Maurice Buchbinder M D LAAC WATCHMAN Next Clinical

Maurice Buchbinder, M. D. LAAC WATCHMAN™ Next Clinical Trial Maurice Buchbinder, MDCM, FACC, FSCAI Medical Director Foundation for Cardiovascular Medicine San Diego, California Professor of Clinical Medicine Stanford Hospital and Clinics Stanford, California

Maurice Buchbinder, M. D. DISCLOSURE STATEMENT OF FINANCIAL INTEREST In the past 12 months, I or my spouse/partner has had a financial interest/arrangement with the organization(s) listed below. BSCI • Scientific Advisory Board Member • Speaker Bureau • Equity Ownership
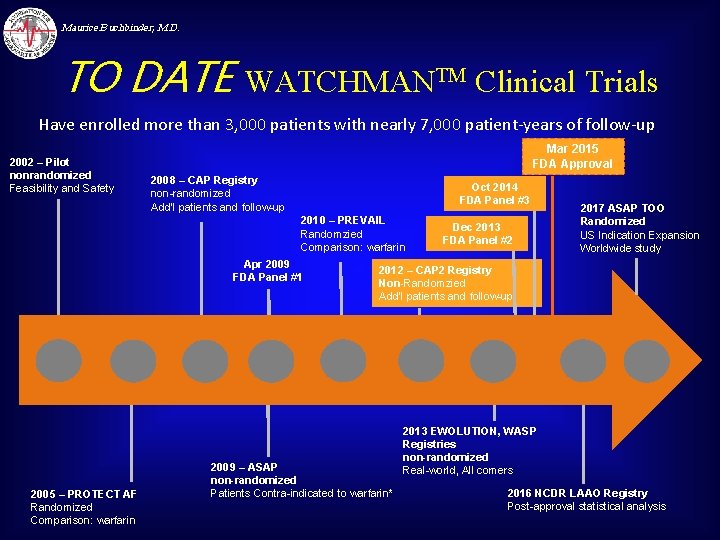
Maurice Buchbinder, M. D. TO DATE WATCHMANTM Clinical Trials Have enrolled more than 3, 000 patients with nearly 7, 000 patient-years of follow-up 2002 – Pilot nonrandomized Feasibility and Safety Mar 2015 FDA Approval 2008 – CAP Registry non-randomized Add’l patients and follow-up Oct 2014 FDA Panel #3 2010 – PREVAIL Randomzied Comparison: warfarin Apr 2009 FDA Panel #1 2005 – PROTECT AF Randomized Comparison: warfarin Dec 2013 FDA Panel #2 2017 ASAP TOO Randomized US Indication Expansion Worldwide study 2012 – CAP 2 Registry Non-Randomzied Add’l patients and follow-up 2009 – ASAP non-randomized Patients Contra-indicated to warfarin* 2013 EWOLUTION, WASP Registries non-randomized Real-world, All comers 2016 NCDR LAAO Registry Post-approval statistical analysis

Maurice Buchbinder, M. D. Highlights of WATCHMAN™ Clinical Trial Findings Safe Alternative to Long-Term Warfarin Therapy, with Comparable Stroke Risk Reduction SAFE • • • >95% implant success rate < 1 -3% M. A. E. (1. 2% rate of pericardial effusion) < 0. 4% need for urgent surgical intervention PROTECT AF / PREVAIL Meta Analysis: EFFECTIVE • 27% reduction all cause mortality* EWOLUTION Registry (Late Breaker Clinical Trial at HRS): • 52% reduction CV/unexplained death* • 84% reduction stroke rate versus expected stroke rate** • 72% reduction major bleeding > 6 months post procedure* • 48% reduction major bleeds versus expected bleeding rate*** • 92% of patient were able to stop taking warfarin after 45 days • 99% of patients were able to stop taking warfarin at 1 year SUCCESSFUL * versus rate on warfarin, ** versus projected rate on no therapy, *** versus projected rate on warfarin
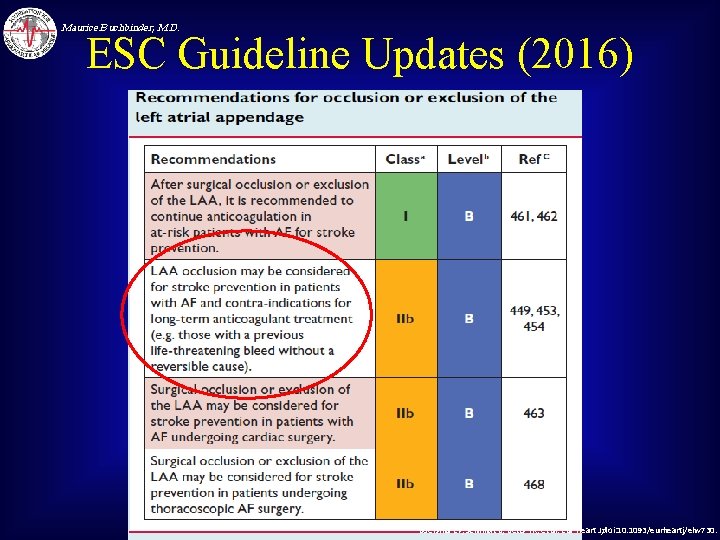
Maurice Buchbinder, M. D. ESC Guideline Updates (2016) Boersma LV, Schmidt B, Betts TR, et al. Eur Heart J; doi: 10. 1093/eurheartj/ehv 730.

