cell cycle progression in response to inducers of senescence • Crucial for allowing normal cells to sense and respond to senescence signals

Cellular Senescence An important tumor suppressor mechanism What does cellular senescence have to do with aging? • The senescent phenotype entails changes in cell function • Aging is a consequence of the decling force of natural selection with age
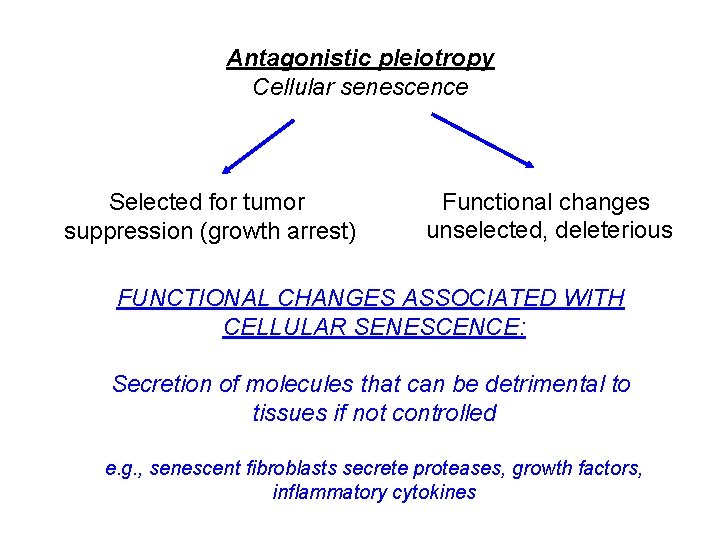
Antagonistic pleiotropy Cellular senescence Selected for tumor suppression (growth arrest) Functional changes unselected, deleterious FUNCTIONAL CHANGES ASSOCIATED WITH CELLULAR SENESCENCE: Secretion of molecules that can be detrimental to tissues if not controlled e. g. , senescent fibroblasts secrete proteases, growth factors, inflammatory cytokines

Functional Assessment • Be familiar with: – What comprises geriatric assessment – What programs are used to test assessment and what are their parameters – Why do women have more disability – Compare/Contrast aging with physical inactivity • Know some parameters – Aging vs. Disease

Geriatric Assessment Involves a multi-dimensional diagnostic process designed to qualify an elderly individual in terms of: • Functional capabilities • Disabilities • Medical & Psychological characteristics A list of typical assessments is summarized in Table 3. 3 For our discussion, we will consider particularly: • Activities of Daily Living (ADL) • Instrumental Activities of Daily Living (IADL) **See Table 3. 4**

Assessment Programs include tests that are grouped into three categories: 1. Tests examining general physical health 2. Tests measuring ability to perform basic self care (ADLs) 3. Tests measuring ability to perform more complex activities (IADLs), reflecting the ability to live independently in the community

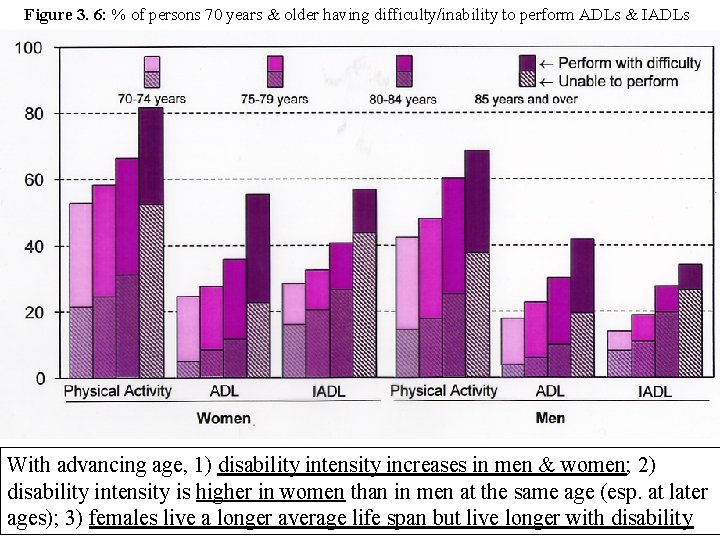
Figure 3. 6: % of persons 70 years & older having difficulty/inability to perform ADLs & IADLs With advancing age, 1) disability intensity increases in men & women; 2) disability intensity is higher in women than in men at the same age (esp. at later ages); 3) females live a longer average life span but live longer with disability


