Matter What do you know about matter Matter

Matter What do you know about matter?

Matter: Anything that has mass and takes up ______.

Three Kinds (classes) of Matter Atom (pure substance) – simplest form of matter. Compound (pure substance) – Two or more atoms bonded chemically. Mixture – Two or more pure substances combined so that the identity of the substances does not change.
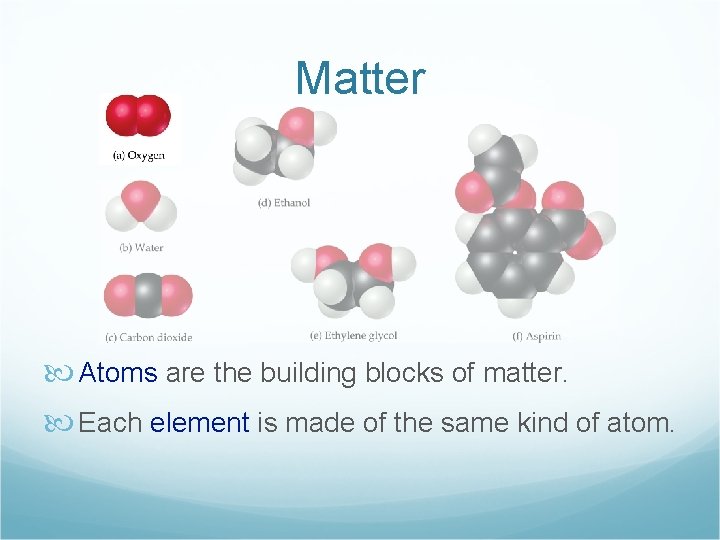
Matter Atoms are the building blocks of matter. Each element is made of the same kind of atom.
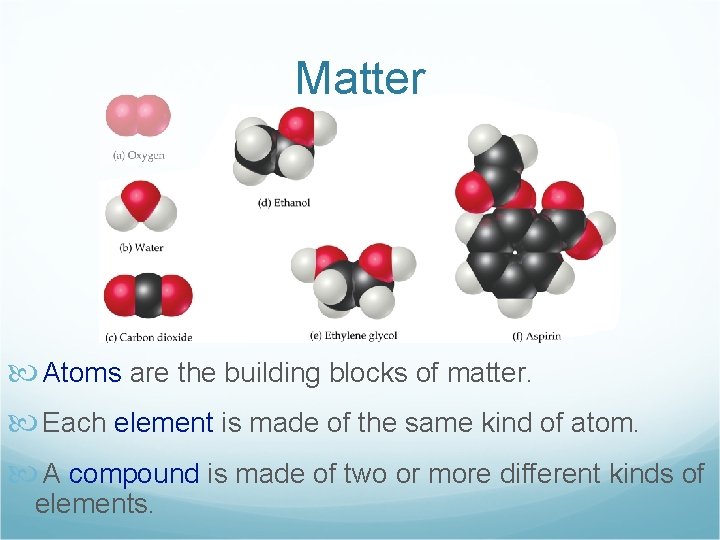
Matter Atoms are the building blocks of matter. Each element is made of the same kind of atom. A compound is made of two or more different kinds of elements.
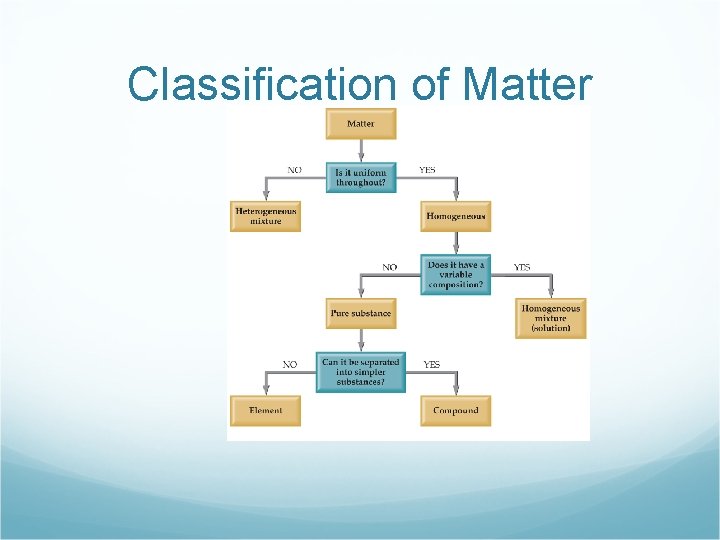
Classification of Matter

Mixtures and Compounds

Chemical Reactions In the course of a chemical reaction, the reacting substances are ______ to new substances.

