MCB 101 Final Exam 1 30 4 30

MCB 101 Final Exam: 1: 30 – 4: 30 PM Friday, 5/3/2019 sections A, B, C & D in 1024 Chem. Annex sections E, F & G in 100 Gregory Hall Conflict Final Exam: Contact KC if you need to take the conflict exam! Times: 8 – 11 am Fri. 5/3/2019 1: 30 – 4: 30 pm Mon. 5/6/2019 7 – 10 pm Mon. 5/6/2019 8 – 11 am Tue. 5/7/2019 What is the nature of your conflict? What is the best time for you to take the exam?

Exam – 3 (the final) will be worth 100 points, the format will be similar to exams 1 and 2. Material Covered: The test will mostly cover Experiments 15 - 19, however, there will be questions that come from earlier material that is conceptually linked to materials in Experiments 15 – 19. Example: In experiment 15 c we looked at the effects of disinfectants and antiseptics on the growth of a Gram negative bacterium and a Gram positive bacterium. Review the differences between Gram negative & Gram positive bacteria.

Lab Practical Parts C & D - Serial Dilutions and Spread Plate (for a viable plate count) - Dilution Calculations Date: April 22 – 23, 2019 during regular lab times. Study Guide - Make sure that you know how to use a micropipette! - Do the dilution calculation problems on pages 208 – 211

MCB 101 Introductory Microbiology Lab Lecture 11 April 19, 2019 Week of April 15 – 19 - Ex. 16 Preservatives and Mold Inhibition, Results - Ex. 17 Pasteurization of Milk, Set Up and Results - Ex. 18 A Fermented Milk Products, Curds and Whey Week of April 22 - 26 -Ex. 18 B Yogurt – set up & results, Ex. 18 C cottage cheese – set up - Ex. 19 Nitrogen Metabolism and Soil Bacteria (start) April 22 – 23 Lab Practical Parts C & D - Dilution & Spread Plate, Dilution Calculations LON-CAPA Pre-Lab 13 over Enrichments and The Nitrogen Cycle Due Monday, 4/22/19 at 8: 00 AM LON-CAPA Quiz 11 Fermented Milk Products and The Nitrogen Cycle, Due Friday, 4/26/19 at 8: 30 AM

The Composition and Chemistry of Cow’s Milk - water-based (p. H near neutral) - butterfat globules (in emulsion, % varies) - carbohydrates, lactose, concentration = 4 – 6% - proteins, casein and lactoglobulin, conc. = ~3. 5% - salts & minerals (good source of calcium phosphate)

MCB 101 Lecture 11 April 19, 2019 Put the following liquids in order ranked from lowest p. H (acidic) to highest p. H (basic). - whey from yogurt - whey from cheese - ammonia based window cleaner - 0. 10 M Hydrochloric Acid - milk Which order is correct? (low p. H to high p. H) A. B. C. D. E. milk, cheese, HCl, yogurt, NH 3 yogurt, cheese, HCl, NH 3, milk HCl, yogurt, cheese, milk, NH 3 HCl, milk, cheese, yogurt, NH 3 HCl, cheese, milk, NH 3, yogurt

MCB 101 Lecture 11 April 19, 2019 Put the following liquids in order ranked from lowest p. H (acidic) to highest p. H (basic). - whey from yogurt - whey from cheese - ammonia based window cleaner - 0. 10 M Hydrochloric Acid - milk Which order is correct? (low p. H to high p. H) A. milk, cheese, HCl, yogurt, NH 3 B. yogurt, cheese, HCl, NH 3, milk C. HCl, yogurt, cheese, milk, NH 3 1 4. 5 5. 5 7 10 D. HCl, milk, cheese, yogurt, NH 3 E. HCl, cheese, milk, NH 3, yogurt

Milk is a good for a young mammal. Therefore, milk is a good for a wide variety of chemoheterotrophic bacteria. Excessive bacterial growth causes spoilage. Different Types of Spoilage Taste Microbial Cause Sour fermentation of sugars with the production of small organic acids Rancid breakdown of fats with the production of ketones and 3 C – 8 C organic acids Putrid breakdown of proteins with the production of ammonia and amines

A key goal in cheese and yogurt production is to extend the shelf life of the milk. 1) A low p. H can inhibit growth of bacteria that can cause milk to become putrid or rancid. 2) Precipitation of casein to make a curd can also slow down the microbial digestion of this protein. Lactic acid bacteria can sour milk without making unpleasant fermentation products. A low p. H reduces the solubility of casein.

