MC006247 Rev 0 Sponsored by Teleflex Optimizing Efficiency

MC-006247 Rev 0 Sponsored by Teleflex Optimizing Efficiency in TAVR Perspectives from Program Directors Rhythm Management Strategies Paul D. Mahoney, MD, FACC, FSCAI Director, Sentara Heart Structural Heart Programs Sentara Norfolk General Hospital Norfolk, Virginia

MC-006247 Rev 0 Paul Mahoney, MD, FACC Consultant: Edwards Medtronic Teleflex Proctor: Edwards Medtronic Boston Scientific Research Support: Edwards Medtronic Boston Scientific Abbott

Management of Conduction Disturbances Post. TAVR MC-006247 Rev 0 • New conduction system abnormalities after TAVR are common and can be associated with increased morbidity, cost, and length of stay (LOS) • Ideal strategy for rhythm management has not been identified • Recent ACC/AHA Consensus document offered less than definitive guidance • As TAVR volume increases and procedural efficiencies are emphasized, what strategies can be used for management of new conduction disturbances post TAVR?

ACC/AHA Consensus Document MC-006247 Rev 0 • “This expert report document represents an initial effort to provide a comprehensive and structured guide for managing patients with conduction disturbances post-TAVR and a framework for future research…” • “…It should be considered a work in progress and the recommendations regarded as suggestions based on current evidence and consensus opinion of a group of experts in the field. ” Rodés-Cabau JACC, Volume 74, Issue 8, August 2019
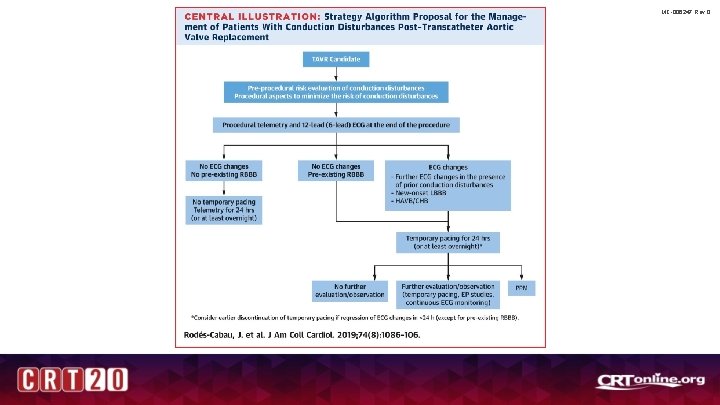
MC-006247 Rev 0

MC-006247 Rev 0 OMG 2019 American College of Cardiology Foundation

Define the Problem: Post TAVR Conduction MC-006247 Rev 0 • Intraprocedural Complete Heart Block (CHB) that is transient – “Transient”: resolves within an hour of the procedure and does not recur • Intraprocedural CHB that persists • Late CHB: more than an hour after TAVR • New BBB – Persistent vs transient – Width of the QRS • Bradycardia is not TAVR related

Procedural and Patient Specific Risk Factors for PPM After TAVR MC-006247 Rev 0 • Patient Risk Factors for PPM – Pre-existing RBBB – Pre-existing 1 st degree AV block – Age • Valve deployment matters – High implant associated with lower PPM rate (esp. with Evolut™) – Oversizing associated with Higher PPM rate – Evolut™ vs SAPIEN™ (can be mitigated) Rodés-Cabau JACC, Volume 74, Issue 8, August 2019
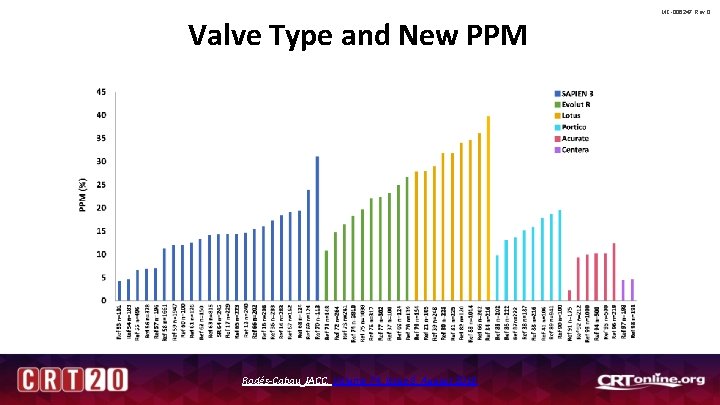
Valve Type and New PPM Rodés-Cabau JACC, Volume 74, Issue 8, August 2019 MC-006247 Rev 0
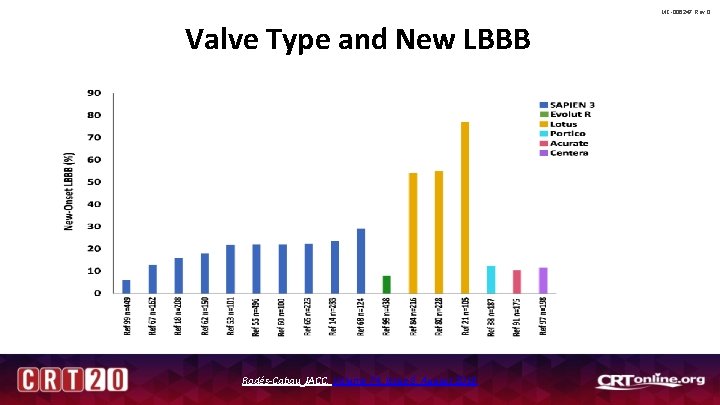
MC-006247 Rev 0 Valve Type and New LBBB Rodés-Cabau JACC, Volume 74, Issue 8, August 2019

