MCB 100 Introductory Microbiology January 25 2019 Chemistry
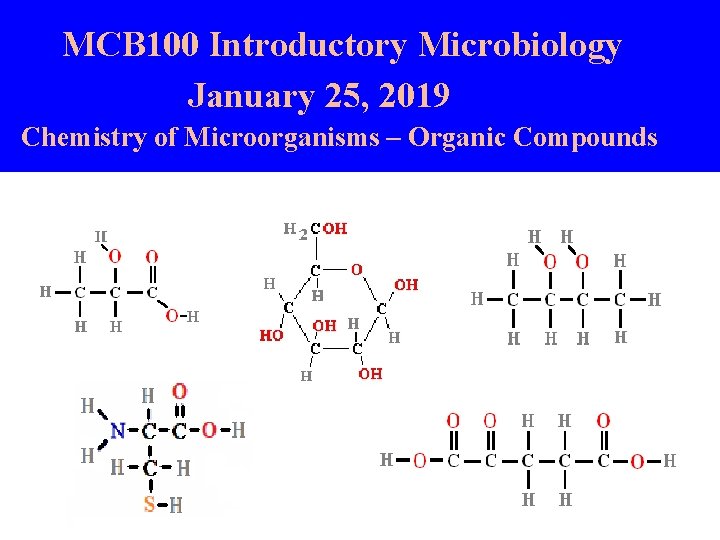
MCB 100 Introductory Microbiology January 25, 2019 Chemistry of Microorganisms – Organic Compounds
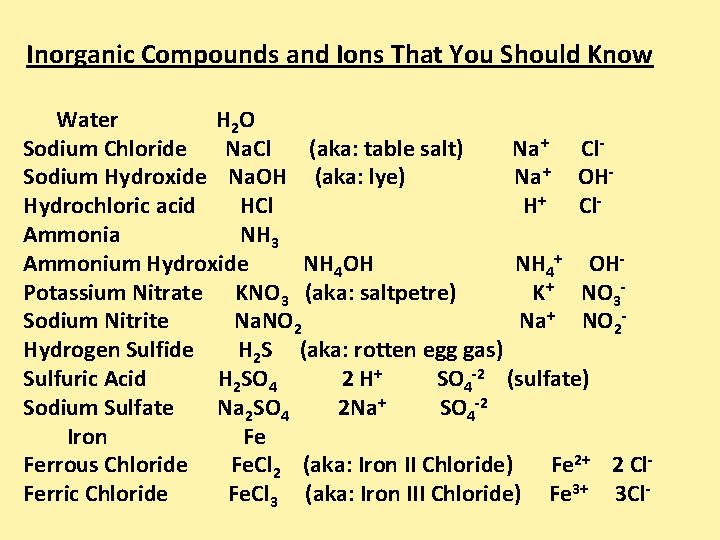
Inorganic Compounds and Ions That You Should Know Water H 2 O Sodium Chloride Na. Cl (aka: table salt) Na+ Cl. Sodium Hydroxide Na. OH (aka: lye) Na+ OHHydrochloric acid HCl H+ Cl. Ammonia NH 3 Ammonium Hydroxide NH 4 OH NH 4+ OHPotassium Nitrate KNO 3 (aka: saltpetre) K+ NO 3 Sodium Nitrite Na. NO 2 Na+ NO 2 Hydrogen Sulfide H 2 S (aka: rotten egg gas) Sulfuric Acid H 2 SO 4 2 H+ SO 4 -2 (sulfate) Sodium Sulfate Na 2 SO 4 2 Na+ SO 4 -2 Iron Fe Ferrous Chloride Fe. Cl 2 (aka: Iron II Chloride) Fe 2+ 2 Cl. Ferric Chloride Fe. Cl 3 (aka: Iron III Chloride) Fe 3+ 3 Cl-

Properties of Living Matter - Living matter contains carbon. The atoms in an organic molecule are held together by covalent bonds. Living organisms are mostly made of about 20 different elements. These six elements make up most of the mass of organic compounds: Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorous, Sulfur C H O N P S - These six elements can form covalent bonds. - The proteins, nucleic acids, lipids and carbohydrates that form living matter are large complicated molecules. (AKA: Biological Macromolecules) - There are several thousand different types of proteins found in a typical cell. - Biological macromolecules stick to each other in very specific ways due to noncovalent attractions. This produces a higher level of order. Enzyme subunits can stick to each other and work together. Regulatory proteins can bind to specific DNA sequences. Ribosomal subunits stick together. - However, a cell is more than a mixture of molecules, it is an organized system.
