MCB 100 Introductory Microbiology September 5 2018 Flavin

MCB 100 Introductory Microbiology September 5, 2018 Flavin Adenine Dinucleotide is an enzyme cofactor that acts as an electron carrier. It is found in almost all living organisms. Image From: wikipedia/commons/3/36/FAD. png
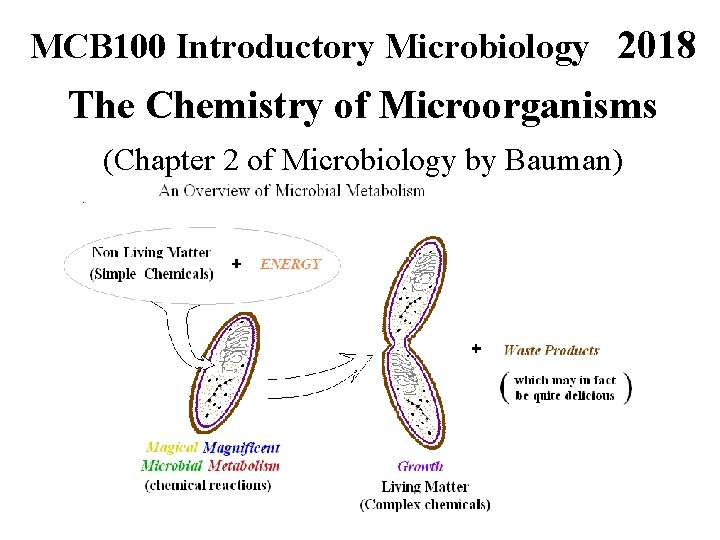
MCB 100 Introductory Microbiology 2018 The Chemistry of Microorganisms (Chapter 2 of Microbiology by Bauman)

Some Important Chemistry Terms Mixture: a substance (or sample) that can be broken down into different parts by physical manipulations that don’t necessarily involve chemical changes Compound: a pure substance that can be broken down into different parts only by chemical reactions Element: a pure substance that can’t be broken down into different parts by chemical reactions Molecule: the smallest particle possible for a compound Atom: the smallest particle possible for an element

What is an Organic Compound? An Organic Compound, to a chemist, is a chemical substance that is composed of at least 2 carbon atoms that are covalently bonded and at least 1 hydrogen atom. Organic = from an organism Methane, CH 4, isn’t considered to be organic because it has only 1 carbon atom and can be made by volcanoes, which are definitely not living creatures. But, methane can be made by living creatures, and organic chemists often use methane derivatives as examples when teaching concepts in organic chemistry. Urea (NH 2 -CO-NH 2) is considered to be organic because it is found in urine, which comes from animals. Also, in 1828, Friedrich Wohler showed that urea could be synthesized from inorganic substances. This was an important historical event in the development of chemistry as a science.

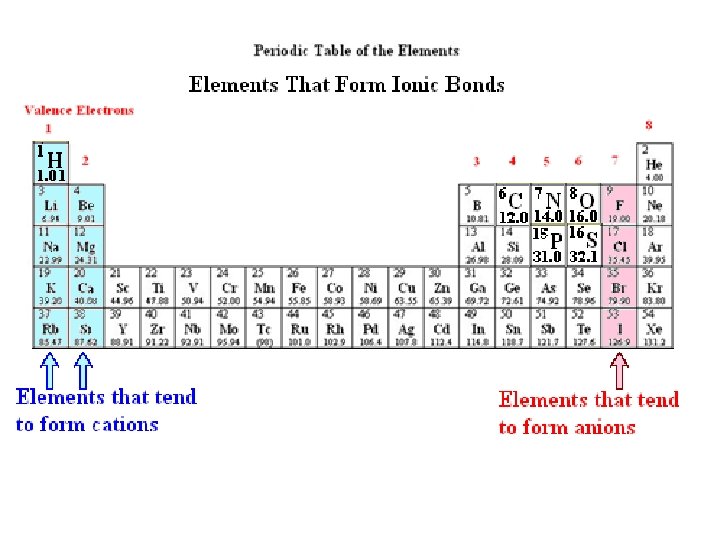
Elements That Are Common In Living Matter Choose the statement that is TRUE. A. Ca is the symbol for carbon. B. Sodium and Chlorine atoms form chemical associations with other elements primarily through covalent bonds. C. An oxygen atom can normally form 4 covalent bonds. D. The chemical symbol for sodium is S. E. Nitrogen atoms, when paired with hydrogen, carbon or other nitrogen atoms, normally form 3 covalent bonds.