Casein – the major storage protein in milk There a few different types of protein molecules found in casein. A clump of casein molecules is called a micelle. Casein micelles contain calcium phosphate as well as protein. A soluble casein particle is called a submicelle by some people. The outer surface of a casein submicelle includes k-casein, (kappa-casein) which is hydrophilic.
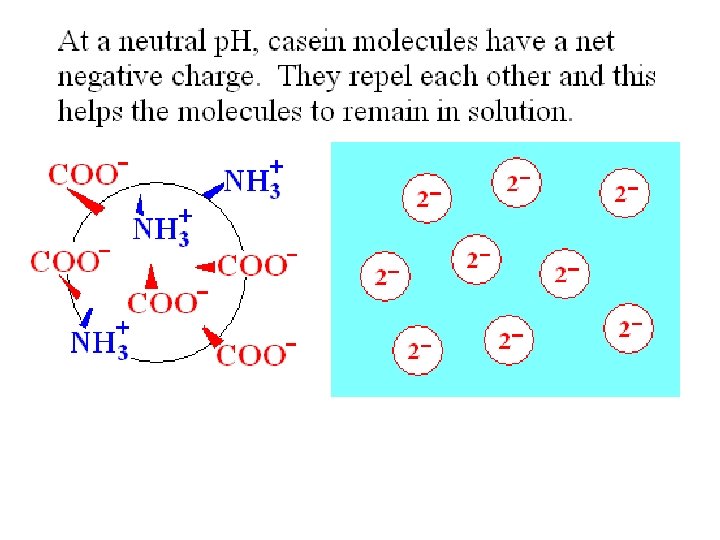
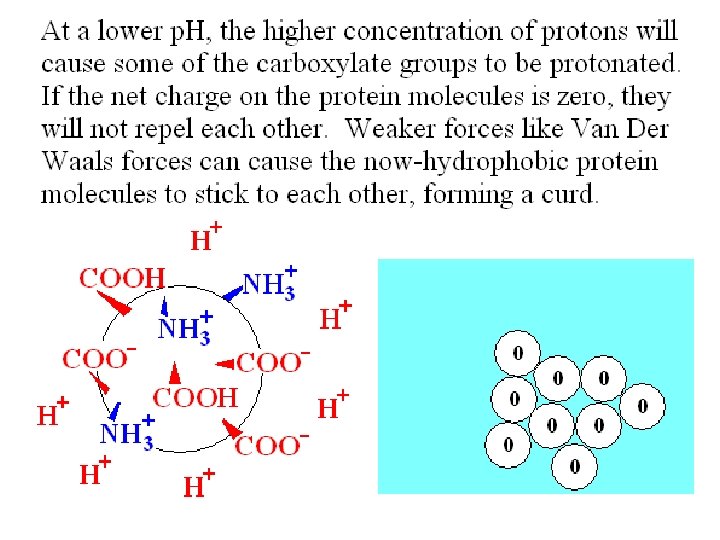

All casein proteins become insoluble at their isoelectric point, p. H = 4. 6. Adding acid, either by adding vinegar or by bacterial fermentation of lactose to make lactic acid, causes curdling when casein molecules clump together.