Sentara TAVR Program: Coastal Virginia • 2000 TAVR • 450 TAVR 2019 – 210 SAPIEN™ – 225 Evolut™ – 15 LOTUS Edge™/Portico™/ACURATE neo ™ • 99% Conscious Sedation • 80% next day discharge • 5 TAVR/day; one team • 2019 Pacemaker Rate: 4. 6% – SAPIEN™: 4. 2% – Evolut™: 5. 1% • 2018 Pacemaker Rate: 7% • 2017 Pacemaker Rate: 8. 5% • 2016 Pacemker Rate: 11% MC-006247 Rev 0

MC-006247 Rev 0 Developing Strategy • Programmatic effort for procedural efficiency • Unified approach for rhythm management • Met with EP leadership and developed protocolized strategy; avoid “re-inventing the wheel” on case by case basis

Develop a Protocol Driven, Institution Specific Approach with EP • Intra-procedural • Post-Procedural • Post-Discharge MC-006247 Rev 0

Intraprocedural • Which temp wire to start? – Non-RBBB: any temp wire – RBBB: Active fixation lead exteriorized to pacer box via 7 Fr. Sheath; plan to leave in overnight MC-006247 Rev 0

MC-006247 Rev 0 Active Fixation Lead Regular Temp Wire IS-1 Adapter Images courtesy of Paul Mahoney, MD Standard Pacemaker box
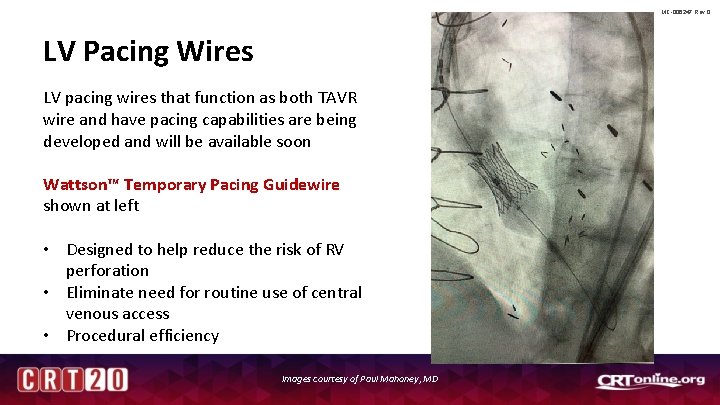
MC-006247 Rev 0 LV Pacing Wires LV pacing wires that function as both TAVR wire and have pacing capabilities are being developed and will be available soon Wattson™ Temporary Pacing Guidewire shown at left • Designed to help reduce the risk of RV perforation • Eliminate need for routine use of central venous access • Procedural efficiency Images courtesy of Paul Mahoney, MD