Elements That Are Common In Living Matter Choose the statement that is TRUE. A. Ca is the symbol for carbon. False - Ca is Calcium, C is Carbon B. Sodium and Chlorine atoms form chemical associations with other elements primarily through covalent bonds. False – Na. Cl is an ionic compound C. An oxygen atom can normally form 4 covalent bonds. F 2 bonds D. The chemical symbol for sodium is S. F Sodium is Na, S is Sulfur E. Nitrogen atoms, when paired with hydrogen, carbon or other nitrogen atoms, normally form 3 covalent bonds. True - NH 3 H 3 C-NH 2 N=N
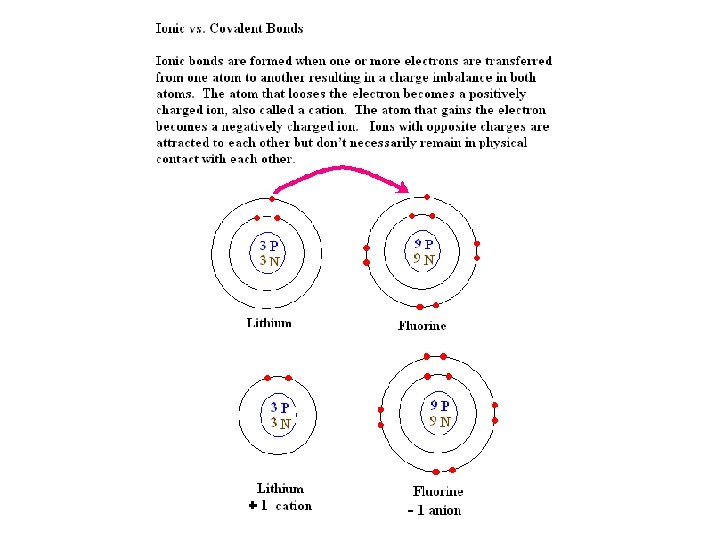

REDOX REACTIONS An Oxidation-Reduction reaction involves a transfer of electrons from one atom (or group of atoms) to another atom (or group). An oxidation is a loss of electrons. A reduction is a gain of electrons. Because: electrons have a negative charge. 2 Na + Cl 2 2 Na. Cl (Na+, Cl-) The sodium atoms go from a redox state of 0 to +1 when they lose an electron, while the chlorine atoms go from a redox state of 0 (in Cl 2) to -1 when they gain an electron. The sodium is the reducing reagent because it supplies the electron that reduces the charge of the chlorine atom. The chlorine is the oxidizing reagent because it accepts (or takes) the electron from the sodium atom, thus causing the sodium to become a positively charged ion.
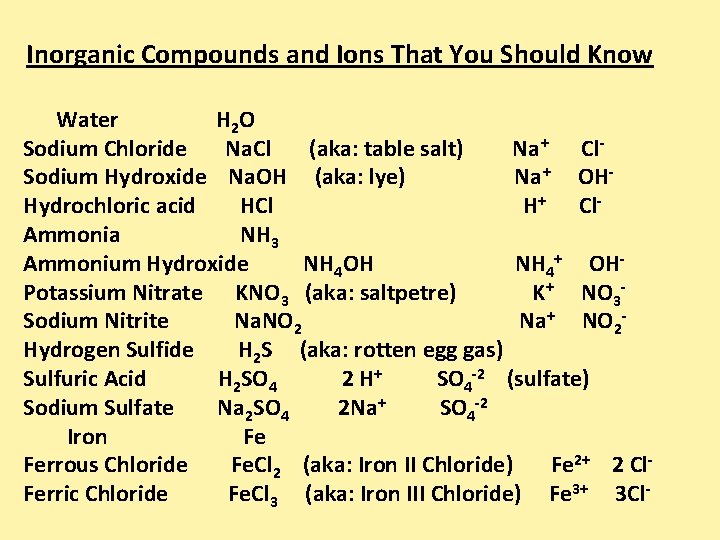
Inorganic Compounds and Ions That You Should Know Water H 2 O Sodium Chloride Na. Cl (aka: table salt) Na+ Cl. Sodium Hydroxide Na. OH (aka: lye) Na+ OHHydrochloric acid HCl H+ Cl. Ammonia NH 3 Ammonium Hydroxide NH 4 OH NH 4+ OHPotassium Nitrate KNO 3 (aka: saltpetre) K+ NO 3 Sodium Nitrite Na. NO 2 Na+ NO 2 Hydrogen Sulfide H 2 S (aka: rotten egg gas) Sulfuric Acid H 2 SO 4 2 H+ SO 4 -2 (sulfate) Sodium Sulfate Na 2 SO 4 2 Na+ SO 4 -2 Iron Fe Ferrous Chloride Fe. Cl 2 (aka: Iron II Chloride) Fe 2+ 2 Cl. Ferric Chloride Fe. Cl 3 (aka: Iron III Chloride) Fe 3+ 3 Cl-
- Slides: 10