Yogurt

Lactic Acid Fermentation Lactose is a disaccharide sugar found in milk. Lactic acid bacteria can ferment lactose and convert it to Lactic Acid 100% of the sugar is converted to lactic acid No gas is produced (unusual for fermentation) Low p. H inhibits other microorganisms Creates a smooth sour taste Fermentation of Lactose C 12 H 22 O 11 + H 2 O 4 C 3 H 6 O 3 lactose + water lactic acid 342 g/mole + 18 g/mole 360 g/4 moles

Fermentations Fermentation is anaerobic metabolism where the terminal electron acceptor is organic compound such as pyruvate. EXAMPLES of Fermentation Products Organic Acids Lactic acid, Acetic acid, Formic acid, Propionic acid Alcohols Ethanol, Propanol, Butanol, 2, 3 -Butanediol Aldehydes and Ketones Acetone, Butyraldehyde Gases Hydrogen, Carbon Dioxide

Lactic Acid Bacteria Two types of bacteria are needed to make yogurt. - Streptococcus thermophilus - Lactobacillus bulgaricus We will use yogurt containing live active culture to inoculate the milk. Microbial Succession First, Streptococcus thermophilus (G+ cocci) ferments lactose to lactic acid. Acid inhibits growth of Streptococcus thermophilus when p. H goes below 5. 0. Second, Lactobacillus bulgaricus (G+ bacilli), because it can grow under more acidic conditions continues to produce lactic acid, lowering the p. H to ~ 4. 0. The acidic condition causes the milk protein casein to coagulate forming a semi- solid curd.

Yogurt Production Protocol - Scald the milk. (Heat to 80 o. C, do not boil. ) This kills contaminating bacteria and drives off oxygen. (Fermentation is an anaerobic process. ) - Cool the milk to 65 C. - Inoculate the milk with lactic acid bacteria (use yogurt) S. thermophilus tolerates higher temperature and grows fastest at neutral p. H. L. bugaricus prefers a lower temperature (40 - 45 C) but can tolerate a lower p. H. - Incubate at 45 C for 16 - 24 hours The amount of lactic acid affects flavor and consistency of the yogurt. The temperature and p. H must be closely monitored in food industry. Stop the fermentation by refrigeration when the product is perfect. Add fruit or flavoring if desired.

MCB 101 Lecture 11 4/19/19 What two species of bacteria are used in yogurt production? A. Escherichi coli & Staphylococcus aureus B. Salmonella typhi & Shigella dysenteriae C. Lactococcus lactis & Streptococcus mutans D. Proteus vulgaris & Morganella morganii E. Lactobacillus bulgaricus & Streptococcus thermophilus

What two species of bacteria are used in yogurt production? A. Escherichi coli & Staphylococcus aureus feces & boils B. Salmonella typhi & Shigella dysenteriae septicemia & bloody diarrhea C. Lactococcus lactis & Streptococcus mutans cheese & tooth decay D. Proteus vulgaris & Morganella morganii bladder infections & fecal bacteria E. Lactobacillus bulgaricus & Streptococcus thermophilus

MCB 101 Lecture 11 4/19/19 What is the incubation temperature for yogurt production? A. B. C. D. E. 0 o. C 20 o. C 37 o. C 45 o. C 63 o. C

MCB 101 Lecture 11 4/19/19 What is the incubation temperature for yogurt production? A. 0 o. C B. 20 o. C C. 37 o. C ice room temperature human body temperature D. 45 o. C E. 63 o. C pasteurization temperature