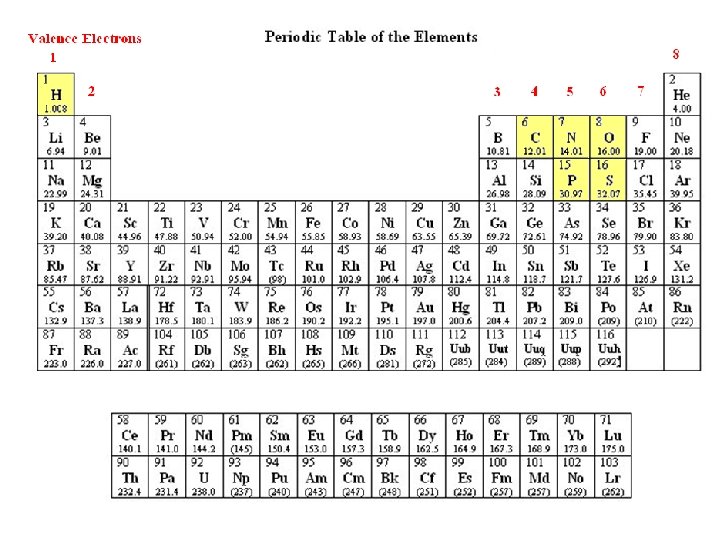

Twelve elements that are common in living matter Element Symbol Carbon C Hydrogen H Oxygen O Nitrogen N Phosphorous P Sulfur S Chlorine Cl Sodium Na Potassium K Calcium Ca Magnesium Mg Iron Fe Valence Electrons 4 1 6 5 5 6 7 1 1 2 2 2 Bond Number 4 1 2 3 3 or 5 2 or 6 1 1 1 2 2 2 or 3 Bond Type covalent (ionic, cation) covalent ionic, anion ionic, cation ionic, cation

Compounds made of atoms that are held together by ionic bonds tend to be simple. Compound Sodium Chloride Calcium Bromide Potassium Iodide Formula Na. Cl Ca. Br 2 KI # of atoms 2 3 2 F. W. 58. 