Maurice Buchbinder, M. D. Current US Indication The WATCHMAN Device is indicated to reduce the risk of thromboembolism from the left atrial appendage in subjects with non-valvular atrial fibrillation who: • Are at increased risk for stroke and systemic embolism based on CHADS 2 or CHA 2 DS 2 -VASc scores; • Are deemed by their physicians to be suitable for warfarin for 45 days post implantation; and • Have an appropriate rationale to seek a nonpharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin

Maurice Buchbinder, M. D. WATCHMAN’s comprehensive clinical plan is designed to ADDRESS and TREAT all patients with NVAF Non-Valvular Atrial Fibrillation Patient Population Unable to take oral anticoagulant Reasons to seek an alternative to long term blood thinners NCDR Registry ASAP Too Able to take long term oral anticoagulant (ongoing) SALUTE: JPN approval trial (2017 E complete) (2021 E complete) WM with TAVR ISR (2017 E start) WM with Ablation (2018 E start) Prior Intracranial Hemorrhage Prior GI Bleeds (2018 E start) (2019 E start) Renal Dysfunction (2019 E start) Ablation with WM vs. Ablation with NOAC (2019 start)

Maurice Buchbinder, M. D. WATCHMAN’s comprehensive clinical plan is designed to ADDRESS and TREAT all patients with NVAF Non-Valvular Atrial Fibrillation Patient Population OAT Risk >>Benefit Unable to take Reasons to seek an alternative to long term blood thinners NCDR Registry ASAP Too (2021 E complete) OAT Risk << Benefit Able to take long term blood thinners (ongoing) SALUTE: JPN approval trial (2017 E complete) WM with TAVR ISR (2017 E start) WM with Ablation (2018 E start) Prior Intracranial Hemorrhage Prior GI Bleeds (2018 E start) (2019 E start) Renal Dysfunction (2019 E start) Ablation with WM vs. Ablation with NOAC (2019 start)

Maurice Buchbinder, M. D. ASAP-TOO Update
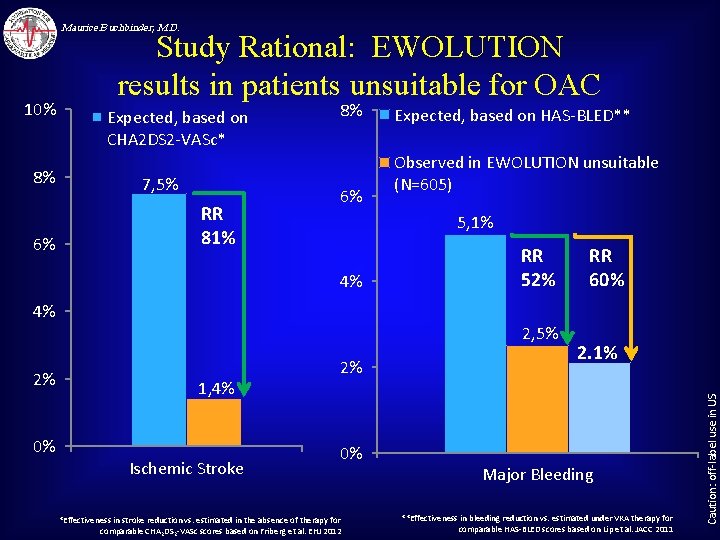
Maurice Buchbinder, M. D. 8% 6% Expected, based on CHA 2 DS 2 -VASc* 7, 5% RR 81% 8% 6% 4% 0% Observed in EWOLUTION unsuitable (N=605) 5, 1% 4% 2% Expected, based on HAS-BLED** RR 52% 2, 5% 1, 4% Ischemic Stroke 2% 0% *Effectiveness in stroke reduction vs. estimated in the absence of therapy for comparable CHA 2 DS 2 -VASc scores based on Friberg et al. EHJ 2012 RR 60% 2. 1% Major Bleeding **Effectiveness in bleeding reduction vs. estimated under VKA therapy for comparable HAS-BLED scores based on Lip et al. JACC 2011 Caution: off-label use in US 10% Study Rational: EWOLUTION results in patients unsuitable for OAC