Aging is associated with increased incidence of: • Diseases • Accidents • Stress

Aging should be differentiated from Disease: • is selective; it varies with the species, tissue, organ, cell molecule • may depend on intrinsic & extrinsic factors • is discontinuous (may progress, regress or be arrested) • is occasionally deleterious, damage is often variable/reversible • is often treatable with known cause(s)

Assessment of Physiological Age in Humans Physiological age depends on Physiologic competence: good to optimal function of all body systems & Health status: absence of disease Physiological age may or may not coincide with chronological age


Epidemiology • Be familiar with: – What is epidemiology and why is it useful – Why is it thought that older people are at an elevated risk for disease – What are the major age associated causes of death – Understand why falls are a problem for the elderly

EPIDEMIOLOGY OF AGING • THE STUDY OF THE AGE-RELATED DISTRIBUTION AND CAUSES OF DISEASE, DISABILITY, AND MORTALITY IN HUMAN POPULATIONS.

EPIDEMIOLOGY OF AGING • ACCUMULATION OF ENVIRONMENTAL/BEHAVIORAL INSULTS. • REDUCED IMMUNOLOGICAL SURVEILLANCE

EPIDEMIOLOGY OF AGING • WHY IMPORTANT? – AGING OF THE HUMAN POPULATION – HEALTH AND VITALITY OF AN AGING POPULATION – QUALITY OF LIFE AND COST OF CARE

EPIDEMIOLOGY OF AGING • MAJOR AGE-ASSOCIATED CAUSES OF DEATH – CANCER – CARDIOVASCULAR DISEASE – CHRONIC OBSTRUCTIVE PULMONARY DISEASE – DIABETES

EPIDEMIOLOGY OF AGING AGE-SPECIFIC COLORECTAL CANCER INCIDENCE RATES (Per 100, 000 in population <65 65+ WM WF 20. 4 14. 7 408. 0 269. 3 ) BM BF 25. 3 20. 4 385. 8 286. 1

EPIDEMIOLOGY OF AGING COGNITIVE FUNCTION Moderate/Severe Memory Impairment 65 -69 85+ Male 5. 3 37. 3 Female 3. 8 35. 0

EPIDEMIOLOGY OF AGING • FALLS • 30% OF PEOPLE AGED 65+ FALL EACH YEAR. • 10 -15% OF THOSE FALLS ARE CONSIDERED “SERIOUS/NON-FATAL” • FALLS REPRESENT THE LEADING CAUSE OF ACCIDENTAL DEATH IN PEOPLE AGED 65 AND OLDER. • FEAR OF FALLING IS A LEADING REASON FOR NOT ENGAGING IN PHYSICAL ACTIVITY.

EPIDEMIOLOGY OF AGING • CAUSES OF FALLS IN THE ELDERLY • - DIZZINESS • - POOR COGNITIVE FUNCTION • - VISION PROBLEMS • - GENERAL FRAILTY • - ENVIRONMENTAL HAZARDS