5 199. 9 166. 0 Compounds made of atoms that are held together by covalent bonds can be very large and complex. Compound Formula # of atoms Ethyl Alcohol C 2 H 6 O 9 Glucose C 6 H 12 O 6 24 Lactose C 12 H 22 O 11 45 Alanine C 3 H 7 O 2 N 13 Tryptophan C 11 H 12 O 2 N 2 27 typical protein 300 amino acids 5000 ATP C 10 H 14 O 13 N 5 P 3 45 small plasmid 5000 nucleotide base pairs human chromosome 100 million base pairs F. W. 46 180 342 89 204 40, 000 505 3, 300, 000 66, 000, 000
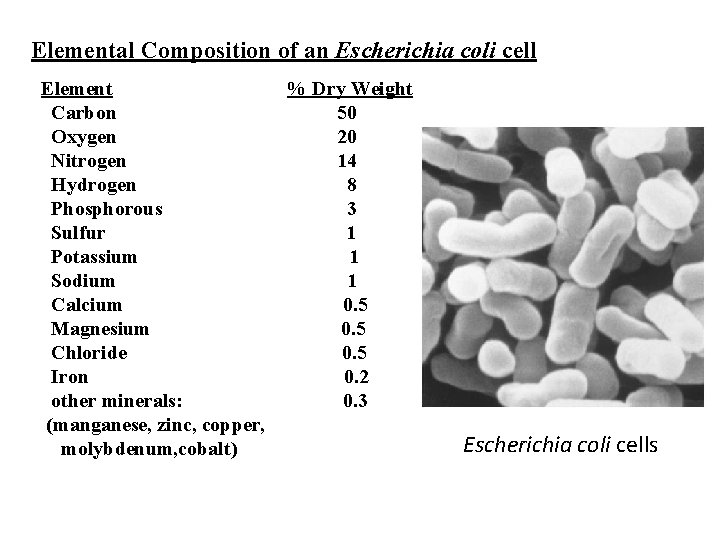
Elemental Composition of an Escherichia coli cell Element Carbon Oxygen Nitrogen Hydrogen Phosphorous Sulfur Potassium Sodium Calcium Magnesium Chloride Iron other minerals: (manganese, zinc, copper, molybdenum, cobalt) % Dry Weight 50 20 14 8 3 1 1 1 0. 5 0. 2 0. 3 Escherichia coli cells

The Basis of Organic Functional Groups
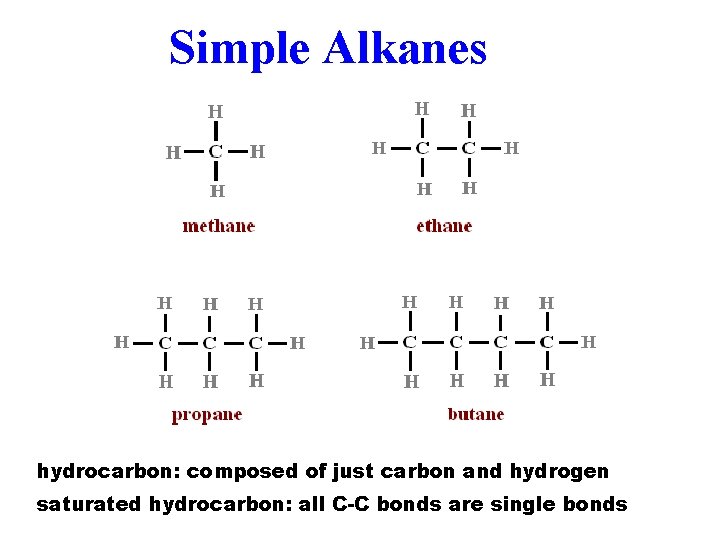
Simple Alkanes hydrocarbon: composed of just carbon and hydrogen saturated hydrocarbon: all C-C bonds are single bonds