Cheese

In cheese production, curd formation is achieved using a combination of bacterial lactic acid fermentation plus rennet. Rennet is a proteolytic enzyme derived from the digestive tract of a calf that modifies casein protein molecules making them less soluble. This allows for curd formation at a slightly higher p. H than is typical for yogurt.
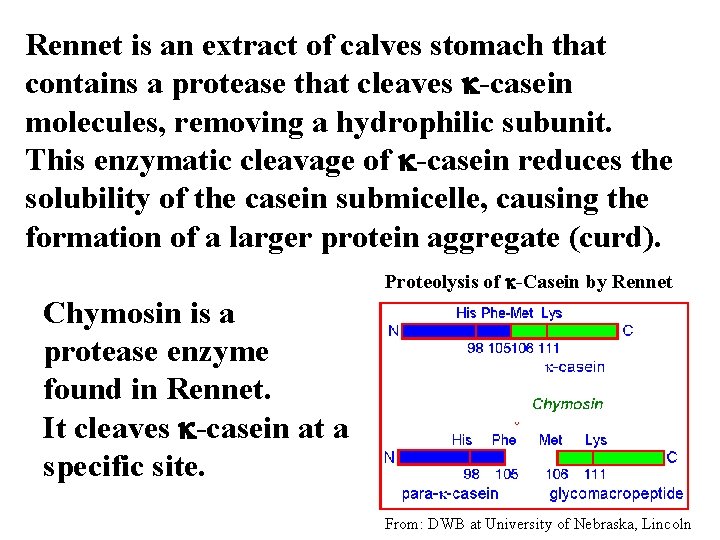
Rennet is an extract of calves stomach that contains a protease that cleaves k-casein molecules, removing a hydrophilic subunit. This enzymatic cleavage of k-casein reduces the solubility of the casein submicelle, causing the formation of a larger protein aggregate (curd). Proteolysis of k-Casein by Rennet Chymosin is a protease enzyme found in Rennet. It cleaves k-casein at a specific site. From: DWB at University of Nebraska, Lincoln
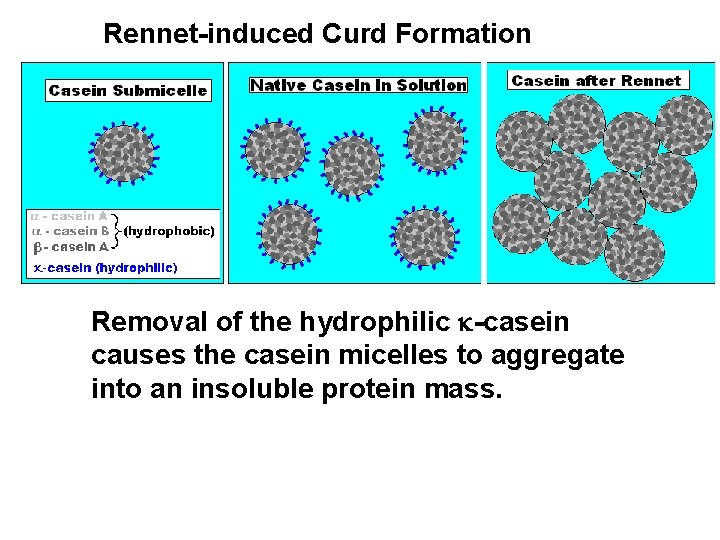
Rennet-induced Curd Formation Removal of the hydrophilic k-casein causes the casein micelles to aggregate into an insoluble protein mass.

The cheese curd is cut into smaller pieces and gently heated to drive out excess liquid (whey). Then the curds are drained and (usually) salted.

Right: Cheese curds are placed in a mold. Bottom left: Cheese curds in molds. Bottom right: Traditional wooden cheese press.

Experiment - 19 The Role of Bacteria in the Nitrogen Cycle - Nitrogen is essential to all living things. - Nitrogen is found in DNA, RNA, proteins, modified sugars and many enzyme cofactors. - 70% of the atmosphere is N 2 gas, but most organisms cannot utilize this form of nitrogen. - Some bacteria can reduce N 2 to ammonia and other forms that plants & animals can utilize. - Many bacteria can convert nitrogenous compounds from one form to another.
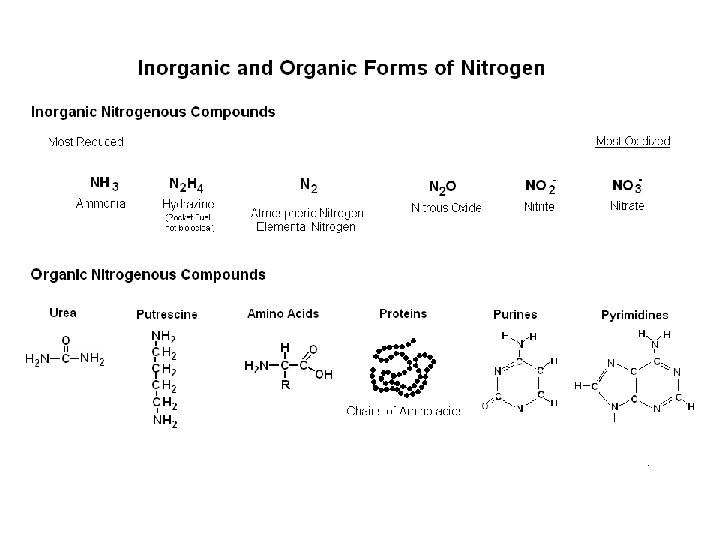