Telomeres • Be familiar with: – What telomeres are – Why telomeres are important – Consequence of telomere end-shortening – What telomerase is – The telomere hypothesis of aging
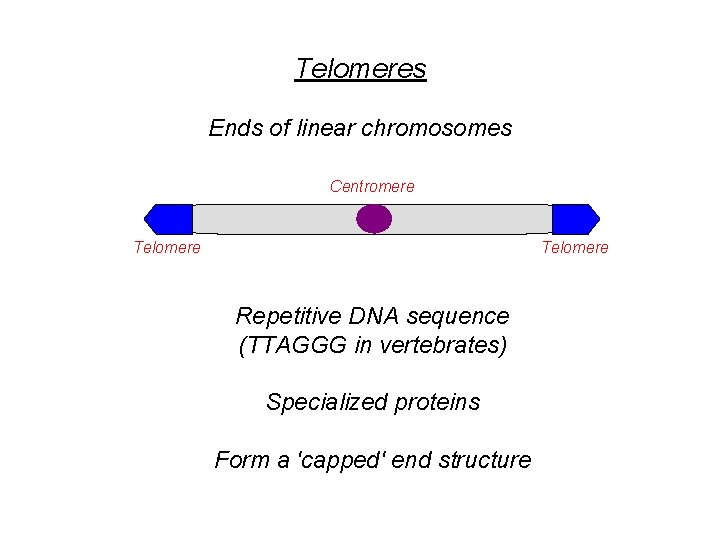
Telomeres Ends of linear chromosomes Centromere Telomere Repetitive DNA sequence (TTAGGG in vertebrates) Specialized proteins Form a 'capped' end structure
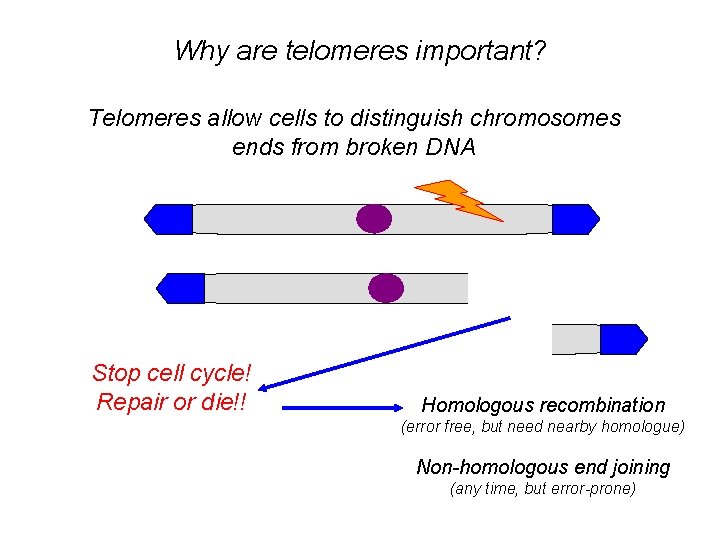
Why are telomeres important? Telomeres allow cells to distinguish chromosomes ends from broken DNA Stop cell cycle! Repair or die!! Homologous recombination (error free, but need nearby homologue) Non-homologous end joining (any time, but error-prone)
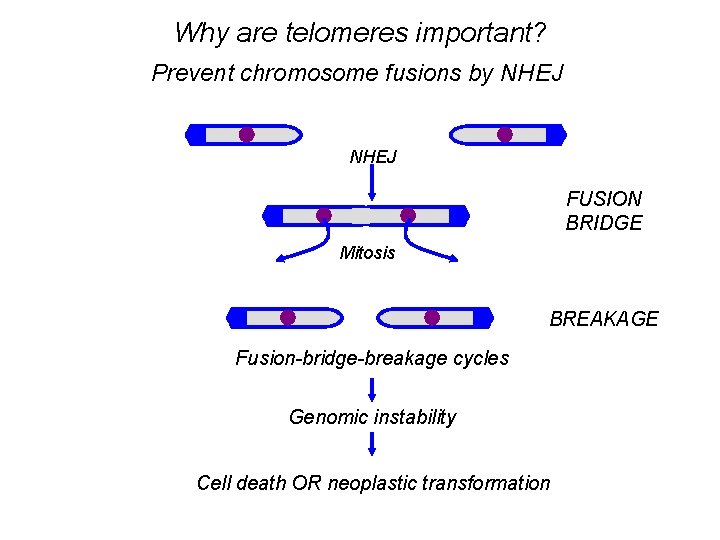
Why are telomeres important? Prevent chromosome fusions by NHEJ FUSION BRIDGE Mitosis BREAKAGE Fusion-bridge-breakage cycles Genomic instability Cell death OR neoplastic transformation
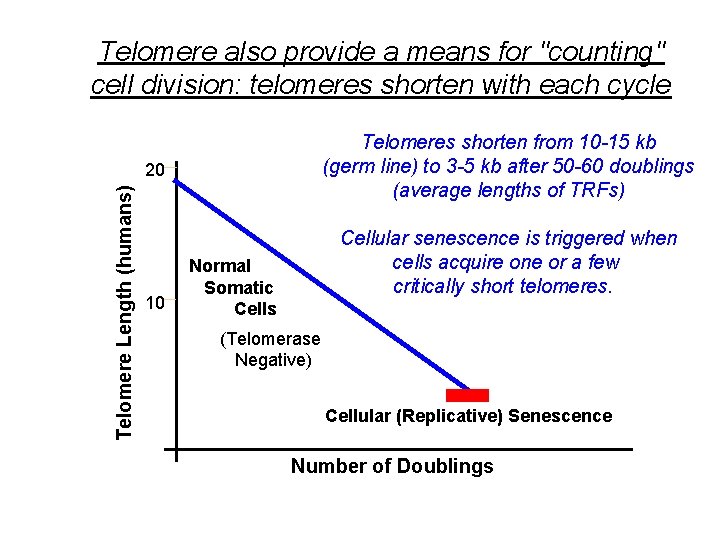
Telomere also provide a means for "counting" cell division: telomeres shorten with each cycle Telomeres shorten from 10 -15 kb (germ line) to 3 -5 kb after 50 -60 doublings (average lengths of TRFs) Telomere Length (humans) 20 10 Cellular senescence is triggered when cells acquire one or a few critically short telomeres. Normal Somatic Cells (Telomerase Negative) Cellular (Replicative) Senescence Number of Doublings