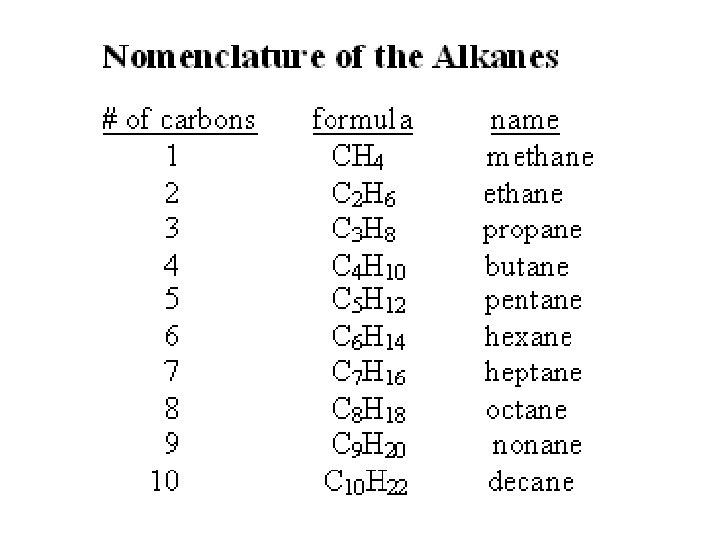
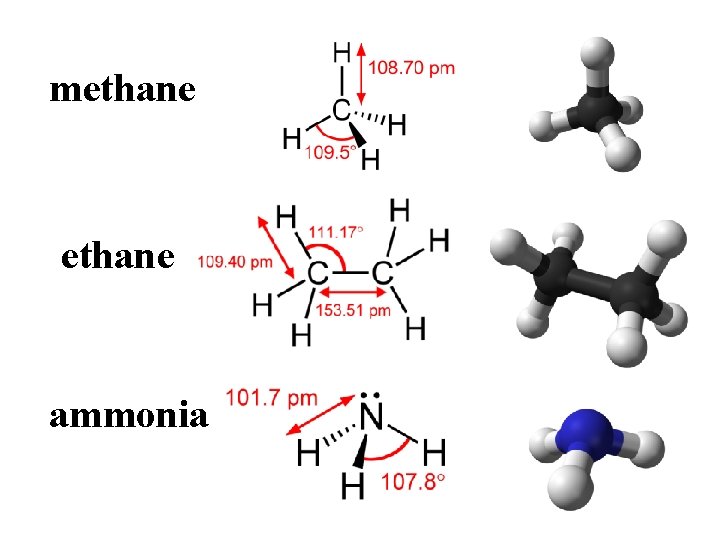
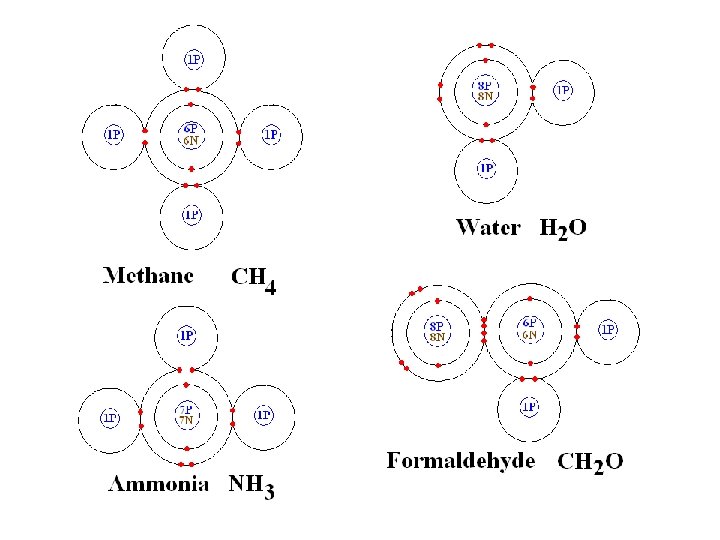
methane ammonia
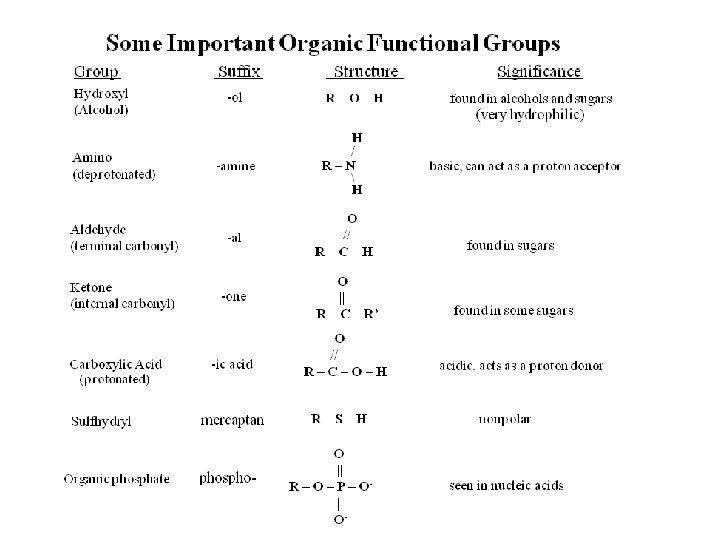
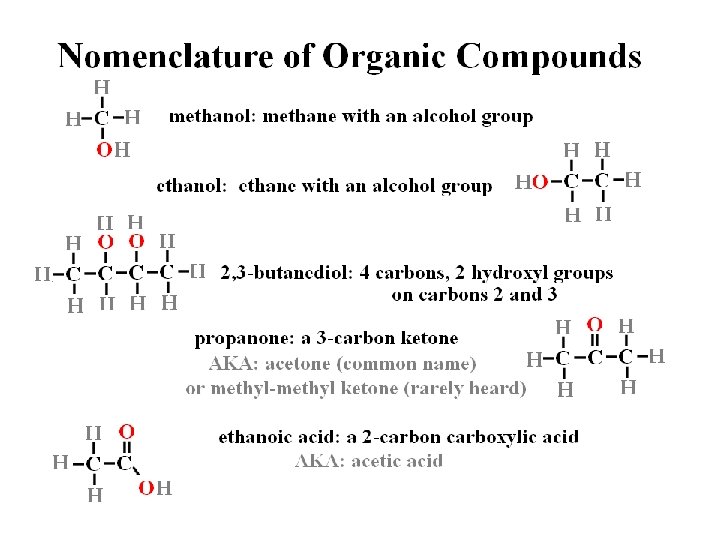
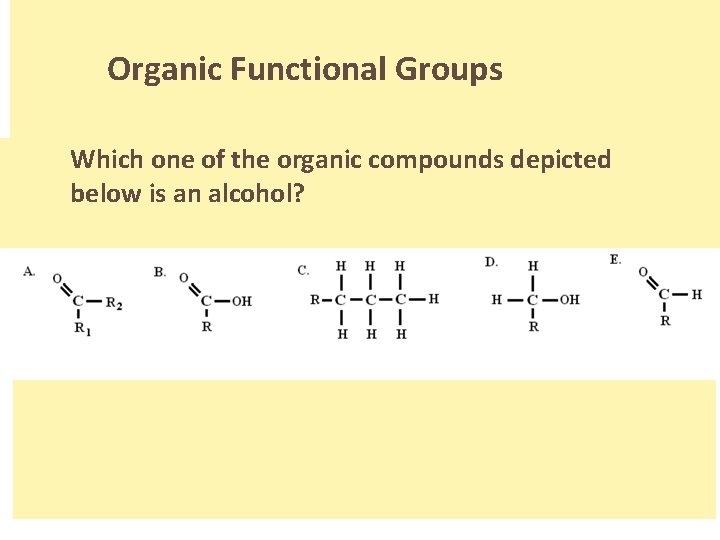
Organic Functional Groups Which one of the organic compounds depicted below is an alcohol? . .
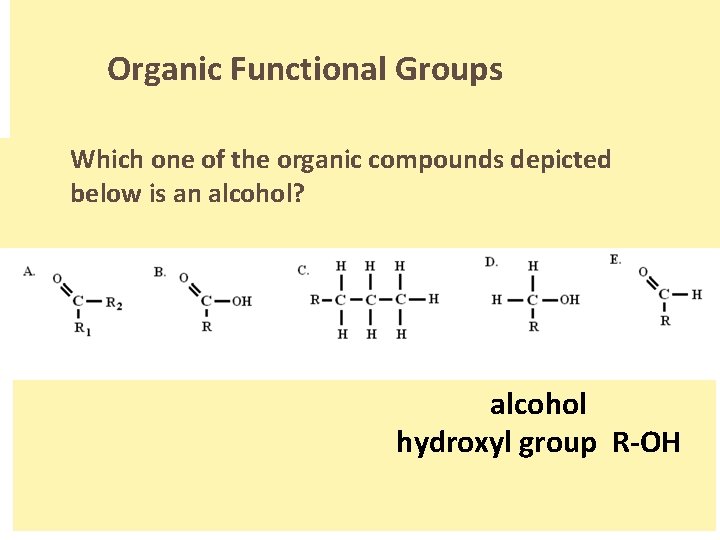
Organic Functional Groups Which one of the organic compounds depicted below is an alcohol? alcohol hydroxyl group R-OH.
