Matterhas mass and occupies space Properties Chemicalexhibited by

Matter-has mass and occupies space Properties: Chemical-exhibited by matter as it undergoes changes in composition Physical-exhibited by matter without a change in composition. Extensive-depends on the amount of matter Intensive-independent of the amount of matter Physical & Intensive:

Chemical Change-one or more substances are either used up or formed and energy is absorbed or released. Physical Change-change in physical properties without a change in composition. Energy-capacity to do work or transfer heat. Kinetic Energy (KE)-motion Potential Energy (PE)-position, condition or composition. Release of Energy - Exothermic Absorption of Energy- Endothermic

Law of Conservation of Matter-There is no observable change in the quantity of matter during a chemical reaction or during a physical Change. Law of Conservation of Energy-Energy cannot be created or destroyed in a chemical reaction or in a physical change. It can only be converted from one form to another. E=mc 2 Combined-The combined amount of matter and energy in the universe is fixed. 2 Mg + O 2 -----> 2 Mg. O

Chemical Changes P 4(s)+6 Cl 2(g)----->4 PCl 3(l)

Physical Change Chemical Change H 2 O O 2
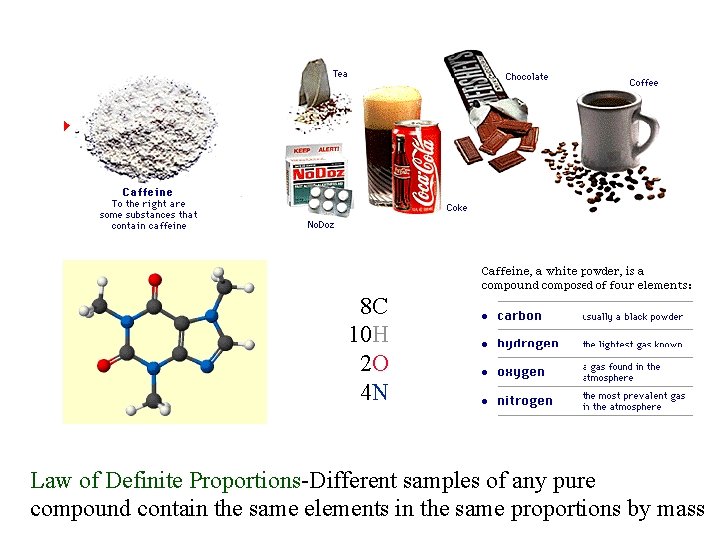
8 C 10 H 2 O 4 N Law of Definite Proportions-Different samples of any pure compound contain the same elements in the same proportions by mass

Separation of Mixtures
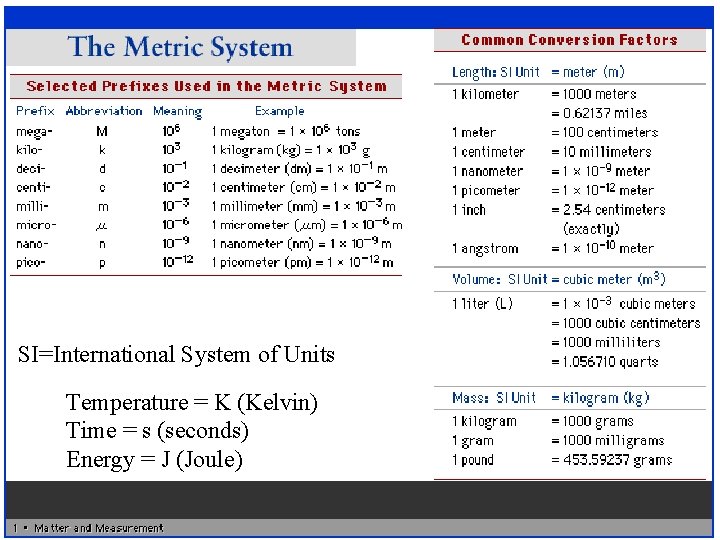
SI=International System of Units Temperature = K (Kelvin) Time = s (seconds) Energy = J (Joule)

% Composition %A (mass)= Parts of A (by mass) 100 parts mixture (by mass) Mass A 8 C 10 H 2 O 4 N Mass mixture 49. 5% C 5. 15% H 16. 5% O 28. 9% N 49. 5 grams of Carbon 100 grams of caffeine
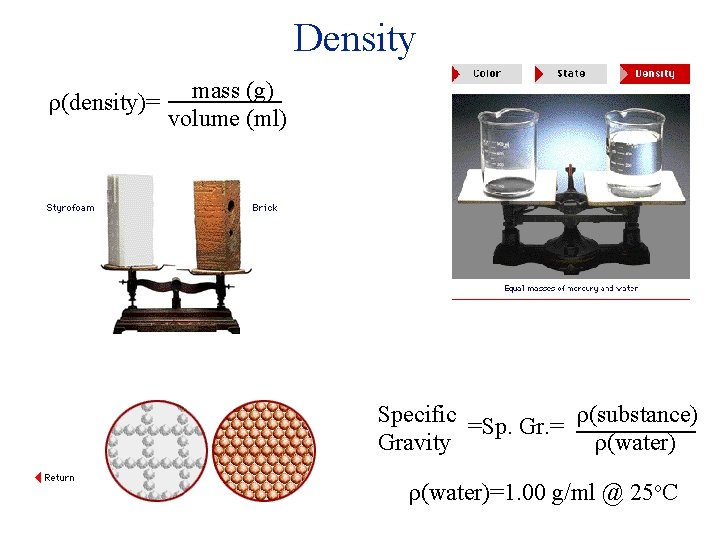
Density (density)= mass (g) volume (ml) Specific =Sp. Gr. = (substance) Gravity (water)=1. 00 g/ml @ 25 o. C

K=o. C+273. 15

Heat Transfer Specific Heat = (Amount of heat in J) (mass of substance in g)(Temperature change in K/o. C) Heat Capacity= (Csp)(mass) units=(J/ o. C)
- Slides: 12