TELOMERASE: Key to replicative immortality Enzyme (reverse transcriptase) with RNA and protein components Adds telomeric repeat DNA directly to 3' overhang (uses its own RNA as a template) Vertebrate repeat DNA on 3' end: TTAGGG Telomerase RNA template: AAUCCC

The telomere hypothesis of aging Telomeres shorten with each cell division and therefore with age TRUE Short telomeres cause cell senescence and senescent cells may contribute to aging TRUE HYPOTHESIS: Telomere shortening causes aging and telomerase will prevent aging TRUE OR FALSE?

The telomere hypothesis of aging Telomere length is not related to life span (mice vs human; M musculus vs M spretus) Telomeres contribute to aging ONLY if senescent cells contribute to aging Telomerase protects against replicative senescence but not senescence induce by other causes

Evolutionary Theory of Lifespan • Be familiar with: – – – – Evolutionary theory of lifespan Aging in nature Disposable soma Antagonistic Pleiotropy Mutation Accumulation Trait that correlate with longevity Model systems that agree and disagree with theory
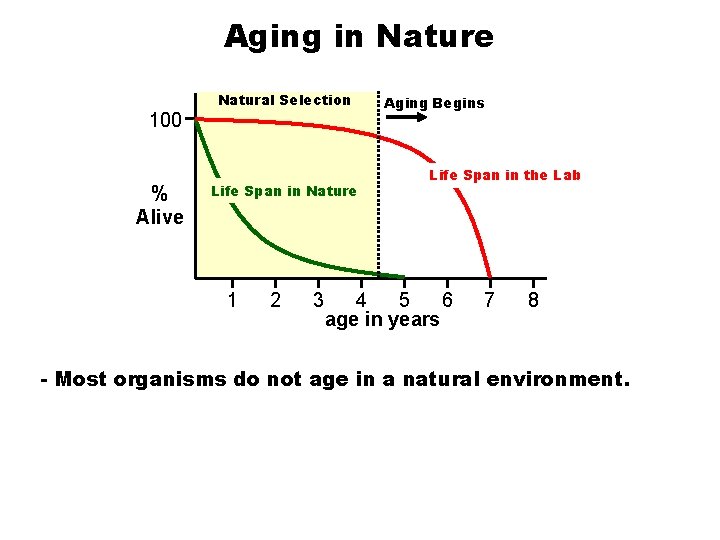
Aging in Nature 100 % Alive Natural Selection Life Span in Nature 1 2 3 Aging Begins Life Span in the Lab 4 5 6 age in years 7 8 - Most organisms do not age in a natural environment.

Lifespan and extrinsic mortality: -If mortality is high an organism will die from predation or other hazards before it grows old. -Therefore, if extrinsic mortality limits survival there is no reason to evolve a life span that is longer than an organism would normally survive in nature.

Evolutionary Theories of Aging Disposable Soma - Somatic cells are maintained only to ensure continued reproductive success, following reproduction the soma is disposable. (life span theory) Antagonistic Pleiotropy - Genes that are beneficial at younger ages are deleterious at older ages. (Pleiotropism = The control by a single gene of several distinct and seemingly unrelated phenotypic effects) Mutation Accumulation - Mutations that affect health at older ages are not selected against (no strong evidence).