Nitrogen Cycle • Some bacteria that are found in soil can fix nitrogen. (that is: reduce N 2 to ammonia NH 3) • Some soil bacteria can oxidize NH 3 to nitrite (NO 2 -) or nitrate (NO 3 -), under aerobic conditions, while other bacteria can reduce nitrate or nitrite to N 2. • Plants absorb nitrate NO 3 - or ammonia NH 3 from the soil and use it to make amino acids etc. • Animals eat plants or meat to get nitrogen (protein). • Nitrogen is excreted in waste and returned to soil in the form of ammonia or urea. • When dead plants and animals decompose, organic nitrogen is broken down to ammonia and amines and released into soil.
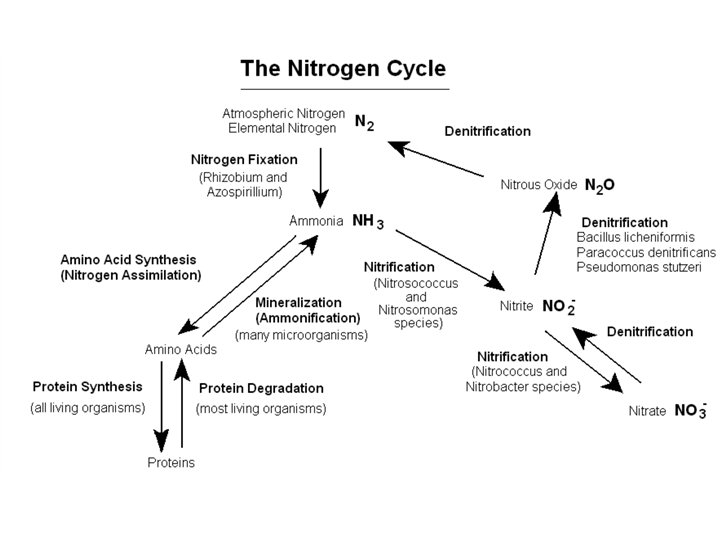

Basic Processes of the Nitrogen Cycle - Nitrogen fixation (reduction of N 2 to ammonia) - Nitrogen Assimilation (ammonia amino acids etc. ) - Mineralization (degradation of organic N – compounds) - Denitrification (nitrate reduction to N 2) - Nitrification (oxidation of ammonia to nitrate) Plants need a source of nitrogen to survive. Nitrogen Fixing Bacteria - Azotobacter and Bacillus are free-living soil bacteria that fix N 2 and release nitrogen components into the soil where it’s available to plants. - Rhizobium species are symbiotic within root nodules of legumes.

MCB 101 Lecture 11 April 19, 2019 4) Which one of these organic compounds found in cytoplasm does not contain nitrogen? A. DNA D. ATP B. Starch C. NAD+ E. Proteins

MCB 101 Lecture 11 April 19, 2019 4) Which one of these organic compounds found in cytoplasm does not contain nitrogen? A. DNA D. ATP B. Starch C. NAD+ E. Proteins

Nitrogen Fixation (no experiment will be done in the lab) Gaseous N 2 is reduced to ammonia (NH 3) 70% of the atmosphere is N 2. Only certain prokaryotes can fix nitrogen. N 2 fixation consumes a lot of energy and reducing power. The reaction is catalyzed by nitrogenase, which is an oxygen sensitive enzyme. (Aerobic nitrogen fixers must protect their nitrogenase from oxygen. ) N 2 + 4 -5 (NADPH + H+) + 12 – 16 ATP 2 NH 3 + 4 -5 NADP+ + 1 -2 H 2 + 12 – 16 (ADP + Pi) Ammonia can then be assimilated to make amino acids. a-keto-glutarate + NH 3 + NADPH + H+ glutamic acid + NADP + + H 2 O