Maurice Buchbinder, M. D. ASAP-TOO Study Objective • The primary objective of this study is to establish the safety and effectiveness of the WATCHMAN™ Left Atrial Appendage Closure (LAAC) Device for patients with non-valvular atrial fibrillation who are deemed not to be eligible for anti-coagulation therapy to reduce the risk of stroke. • The device is intended to reduce the risk of ischemic stroke and systemic embolism.

Maurice Buchbinder, M. D. ASAP-TOO (NCT 02928497): Overview Study Objective Evaluate LAA Closure with WATCHMAN in NVAF patients deemed not suitable for oral anti-coagulation therapy Study Design Prospective, multi-center Randomized 2: 1 (Watchman vs Control) Considering Group Sequential Design Effectiveness Endpoint Time to first occurrence of ischemic stroke or systemic embolism Primary Endpoint Safety Endpoint 7 -day rate of all-cause death, ischemic stroke, systemic embolism, or device- or procedure- related events requiring open cardiac surgery or major endovascular intervention Patient Population 888 Number of Sites 100 global sites Follow-up* • • 45 Day with TEE 6, 18 month phone visit 12 month with TEE Bi-annually for years 2 -5 * Brain imaging required at baseline if prior stroke or TIA
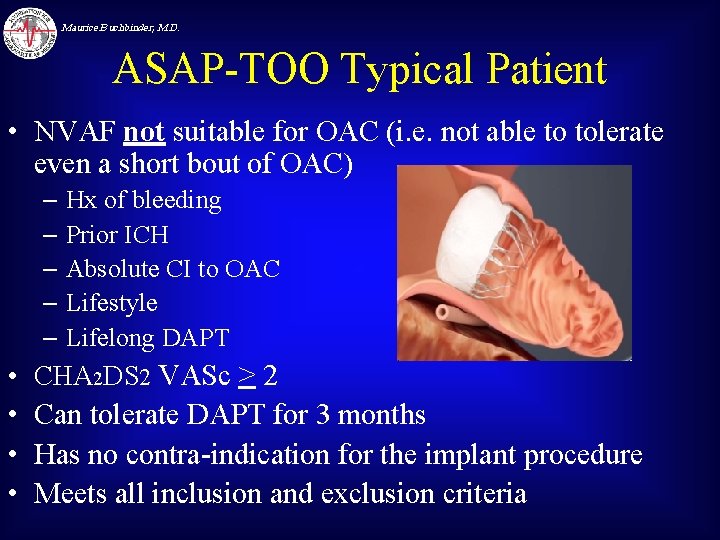
Maurice Buchbinder, M. D. ASAP-TOO Typical Patient • NVAF not suitable for OAC (i. e. not able to tolerate even a short bout of OAC) – Hx of bleeding – Prior ICH – Absolute CI to OAC – Lifestyle – Lifelong DAPT • CHA 2 DS 2 VASc > 2 • Can tolerate DAPT for 3 months • Has no contra-indication for the implant procedure • Meets all inclusion and exclusion criteria