Chemical Reactions

Separation of Mixtures

Distillation: Separates ______ mixture on the basis of differences in boiling point.

Filtration: Separates solid substances from ______ and solutions.

Chromatography: Separates substances on the basis of differences in ______ in a solvent.

Energy What do you know about energy?

Matter and Energy How do matter and energy interact?

Particle Theory of Matter All Matter is made up of tiny particles. All particles of one substance are the same. Different substances are made of different particles. There are spaces between the particles.

Particle Theory of Matter The particles are always moving. The more energy that the particles have, the faster they move. There attractive forces among particles. These forces are stronger when the particles are close together.

States (phases) of Matter Physical state is based on The nature of the bonds between the particles (potential energy) The motion of the particles (kinetic energy)

Solids Rigid Definite shape Definite volume

Solids Potential energy of bonds limits particle motion Particles vibrate in relatively fixed positions (low kinetic energy)

Liquids Definite volume Takes shape of container

Liquids Potential energy of the bonds is lower than in solids Particles have more kinetic energy than in solid state

Gases No definite shape No definite volume Takes shape of container

Gases Potential energy between particles is zero Greater kinetic energy than in solid and liquid states

Phase Changes Matter changes state by the addition of energy (heat) which Increases the kinetic energy of the particles Overcomes the potential energy between the particles to loosen/break the bonds