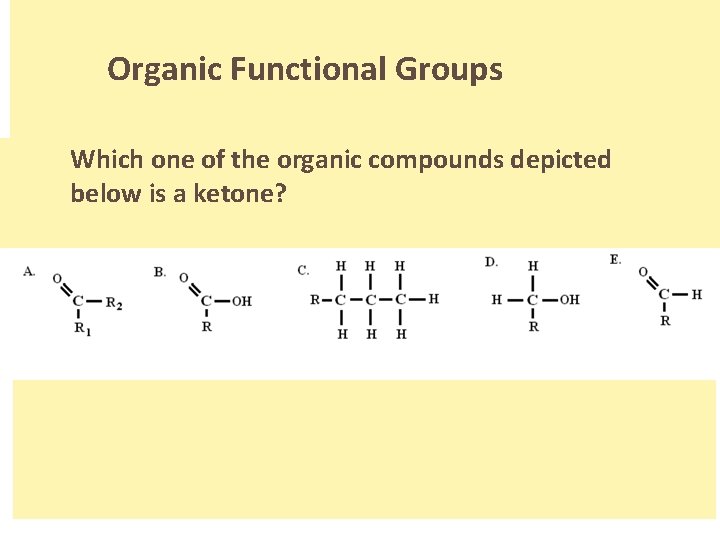
Organic Functional Groups Which one of the organic compounds depicted below is a ketone? . .
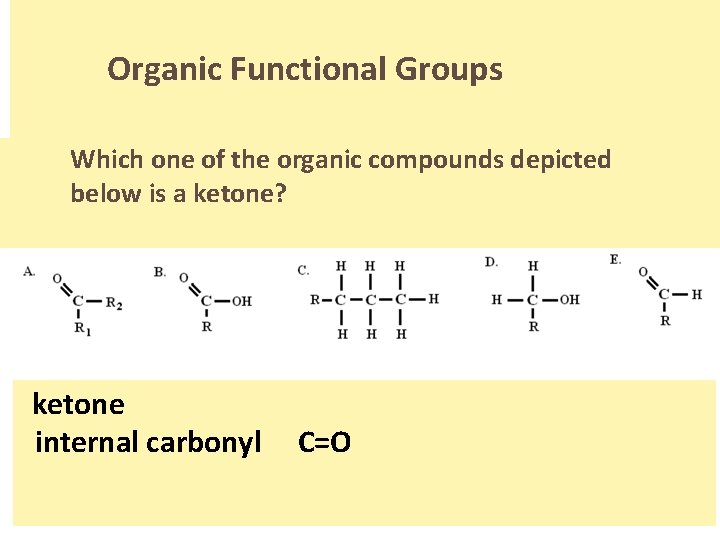
Organic Functional Groups Which one of the organic compounds depicted below is a ketone? . ketone internal carbonyl C=O.
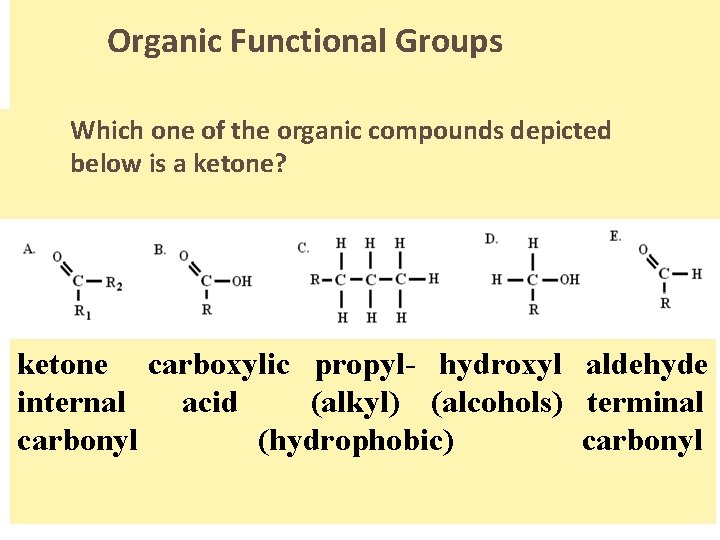
Organic Functional Groups Which one of the organic compounds depicted below is a ketone? ketone carboxylic propyl- hydroxyl aldehyde internal acid (alkyl) (alcohols) terminal carbonyl (hydrophobic) carbonyl.