Opossums and Life Span - ultimate prey, ~ 80% die from predation - typically reproduce once - age very rapidly -Hypothesis: The presence of predators limits life span, natural selection favors somatic maintenance for only as long as an average opossum can be expected to live. -How could you test this hypothesis? Steve Austad, U. of Idaho

Sapelo Island Opossums - no predators (out in daytime) - longer average life span - reproduce twice (fewer offspring/litter) -Are these changes due to a lack of predators, or a physiological change that delays the aging process? Physiological Change - Sapelo island opossums not only live longer, they age slower than mainland animals. -Sapelo Island opossums have less oxidative damage than mainland opossums. (collagen X-linking)
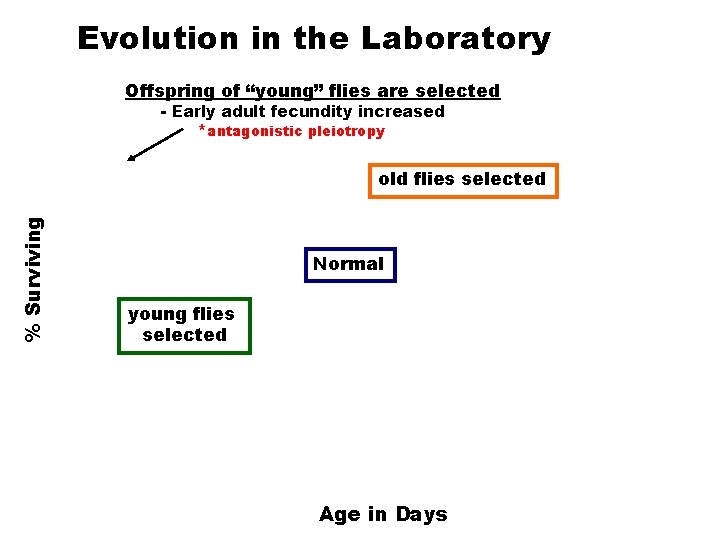
Evolution in the Laboratory Offspring of “young” flies are selected - Early adult fecundity increased *antagonistic pleiotropy % Surviving old flies selected Normal young flies selected Age in Days

Summary of Drosophila Selection 1) Selection at age of reproduction can alter the lifespan of Drosophila (lifespan has been doubled by this technique). 2) Increase in lifespan has a cost, reduced fecundity (reproduction). - antagonistic pleiotropy - 3) Long-lived flies are stress resistant (heat shock, oxidants).
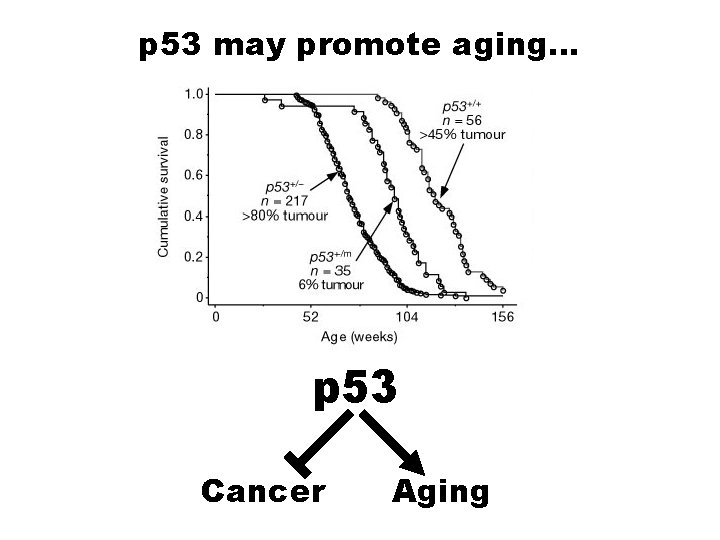
p 53 may promote aging… p 53 Cancer Aging

Why did we evolve a system that limits our lifespan? -to protect against cancer! (Antagonistic Pleiotropy again!)

Life Span versus Aging - can not be selected for, results from an absence of natural selection. Life Span - results from selection and extrinsic mortality Environmental Selection - predators, natural hazards Social Selection - parental investment, sexual behavior

Main Ideas 1. Life span results from selective pressure. 2. Life span is inversely proportional to extrinsic mortality. 3. Aging results from a lack of natural selection with age.