Phase Changes in particle motion are observed using a thermometer Temperature is a measure of the average kinetic energy of the particles
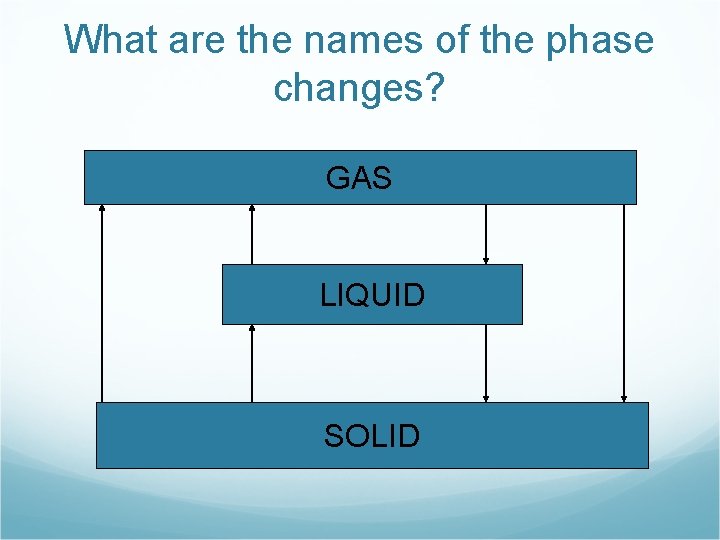
What are the names of the phase changes? GAS LIQUID SOLID
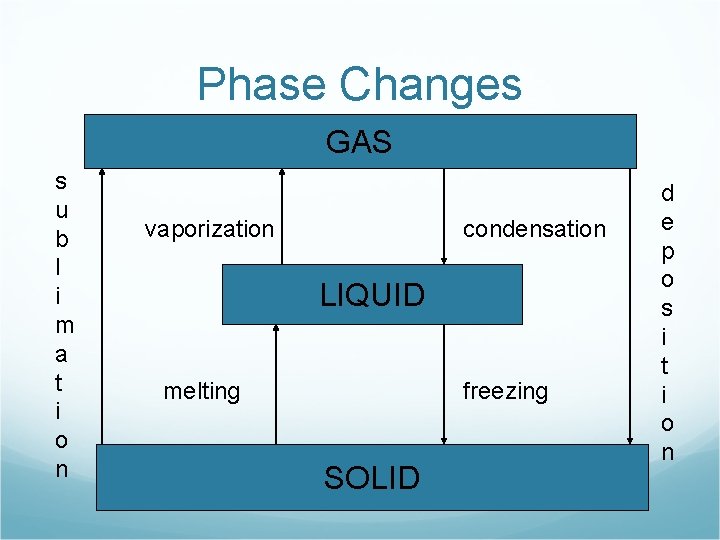
Phase Changes GAS s u b l i m a t i o n vaporization condensation LIQUID melting freezing SOLID d e p o s i t i o n
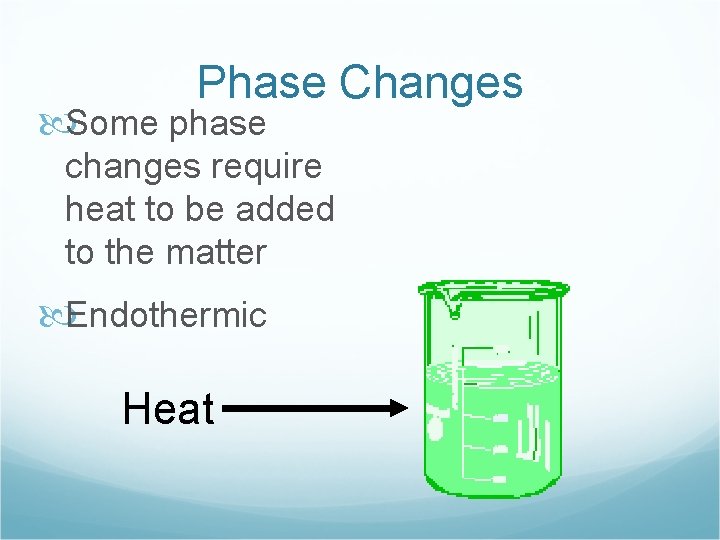
Phase Changes Some phase changes require heat to be added to the matter Endothermic Heat
What is Phase Changes happening to the heat? Heat
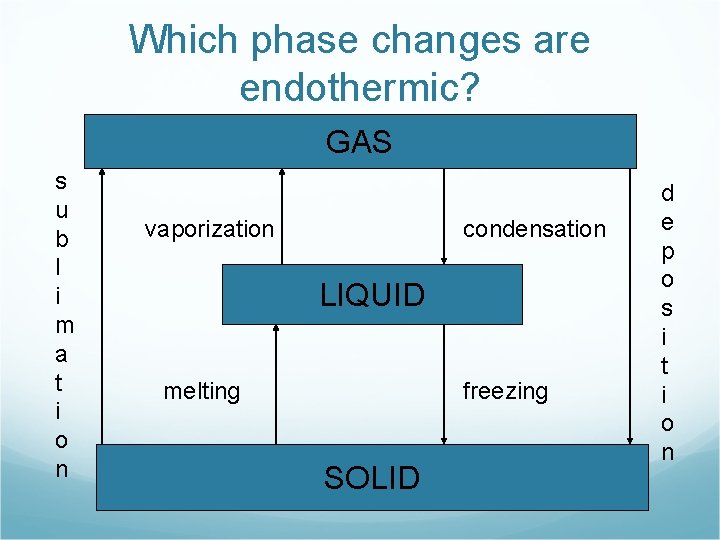
Which phase changes are endothermic? GAS s u b l i m a t i o n vaporization condensation LIQUID melting freezing SOLID d e p o s i t i o n
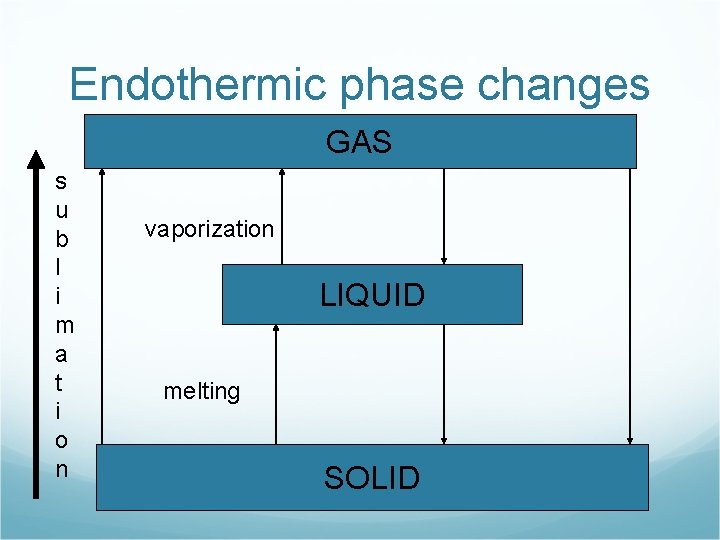
Endothermic phase changes GAS s u b l i m a t i o n vaporization LIQUID melting SOLID