Maurice Buchbinder, M. D. Reasons Unsuitable for OAC • OAC unsuitability should be a shared decision between the subject and study physicians • 2 study physicians (implanting physician + non-implanting physician) must determine that a subject is unsuitable for OAC • Decision and reason(s) subject is unsuitable for OAC must be documented in the subject’s study records and on the case report form • Acceptable reasons include the following categories: – – History of overt bleeding related or unrelated to oral anticoagulants Increased risk of bleeding or bleeding tendencies Contraindications to warfarin and/or direct OAC (DOAC) Other contraindications

Maurice Buchbinder, M. D. Reasons Unsuitable for OAC • History of overt bleeding related or unrelated to oral anticoagulants: – Prior history of intracranial or subdural hemorrhage – Other clinically relevant organ bleeding as defined by requiring hospitalization, transfusion, or medical intervention, including: gastrointestinal, genitourinary, ocular, spinal, pulmonary retroperitoneal, pericardial, or ENT. Last event must be within the 6 -months prior to randomization. – Recurrent epistaxis requiring ER visit, hospitalization or physician intervention. Last event must be within the 6 months prior to randomization.

Maurice Buchbinder, M. D. Reasons Unsuitable for OAC • Increased risk of bleeding or bleeding tendencies: – Gastrointestinal lesions resulting in clinically relevant bleeding as defined by requiring hospitalization, transfusion or medical intervention (e. g. , esophageal varices, diverticular disease with a history of bleeding in which the site was not identified and presumed to be diverticular). Last event must be within the 6 months prior to randomization. – Active inflammatory bowel disease – Peptic ulcer disease with gastrointestinal bleeding in which it is deemed that anticoagulation cannot be safely initiated or restarted following healing of the peptic ulcer. – Uncontrolled seizures – History of traumatic falls with the likelihood of recurrence – Cerebral amyloid angiopathy – Significant thrombocytopenia (defined as platelet count < 50 x 109/L) – Need for lifelong dual antiplatelet therapy

Maurice Buchbinder, M. D. Reasons Unsuitable for OAC • Contraindications to warfarin and/or direct OAC (DOAC) – Severe renal failure (GFR <30 m. L/min/1. 73 m 3) – Allergy to the above agents • Other contraindications including: – Lifestyle or occupational bleeding risk (i. e. anyone who is at risk of trauma as a result of their occupation or their lifestyle. For example, high voltage electrical line workers, airline pilots, manual laborers, extreme sports enthusiasts, etc. ) – Poor control on warfarin (time in therapeutic range <50%) and intolerance to the DOACs – Other medical or social reasons that make OACs unsuitable (For example, an elderly patient with poor social support and a high risk of bleeding resulting in the use of aspirin alone despite a high stroke risk)

Maurice Buchbinder, M. D. Key Inclusion Criteria • The subject has documented paroxysmal, persistent, permanent or longterm/longstanding persistent non-valvular atrial fibrillation (i. e. , the subject has not been diagnosed with rheumatic mitral valvular heart disease). • The subject has a calculated CHA 2 DS 2 -VASc score of 2 or greater. • The subject is deemed by two study physicians to be unsuitable for oral anticoagulation. • The subject is deemed by a study physician to be suitable for the defined protocol pharmacologic regimen of aspirin and clopidogrel* therapy following WATCHMAN Closure Device implant. • The subject had or is planning to have any invasive cardiac procedure within 30 days prior to randomization (e. g. , cardioversion, ablation). *Ticagrelor or prasugrel may be used in place of clopidogrel if subject has another indication or a known resistance to clopidogrel. Prasugrel should only be used if neither clopidogrel nor ticagrelor can be used due to potential for increased bleeding risk.

Maurice Buchbinder, M. D. Key Inclusion Criteria • The subject is planning to have any cardiac or non-cardiac invasive or surgical procedure that would necessitate stopping or modifying the protocol required medication regimen within 90 days after the WATCHMAN Closure Device implant (e. g. , cardioversion, ablation, cataract surgery). • The subject had a prior stroke (of any cause) or TIA within the 30 days prior to randomization. • The subject has a history of atrial septal repair or has an ASD/PFO device. • The subject has an implanted mechanical valve prosthesis in any position.