Oxidants and Anti-Oxidants • Be familiar with: – – – What is the Free Radical Theory of Aging What is a free radical What are the implications of free radicals on aging How oxygen can be toxic What the major oxidants are and what are oxidant sources – What are the major antioxidants – Experimental Models

Free Radicals • Free radicals are unstable • React quickly with other compounds, doing cell and body damage • Once produced, they multiply unless neutralized by anti-oxidants or other free -radical scavengers. • Free radicals rarely occur in nature • Oxygen can have several unpaired electron “pairs”

The Free Radical Theory of Aging “Aging results from the deleterious effects of free radicals produced in the course of cellular metabolism” Harman D. , Aging: A theory based on free radical and radiation chemistry, J. Gerontol. 11: 298, 1956

Free Radical Chemistry • • • Reactive radicals attack indiscriminately Can add to unsaturated bonds Can abstract electrons or hydrogen atoms Propagate chain reactions Can cause bond scission Can cause crosslinking – Crosslinking - Formation of bonds among polymeric chains • Produce secondary toxic agents

Implications for Aging • Some free radical-induced chemical modifications may have unique impacts • Crosslinked products may not be degradable • Scission of bonds in DNA, particularly multiple events may erase vital information

What are the Major Oxidants? • • . Hydroxyl radical (OH. ) Hypochlorite (HOCl) Singlet oxygen 1 O 2 Peroxynitrite (OONO-) Hydrogen peroxide (H 2 O 2) Free or loosely-bound iron, copper or heme. Superoxide radical (O 2 -). Nitric oxide (NO )

Lipid Peroxidation • PUFAs* contain weakly bonded hydrogen atoms between double bonds • Chain reactions are probable because of high local concentrations of double bonds *PUFAs means polyunsaturated fatty acids

Oxidant Sources Table 5. 1 Regulated Unregulated • Enzymes involved • in “Leaky” electron transport • Damaged proteins and lipids cell signaling • Toxins (food, water) • Immune cells • Smoke • Irradiation (UV)

Major Antioxidants Table 5. 2 • • Vitamins E and C Thiols, particularly glutathione Uric acid Superoxide dismutases (Cu/Zn or Mn SOD) Catalase and glutathione peroxidase Heme oxygenases Protein surface groups (Msr)

Glucose and Oxidants • In cell culture models high glucose correlates with oxidant production • Three diabetes-linked effects can be correlated with superoxide production • Insulin pathway and life-span extension

Are Oxidants the Cause of Aging? (Table 5. 8) Pro Con • Caloric restriction may reduce oxidative stress • Life span extension in mutants may be associated with stress resistance • Knockout mice lacking Mn. SOD have restricted survival • Enzyme mimetics extend life span in some aging models. • Some drugs, probably acting as antioxidants, have been claimed to extend lifespan. • Vitamin C, a superb free radical scavenger, is not synthesized by long-lived primates. • Chronic radiation in low doses does not shorten life span (may increase it). • Dietary supplementation with Vitamin E and C does not extend life span. • Tissue comparison (brain vs. muscle) seems incompatible with oxidant/antioxidant models of aging. • Exercise, that increases oxidant stress, improves life span.

Neurodegeneration, Repair, and Plasticity


Neurodegeneration, Repair, and Plasticity • Neuroendocrine theory of aging: Alterations in either the number or the sensitivity of various neuroendocrine receptors gives rise to homeostatic or homeodynamic changes that result in senescence.

Neuron regeneration? How is that possible? • While until the 1990 s we thought neurons couldn’t regenerate, now we’ve seen that certain neurons have the potential to regenerate under specific circumstances. – Neurons in lining of cerebral ventricles – Hippocampus – Neuroglia (astrocytes and oligodendrocytes) – Microglia (“macrophages” of nervous system)

What conditions favor regeneration? • Whole body: – – – Exercise Nutrition Some stress Education Good circulation • Neural microenvironment – Brain metabolism – Hormonal changes

Education and death rates • • Higher education, lower death rates Higher education, lower disability Higher income, lower death rates Comments?