Clover Root Nodules
Bacteroids

Nitrogen Assimilation (no experiment will be done in lab) • Incorporation of ammonia into organic compounds (amino acids) which can be used by bacteria as sole source of nitrogen • Key steps – Synthesis of glutamic acid (glutamate) – Glutamic acid is used to make other amino acids. – Transamination, the movement of an amino group from glutamic acid to pyruvate produces alanine. – Nitrogen rich compounds which can be made include: amino acids, purines and ureides

Mineralization (Experiment 19 a) • Mineralization is the degradation of organic compounds that contain nitrogen such as: DNA, RNA and proteins, to release inorganic nitrogenous compounds such as ammonia (NH 3). • Some Enzymes Involved in Mineralization of Nitrogen (conversion of organic nitrogen to inorganic forms) – Tryptophanase (Indole test) – Phenylalanine deaminase – Cysteine desulfhydrase (Kligler’s H 2 S) – Urease (Urea to Ammonia and CO 2)

Urease Test (Experiment 19 a) • Mammals excrete urea into soil • Plants use ammonia or nitrate but not urea • Some bacteria can break down urea to ammonia and CO 2 • Ammonia can be used by plants or converted to nitrate by aerobic bacteria The urease test distinguishes Proteus from other enteric bacteria. Proteus is urease positive, the broth turns pink Serratia is urease negative, it stays peachy

Ex. 19 b Denitrification - Nitrate Reduction • The reduction of nitrate to nitrite is a form of anaerobic respiration. NADH + H+ + NO 3 - NAD+ + H 2 O + NO 2 - • Nitrogen can be further reduced from nitrite to nitrous oxide (N 2 O) or nitrogen gas (N 2). • Denitrification occurs in the absence of oxygen (O 2) (It’s anaerobic respiration. ) • Nitrogen is removed from soil and lost to the atmosphere in form of gas

Ex. 19 b Nitrate Reductase Test • • Purpose: check for denitrification Inoculate: Nitrate broth and PBE Look for anaerobic growth Look for gas production – nitrous oxide or nitrogen gas • Look for reduction of nitrate to nitrite (or beyond) Media Used for the Nitrate Reductase Test PBE Broth - Peptone - Beef extract Nitrate Broth - PBE Broth + Nitrate

Bacterial Growth in Medium • Proteus = fermentative metabolism = anaerobic • Pseudomonas = oxidative = respiration (Remember the OF-glucose test) • Proteus can grow fermentatively so should be ok in PBE without nitrate and without oxygen. • Pseudomonas should grow poorly in PBE anaerobically since it’s oxidative and has no nitrate to subsitute for oxygen as a terminal electron acceptor • Both are able to reduce nitrate and therefore grow better in PBE + Nitrate than in PBE alone