Maurice Buchbinder, M. D. Key Inclusion Criteria • The subject suffers from New York Heart Association Class IV Congestive Heart Failure. • The subject has LVEF < 30%. • The subject has a life expectancy of less than two years. • The subject has a known or suspected hypercoagulable state. • The subject had or is planning to have any invasive cardiac procedure within 30 days prior to randomization (e. g. , cardioversion, ablation). • The subject had a prior BARC type 3 or 4 bleeding event within the 14 days prior to randomization. Lack of resolution of related clinical sequelae, or planned and pending interventions to resolve bleeding/bleeding source, are a further exclusion regardless of timing of the bleeding event.

Maurice Buchbinder, M. D. Neurological Assessments Assessment Who When Neurologist Assessment Must be conducted by a delegated study Neurologist • Baseline • 12 -Month Visit • 24 -Month Visit Modified Rankin Scale Must be conducted by (MRS) delegated study staff with current MRS certification NIH Stroke Scale (NIHSS) • Baseline • All office and telephone visits • 90 days post stroke/TIA event Must be conducted by • Baseline delegated study staff with • All office visits (3 -M, 12 -M, current NIHSS certification annual) • At the time of stroke/TIA ^May be conducted by non-study staff at the time of the event^

Maurice Buchbinder, M. D. Echo Exclusion Criteria (per baseline TEE) • The subject has intracardiac thrombus, LAA sludge (gelatinous, nonadherent, intracavitary echodensity more layered than dense spontaneous echo contrast (SEC) seen continuously throughout cardiac cycle) or dense SEC visualized by TEE within 2 calendar days prior to randomization. • The subject has an existing pericardial effusion with a circumferential echo -free space >5 mm, and/or the subject has signs/symptoms of acute or chronic pericarditis, and/or there is evidence of tamponade physiology. • The subject has a high-risk patent foramen ovale (PFO) with an atrial septal aneurysm excursion > 15 mm or length ≥ 15 mm. • The subject has a high-risk PFO with a large shunt defined as early (within 3 beats) or substantial passage of bubbles. • The subject has significant mitral valve stenosis (i. e. , MV <1. 5 cm 2). • The subject has complex atheroma with mobile plaque of the descending aorta and/or aortic arch. • The subject has a cardiac tumor.

Maurice Buchbinder, M. D. Randomization WHAT: • Eligible subjects will be randomized 2: 1 to receive the WATCHMAN Device to Control WHEN: • After baseline TEE has been completed and once all echo exclusion criteria have been verified • Randomization must be within 2 calendar days of the baseline TEE • If randomized to device, must go to implant within 2 calendar days of baseline TEE HOW: • Randomization assignment is obtained via Medidata Rave

Maurice Buchbinder, M. D. Randomization Assignments (2 Device: 1 Control) Device Group Treatment Control Group Treatment • WATCHMAN Device Implant with modified post implant medication therapy • Single antiplatelet therapy or no therapy for the duration of the trial at the discretion of the study physician. • Subjects will be allowed to be on dual antiplatelet therapy if indicated. Visit Interval Aspirin Clopidogrel* Discharge to 3 -M Visit Yes, suggested dose: 75 -100 mg Yes, suggested dose: 75 mg 3 -M Visit to 12 -M Visit Yes, suggested dose: 75 -100 mg No, unless other indication Following the 12 -M Visit No, unless other indication *Clopidogrel may be substituted with ticagrelor or prasugrel if the subject requires the medication for other indications (e. g. acute coronary syndromes treated with drug eluting stents) or if the subject has a known resistance to clopidogrel. Prasugrel should only be used if neither clopidogrel nor ticagrelor can be used due to potential for increased bleeding risk.

Maurice Buchbinder, M. D. FOLLOW-UP Clinic/Office Follow up Visits: • The subject is expected to return to the office/clinic at 3, 12, 24, 36, 48, and 60 months (yearly). • This Clinic visit should be the same clinic where the subject was originally seen for their screening. • These visits must be conducted by the PI or personnel delegated by the PI, as noted on the Site Signature and Responsibility Log. (PLEASE REVIEW THIS AND ENSURE IT IS COMPLETE PRIOR TO SUBJECT ENROLLMENT AT YOUR SITE) • The personnel delegated to conduct these visits should be, at minimum, a sub-investigator. Phone Follow up: The subjects will be contacted via phone at 6, 18, 30, 42, and 54 months to review their medications, any adverse events, and to complete the Modified Rankin Scale.