MC-006247 Rev 0 Intraprocedural Conduction Disturbance • Non-RBBB – No change in rhythm: Pull temp wire – Change in rhythm: Screw in lead from neck – No one gets PPM day of procedure – 7 am POD #1: Decision time
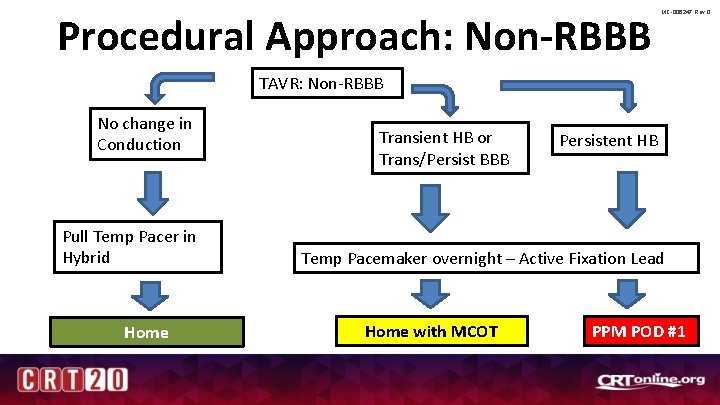
Procedural Approach: Non-RBBB MC-006247 Rev 0 TAVR: Non-RBBB No change in Conduction Pull Temp Pacer in Hybrid Home Transient HB or Trans/Persist BBB Persistent HB Temp Pacemaker overnight – Active Fixation Lead Home with MCOT PPM POD #1
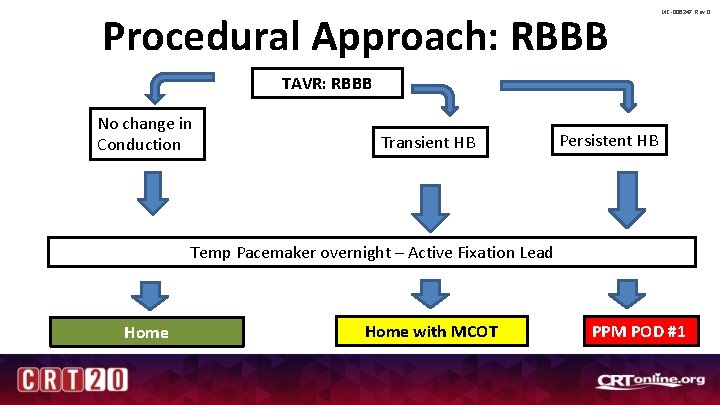
Procedural Approach: RBBB MC-006247 Rev 0 TAVR: RBBB No change in Conduction Transient HB Persistent HB Temp Pacemaker overnight – Active Fixation Lead Home with MCOT PPM POD #1

Post Procedure: Protocolize When EP Gets Involved • Involve EP when: – Pacemaker needed (CHB post implant) – Very Wide QRS (> 160 msec) • Call on day of procedure, for possible pacer POD #1, to reserve time in EP lab MC-006247 Rev 0

MC-006247 Rev 0 Protocolize When EP Gets Involved • Do not call EP for: – Bradycardia – Afib – BBB with QRS < 160 msec – Because you want a monitor

MC-006247 Rev 0 Post-Discharge • 14 -28 day continuous Telemetry • Communicate with patients’ role and reason

MC-006247 Rev 0 Caveats • There is a consensus document; be aware when you deviate from it • Create a written protocol • Track your outcomes

MC-006247 Rev 0 Sentara Experience • 4. 6% PPM rate all comers last 500 TAVR • 20% of patients home with MCOT • 7% of MCOT patients require PPM – None in first 24 hours – CHB Day 2 to Day 16 of monitoring – No deaths (couple near misses, all > 5 days)

MC-006247 Rev 0 The statements herein reflect the personal experience and opinion of the speaker. This information is provided for clinical education purposes and is not intended to be a substitute for sound clinical judgment or decision making, or professional experience relative to diagnostic and treatment options of a specific patient's medical condition. Please see the instructions for use for complete product information. CAUTION: Federal Law (U. S. A. ) restricts this device to sale by or on the order of a physician. Results from case studies are not predictive of results in other cases. Results in other cases may vary. SAPIEN is a trademark of Edwards Lifesciences Corporation. Evolut is a trademark of a Medtronic company. LOTUS Edge and ACURATE neo are trademarks of Boston Scientific Corporation or its affiliates. Portico is a trademark of an Abbott group of companies. Teleflex, the Teleflex logo, and Wattson are trademarks or registered trademarks of Teleflex Incorporated or its affiliates, in the U. S. and/or other countries. All other trademarks and registered trademarks are property of their respective owners. Information in this material is not a substitute for the product Instructions for Use. Not all products may be available in all countries. Please contact your local representative. For more information, please visit teleflex. com. Revised: 02/2020. © 2020 Teleflex Incorporated. All rights reserved. MC-006247 Rev 0
- Slides: 25