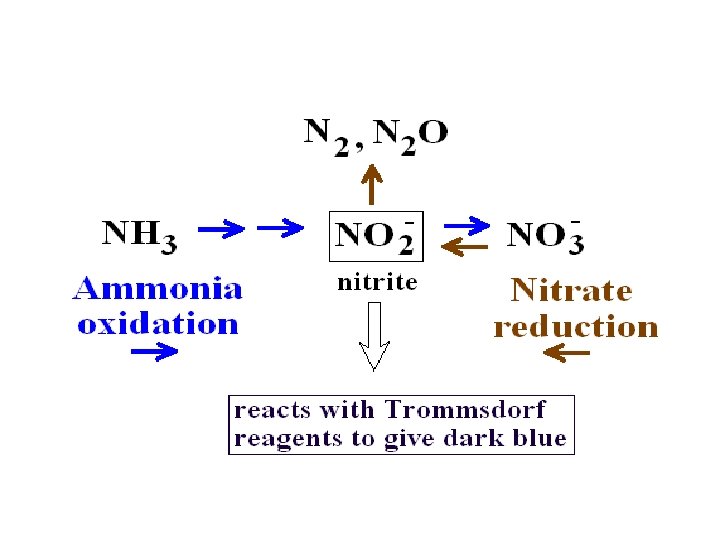
The Chemistry of Trommsdorf Reagents Trommsdorf reagent II is 6 M sulfuric acid. It reacts with nitrite to produce nitrous acid. Nitrous acid can oxidize the iodide in Trommsdorf I to iodine. The iodine can form a blue colored complex with the starch, which is also in Trommsdorf I. Nitrate (NO 3 -) can react with Zinc metal (Zn 0). The zinc is oxidized to Zn+ while nitrate is reduced to nitrite (NO 2 -). Trommsdorf I Zinc Chloride Starch Zinc Iodide 20 g/l 4 g/l 2 g/l Trommsdorf II 6 M H 2 SO 4

Test for Reduction of Nitrate • Add Trommsdorf reagents, I and II (We will use these same reagents for Ex 19 c, Nitrification, but the production of a blue color means something different. ) – The immediate appearance of an inky blue color indicates presence of NITRITE (NO 2 -) – No color change could mean one of two possibilities, either: • No nitrate reduction took place or • Nitrate reduction took place, but the nitrite was further reduced to nitrogen gas (N 2) or nitrous oxide (N 2 O).

Test for Reduction of Nitrate (continued, if no immediate blue color is seen) • Add Zinc metal (Zn 0) • If no nitrate reduction took place, zinc will react with nitrate reducing it to nitrite and the nitrite will produce a blue color because of reaction with the Trommsdorf reagents. This indicates the culture is indeed negative for nitrate reduction. • If no blue color appears after the addition of zinc, it means that the nitrate was reduced beyond nitrite to nitrous oxide or nitrogen so the nitrate is well and truly gone. This indicates the culture is positive for nitrate reduction.

Nitrate Reductase Summary • Step 1 tests for presence of NITRITE – Nitrite + Trommsdorf blue color an immediate positive result shows nitrate was reduced to nitrite, but the nitrite was not reduced any further • Step 2 tests for presence of NITRATE via reduction to nitrite. – Nitrate + zinc nitrite – Nitrite + Trommsdorf blue color A blue color here shows that the nitrate was still present in the medium, that is to say the bacteria are negative for nitrate reduction. If there is no blue color when you add zinc, it indicates a positive result for reduction of nitrate and for the reduction of nitrite to N 2 or N 2 O gas.

Nitrification (ammonia oxidation, Expt. 19 c) • Oxidation of ammonia (NH 3) to nitrate (NO 3 -) by soil bacteria • Aerobic, 2 step process – 1 st Nitroso- bacteria oxidize ammonia to nitrite (NO 2 -) – 2 nd Nitro- bacteria oxidize nitrite to nitrate (NO 3 -) • Nitrate can be used by plants

Test for Nitrification (Experiment 19 c) • Inoculate Ammonia broth with soil • Incubate for 7 to 9 days at Room Temperature – Aerobic (with shaking/rotation) – Anaerobic (+ mineral oil, static) – Uninoculated Ammonia oxidizing bacteria need oxygen to convert the ammonia to nitrite or nitrate. After incubation we test for nitrite and/or nitrate using Trommsdorf reagents.

Scoring of Ammonia Oxidation Blue after Trommsdorf addition – ammonia was oxidized to nitrite – positive for ammonia oxidation Blue after zinc – nitrite was further oxidized to nitrate by nitro-bacteria – still positive for ammonia oxidation Colorless after Trommsdorf and zinc – negative for ammonia oxidation – No nitrite or nitrate present, still ammonia • Remember, this is an aerobic process, so we do not expect the anaerobic tube to be positive. Also note that ammonia does not react with Trommsdorf reagents.
- Slides: 52