ASAP TOO Sites and Randomization Status Maurice Buchbinder, M. D. Sites 53 0 Randomized Patients 40 60 66 0 As of 2/27/18 20 100 80 888 200 400 600 800 Current Goal 100 Current Goal
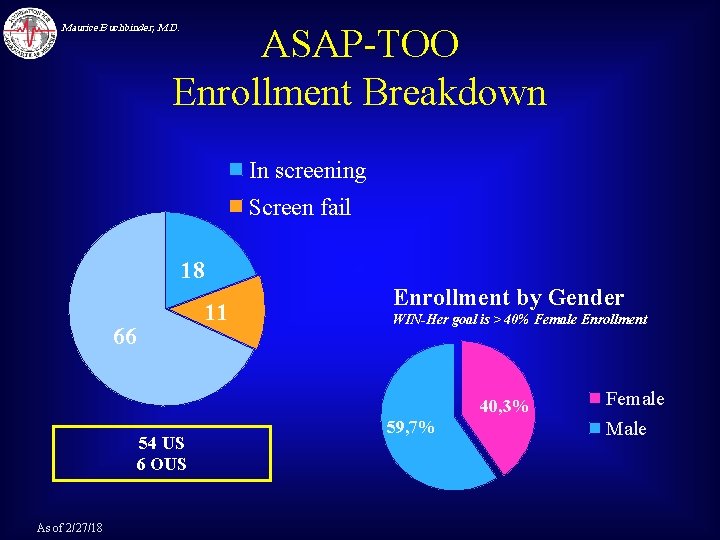
ASAP-TOO Enrollment Breakdown Maurice Buchbinder, M. D. In screening Screen fail 18 66 11 Enrollment by Gender WIN-Her goal is > 40% Female Enrollment 40, 3% 54 US 6 OUS As of 2/27/18 59, 7% Female Male
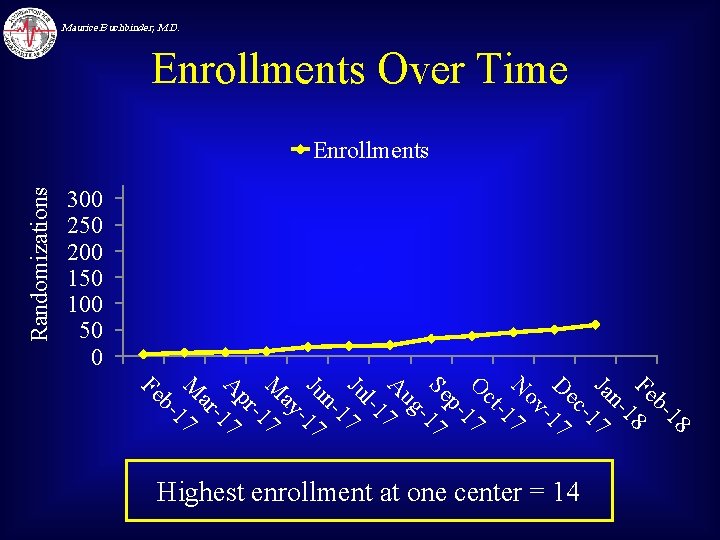
Maurice Buchbinder, M. D. Enrollments Over Time Randomizations Enrollments 300 250 200 150 100 50 0 18 b. Fe 8 1 n. Ja 7 -1 ec D -17 ov N 7 -1 ct O 17 p. Se -17 ug A 7 l-1 Ju 17 n. Ju -17 ay M 17 pr A 17 ar M 17 b- Fe Highest enrollment at one center = 14

Maurice Buchbinder, M. D. WATCHMAN’s comprehensive clinical plan is designed to ADDRESS and TREAT all patients with NVAF Non-Valvular Atrial Fibrillation Patient Population OAT Risk >>Benefit Unable to take Reasons to seek an alternative to long term blood thinners NCDR Registry OAT Risk << Benefit Able to take long term blood thinners (ongoing) SALUTE: JPN approval trial (2017 E complete) ASAP Too (2021 E complete) WM with TAVR ISR (2017 E start) WM with Ablation (2018 E start) Prior Intracranial Hemorrhage Prior GI Bleeds (2018 E start) (2019 E start) Renal Dysfunction (2019 E start) Ablation with WM vs. Ablation with NOAC (2019 start)

Maurice Buchbinder, M. D. WATCHMAN FLX™: Designed to Broaden Treatment Matrix and Improve Ease of Use • Designed for greatly enhanced stability and ease-of-use • Designed for greater apposition to appendage wall − New anchor design, additional anchors and reduced main body taper • Anticipate starting EU and U. S. clinical trials mid-year 2018 Caution: WATCHMAN FLX is an Investigational Device. Limited by Federal (or US) law to investigational use only. Not available for sale.