Why would education decrease death rates? • • • Access to medical care Access to exercise Better nutrition Higher income Responsibility to health behaviors Lower rates of smoking and alcohol

Brain reserve capacity • When these things (listed in previous slide) happen in young life, brain reserve capacity built • This means more neuronal branches and more axonal/dendritic connections, better brain blood supply • Evidence: a person with more education has longer dendritic branching length and more connections • With old age there is denudation of neurons—less branching

Yeast as a Model for Lifespan • Be familiar with: – A molecular cause of yeast aging – SIR 2 – Aging and genetic instability, in yeast and humans

Cellular senescence: finite replicative capacity of mitotically dividing cells • Originally observed in human diploid fibroblasts (Hayflick limit, 1965) • Represents a limit on the number of population doublings • Caused by telomere shortening in cells that do not express telomerase

What about simple eukaryotic cells that do express telomerase? • Cells of baker’s yeast, Saccharomyces cerevisiae, express telomerase • Microbial populations are “immortal”, can be passaged forever • Does this mean these cells are also immortal?

What is the role of telomere length in yeast cellular senescence? • Telomerase is expressed throughout the lifespan • Telomere length is maintained throughout the lifespan • Mutating telomerase does cause cellular senescence: telomere shortening, limited population doublings, genomic instability, ALT

What causes yeast aging? • A clue: exceptions to the rule of the resetting clock • Occasionally, daughters of old mothers are born prematurely aged! • Their lifespan equals the mother’s remaining lifespan • The asymmetry has broken down -- accompanied by loss of size asymmetry (“symmetric buds”) • The daughters of symmetric buds have normal lifespan • Suggests these symmetric buds have inherited a “senescence factor”…

What is the yeast senescence factor? • Some clues (late 1990 s): – Aging is accompanied by fragmentation of the nucleolus – The nucleolus assembles at the site of r. RNA transcription, the r. DNA – Normally, sir 2 tries to keep rdna from popping out. – sir 2 mutants have a short lifespan – sir 2 mutants have high levels of extrachromosomal r. DNA circles (ERCs) – ERCs have the characteristics of the senescence factor…
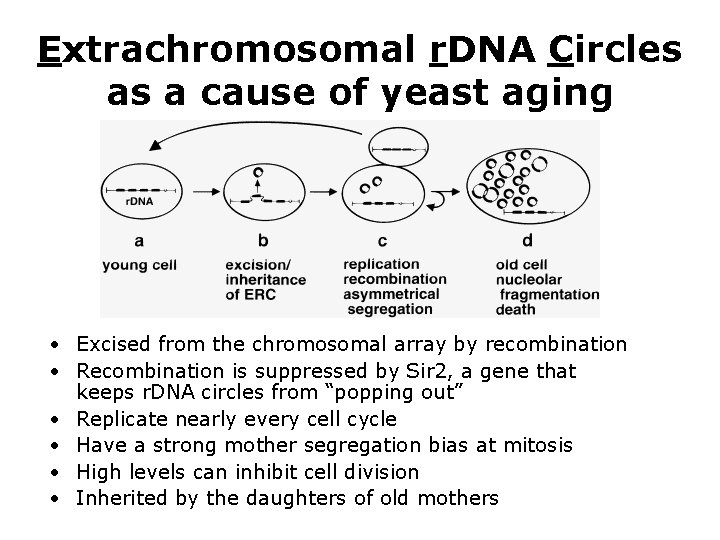
Extrachromosomal r. DNA Circles as a cause of yeast aging • Excised from the chromosomal array by recombination • Recombination is suppressed by Sir 2, a gene that keeps r. DNA circles from “popping out” • Replicate nearly every cell cycle • Have a strong mother segregation bias at mitosis • High levels can inhibit cell division • Inherited by the daughters of old mothers

But, no ERCs in humans! (or mice, or worms, or flies…) Why continue to study yeast aging? • Overexpressing SIR 2 homologs in flies and worms extends lifespan • Perhaps the regulation of lifespan is conserved (and SIR 2 -dependent) while the molecular effectors of aging vary between organisms • Example: calorie restriction (CR)—data is not resolved on this

Calorie Restriction (CR) Extends Lifespan • Decreasing caloric intake (without starvation) lengthens lifespan • Works in yeast, flies, rats, mice, worms, … • Many reports claimed that the CR pathway is SIR 2 -dependent, supporting theory of SIR 2 as master aging regulator • Heated debate over the mechanism by which SIR 2 influences CR pathway • Recent work has shown that in yeast CR is actually SIR 2 -independent

Genetic instability and Aging • Frequencies of mutations and chromosomal rearrangements increase with age in various organisms • Incidence of cancer increases dramatically with age: • Is this due to accumulation of genetic events at a constant rate over the lifetime, or does aging itself alter the rate of new genetic events?