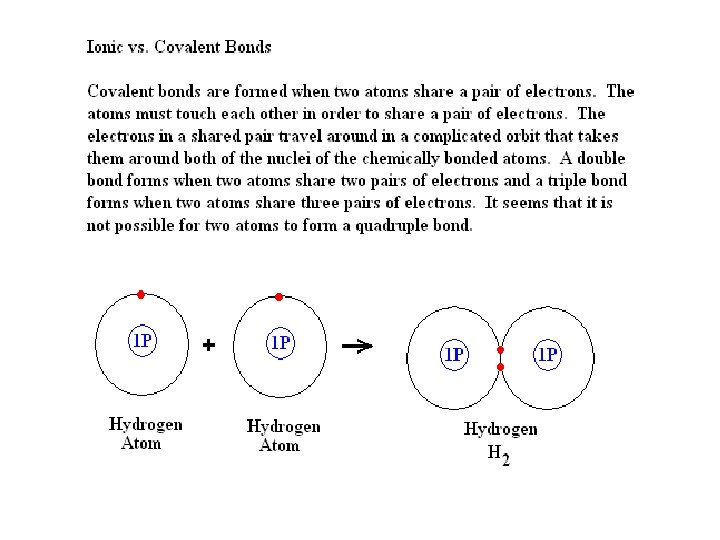
Polar Covalent Bonds vs. Non-polar Covalent Bonds A non-polar covalent bond forms when two atoms share a pair of electrons equally. Both atoms end up with no net charge. If two atoms share a pair of electrons unequally, one ends up with a slight negative charge while the other has a slight positive charge. This is a polar bond. The factor that determines if a pair of electrons will be shared equally or unequally is the electronegativity of the atoms that participate in the chemical bond. Electronegativity is a measure of the affinity of an atom for valence electrons.
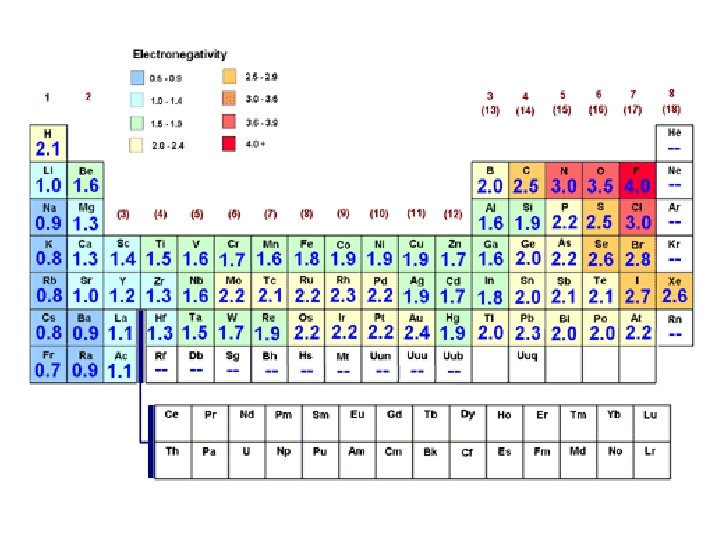

Electronegativity of The Most Common Elements Found in Biological Molecules H 2. 1 C 2. 5 N 3. 0 Examples: Non-Polar Covalent Bonds H–H H / H–C–H H O 3. 5 P 2. 1 S 2. 5 Polar Covalent Bonds H–O H H / H–N H
- Slides: 24