Maurice Buchbinder, M. D. Protection against Embolism for non-valvular AF Patients: Investigational Device Evaluation of the WATCHMAN FLX™ LAA Closure Technology US-only IDE • Single arm non-randomized study design • Up to 490 enrollments • Up to 45 US sites • Primary Safety Endpoint – The occurrence of one of the following events between the time of implant and within 7 days following the procedure or by hospital discharge, whichever is later: all-cause death, ischemic stroke, systemic embolism, or device- or procedure- related events requiring open cardiac surgery or major endovascular intervention such as pseudoaneurysm repair, AV fistula repair, or other major endovascular repair. • • Follow-up at 45 days, 6, 12, 18 and 24 months FPI ~ 1 st half of 2018

Maurice Buchbinder, M. D. CASE STUDY

Maurice Buchbinder, M. D. Case Study – Patient A • Patient profile – medical history • 70 year old woman with chronically persistent AF on warfarin • Moderate dementia of unclear etiology, diabetes mellitus on oral agents • Prior history of lobar hemorrhagic stroke with significant residual deficit ~ 7 years ago • Assisted living facility • No history of hypertension, atherosclerosis, or CAD • Recent CT scan shows multiple cerebral “microbleeds” in a posterior distribution • Presumptive clinical diagnosis of Cerebral Amyloid Angiopathy (CAA) – reason documented for unsuitability to OAC • Increased risk of bleeding: Cerebral Amyloid Angiopathy • Secondary: dementia

Maurice Buchbinder, M. D. Case Study – Patient B • Patient profile – Medical history • 60 year old man • History of persistent AF, hypertension, CAD, PVD, peptic ulcer disease with two prior episodes of acute GI bleeding a year ago which required hospitalization and cessation of warfarin • On apixaban, he experienced duodenal ulcer-associated upper GI bleed three months ago, requiring endoscopic intervention and transfusion of 4 units of PRBC • Recent EGD showed resolution of the acute bleed but GI consultant feels risk of rebleeding is high – reason documented for unsuitability to OAC • Gastrointestinal lesions resulting in clinically relevant bleeding as defined by requiring hospitalization, transfusion or medical intervention (e. g. Esophageal varices, diverticular disease with a history of bleeding in which the site was not identified and presumed to be diverticular). Last event must be within the 6 months prior to randomization. • Peptic ulcer disease with gastrointestinal bleeding in which it is deemed that anticoagulation cannot be safely initiated or restarted following healing of the peptic ulcer.

Maurice Buchbinder, M. D. Case Study – Patient C • Patient profile – Medical history • 48 year old man • History of paroxysmal AF with two prior PVI procedures and residual AF • Prior history: type 1 diabetes mellitus, hypertension, end-stage renal failure on hemodialysis • On ASA 81 mg daily • Prior use of warfarin was associated with calciphylaxis • Prior use of rivaroxaban associated with intolerable headaches – reason documented for unsuitability to OAC • Warfarin contraindication • Unable to take DOAC

Maurice Buchbinder, M. D. Case Study – Patient D • Patient profile – Medical history • 68 year old woman • History of paroxysmal AF, CHADS-VASc = 4, previous ischemic stroke • On warfarin chronically, INR well controlled between 2. 2 and 2. 7 • Experienced sudden monocular blindness due to left intra-ocular hemorrhage - warfarin stopped and reversed with vitamin K • 1 week later, vision loss determined to be permanent, but intraocular bleeding no longer active and clinically stable – reason documented for unsuitability to OAC • Other clinically relevant organ bleeding as defined by requiring hospitalization, transfusion or medical intervention, including: gastrointestinal, genitourinary, ocular, spinal, pulmonary retroperitoneal, pericardial, or ENT. Last event must be within the 6 -months prior to randomization.
- Slides: 36