An Age-induced Hyper-recombinational State • After about 25 divisions, aging mother cells begin to produce daughters that are genetically unstable • High rates of LOH at multiple chromosomes • LOH is caused by recombination, not chromosome loss or deletion • Behaves as a “switch” to a new, unstable state • Hyper-recombinational state is eventually “diluted” in progeny of old cells
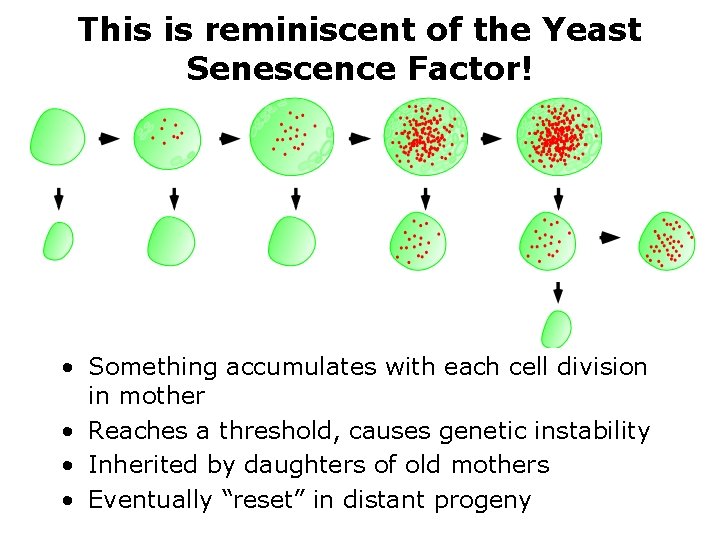
This is reminiscent of the Yeast Senescence Factor! • Something accumulates with each cell division in mother • Reaches a threshold, causes genetic instability • Inherited by daughters of old mothers • Eventually “reset” in distant progeny

Are ERCs the cause? • Mutations that increase ERCs (sir 2) do not accelerate onset of switch • Mutations that decrease ERCs do not delay onset of switch • In fact, onset of switch is unlinked to lifespan! • Suggests an important distinction between longevity and functional senescence! This is key!! There could be different senescent factors affecting different processes of senescence in cell—ie one affecting genomic instability and one affecting division
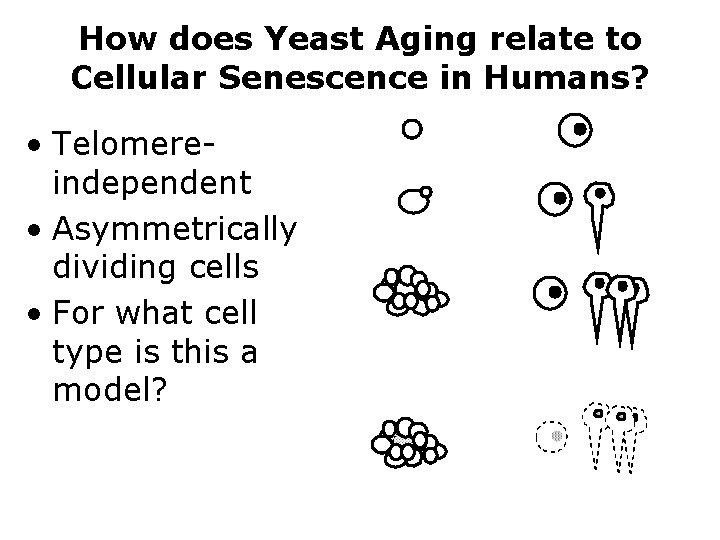
How does Yeast Aging relate to Cellular Senescence in Humans? • Telomereindependent • Asymmetrically dividing cells • For what cell type is this a model?

Stem cells in human aging and cancer • Yeast aging may be a model for continuously dividing, asymmetrically dividing stem cells. And they express telomerase

Conclusions • Yeast aging involves longevity regulation as well as senescence phenotypes unlinked from longevity • Genetic instability increases with age in yeast, by an epigenetic hyperrecombinational switch (not by accumulating gene mutations but by a switch method) • May be a good model for stem cell aging
- Slides: 107