Maximum work or available energy from a system

Maximum work or available energy from a system w

Maximum work or available energy res Control mass process when the system receives heat from a source cm w 12 sur Measure of irreversibility From 1 st law for the CM we get:

Example problem: 100 C NH 3 1 kg of NH 3 is contained in a spring loaded piston/cylinder as saturated liquid at -20 C. Heat is added from a reservoir at 100 C until a final state of 800 kpa, 70 C is reached. Find the work, heat transfer, and entropy generation assuming the process to be internally reversible. p 1=pressure(r 717, t=-20, x=0) v 1=volume(r 717, t=-20, x=0) u 1=intenergy(r 717, t=-20, x=0) s 1=entropy(r 717, t=-20, x=0) v 2=volume(r 717, p=800, t=70) u 2=intenergy(r 717, t=70, p=800) s 2=entropy(r 717, t=70, p=800) w 12=(800+p 1)*. 5*(v 2 -v 1) q 12=w 12+u 2 -u 1 s_gen+q 12/373=s 2 -s 1

Maximum work in a flow process q 1 w 2 Objective: to find the maximum work for the Case shown in picture 1 st Law for the CV: (1) 2 nd Law for the CV (2) (3) Now use eqn (3) in (1) to obtain max work as:
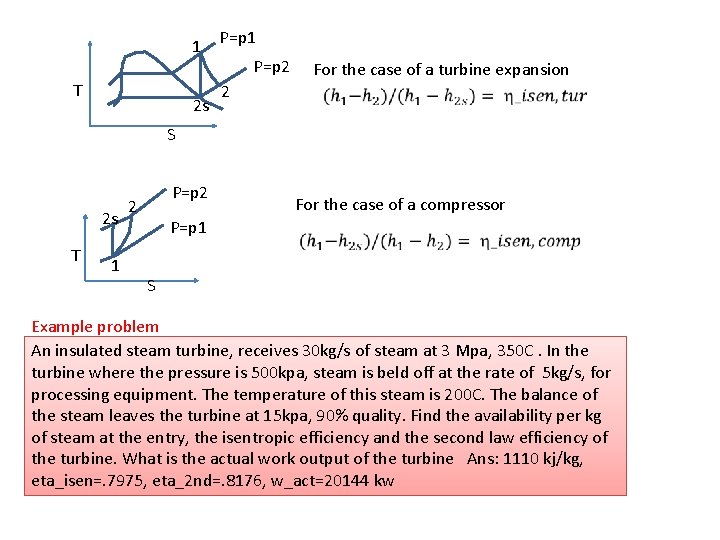
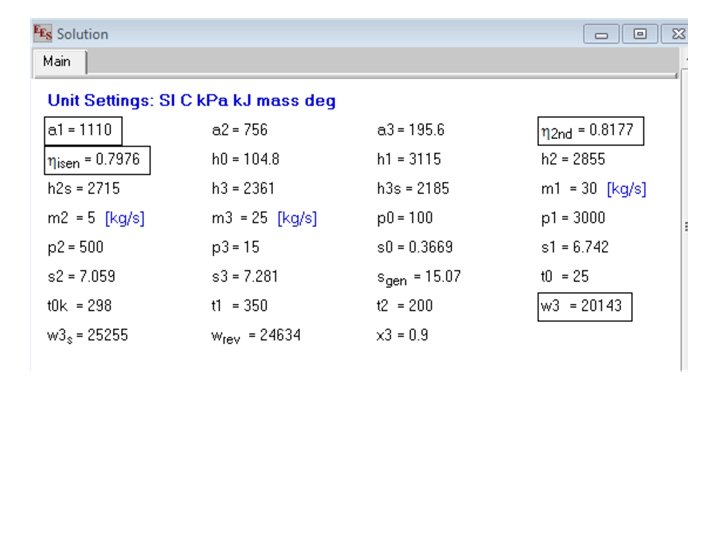
1 T 2 s P=p 1 P=p 2 2 For the case of a turbine expansion S 2 s T 1 P=p 2 2 For the case of a compressor P=p 1 S Example problem An insulated steam turbine, receives 30 kg/s of steam at 3 Mpa, 350 C. In the turbine where the pressure is 500 kpa, steam is beld off at the rate of 5 kg/s, for processing equipment. The temperature of this steam is 200 C. The balance of the steam leaves the turbine at 15 kpa, 90% quality. Find the availability per kg of steam at the entry, the isentropic efficiency and the second law efficiency of the turbine. What is the actual work output of the turbine Ans: 1110 kj/kg, eta_isen=. 7975, eta_2 nd=. 8176, w_act=20144 kw
![m 1=30 [kg/s]; m 2= 5[kg/s]; m 3=25 [kg/s] p 1=3000; p 2=500; p m 1=30 [kg/s]; m 2= 5[kg/s]; m 3=25 [kg/s] p 1=3000; p 2=500; p](http://slidetodoc.com/presentation_image_h2/a860dba7c1631ca8c69665b15e667fe4/image-6.jpg)
m 1=30 [kg/s]; m 2= 5[kg/s]; m 3=25 [kg/s] p 1=3000; p 2=500; p 3=15; x 3=. 9; p 0=100 t 1= 350; t 2=200; t 0=25; t 0 k=298 30 kg/s h 1=enthalpy(steam, p=p 1, t=t 1) 3 MPa, 350 C 1 s 1=entropy(steam, p=p 1, t=t 1) h 0=enthalpy(water, p=p 0, t=t 0) s 0=entropy(water, p=p 0, t=t 0) h 2=enthalpy(steam, p=p 2, t=t 2) s 2=entropy(steam, p=p 2, t=t 2) 2 h 3=enthalpy(steam, p=p 3, x=x 3) 5 kg/s s 3=entropy(steam, p=p 3, x=x 3). 5 MPa, 200 C a 1=h 1 -t 0 k*s 1 -(h 0 -t 0 k*s 0) { availability at entry} a 2=h 2 -t 0 k*s 2 -(h 0 -t 0 k*s 0) a 3=h 3 -t 0 k*s 3 -(h 0 -t 0 k*s 0) m 1*h 1=m 2*h 2+m 3*h 3+w 3 { w 3 = actual work output} m 1*s 1+s_gen=m 2*s 2+m 3*s 3 w_rev=w 3+t 0 k*s_gen eta_2 nd=w 3/w_rev h 2 s=enthalpy(steam, p=p 2, s=s 1) h 3 s=enthalpy(steam, p=p 3, s=s 1) m 1*h 1=m 2*h 2 s+m 3*h 3 s+w 3_s {w 3 s = isentropic work of turbine} eta_isen=w 3/w 3_s 3 25 kg/s 15 k. Pa, 90%quality
- Slides: 7