Endothermic Phase Changes Melting H 2 O(s) + energy H 2 O(l) Vaporization H 2 O(l) + energy H 2 O(g) Sublimination H 2 O(s) + energy H 2 O(g)
Phase Changes Some phase changes require heat to be removed from the matter Exothermic Heat
Phase Changes What is happening as the heat is removed? Heat
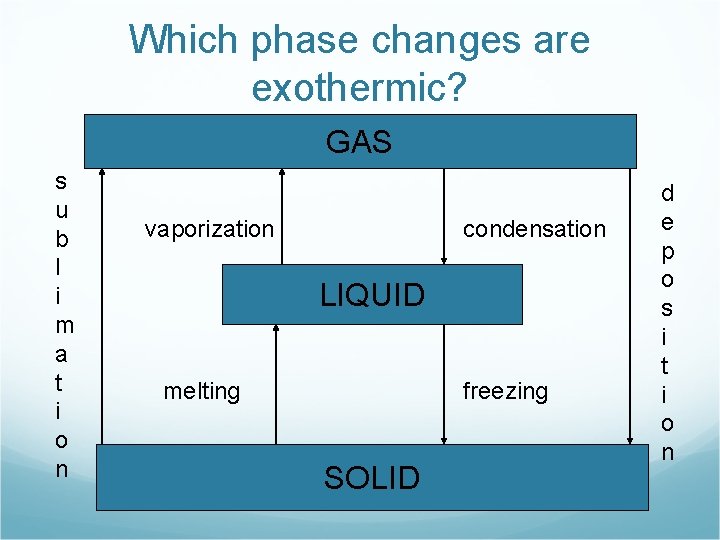
Which phase changes are exothermic? GAS s u b l i m a t i o n vaporization condensation LIQUID melting freezing SOLID d e p o s i t i o n
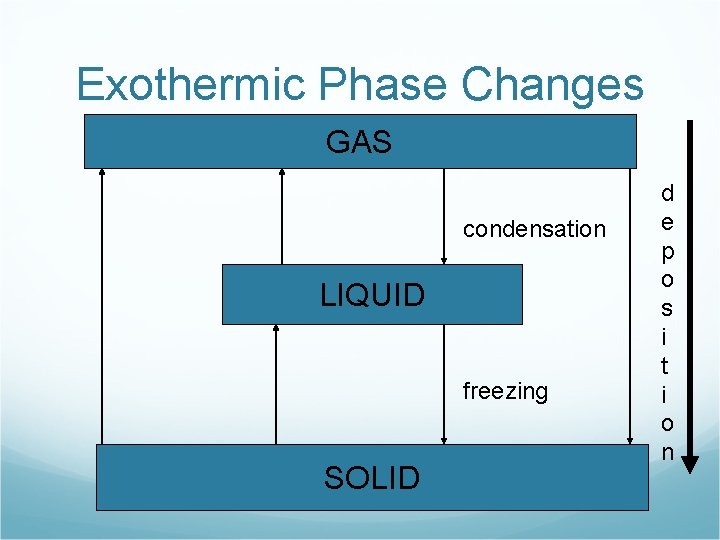
Exothermic Phase Changes GAS condensation LIQUID freezing SOLID d e p o s i t i o n

Exothermic Phase Changes Freezing H 2 O(l) H 2 O(s) + energy Condensation H 2 O(g) H 2 O(l) + energy Deposition H 2 O(g) H 2 O(s) + energy

Phase Changes The temperature at which phase changes occur is a unique property of each type of matter Melting point Boiling point “Known” values measured at 1 atm

Melting Point Matter exists in both solid and liquid states If matter is held at the melting point an equilibrium will occur

Boiling Point Matter exists in both liquid and vapor states If matter is held at the boiling point, an equilibrium will occur

Physical Properties that identify a substance and are unique to that specific substance

Physical Properties Phase changes points Density Color Crystal Structure Atomic/Molecular mass Spectroscopy etc

Physical vs. Chemical Change A Physical Change is a change in the phase or physical appearance of a substance without changing its chemical identity A Chemical Change involves a chemical reaction where the substances involved change chemically To distinguish the two it helps to ask if the substances can be returned to their original chemical makeup,

Physical Changes All phase changes are physical (boiling/evaporation, melting etc) Cutting/Breaking/Bending (reshaping) Separations (filtering, decanting, centrifuge, etc) Creating Solutions (dissolving) Magnetism Mixing

Chemical Changes Burning/Combustion Oxidation/Rusting Precipitating reactions Synthesis Chemical Replacements Decompositions Most Body Interactions (respiration, digestion) Explosions

Evidence of Chemical Changes Light/flame Smoke Bubbles Formation of a precipitate (insoluble solid formed by mixing two solutions) Thermal change Obvious new substance Sound

Evidence of Chemical Changes (cont. ) Loss of mass New or unexpected color “Reacting”
- Slides: 48