MATTER WAVES Louis de Broglie 1892 1987 Matter

MATTER WAVES Louis de Broglie 1892 -1987

Matter Waves Louis de Broglie was a physics graduate student when he suggested that matter had a wave nature. Recall that EMR acts as a wave in some experiments; diffraction, refraction, interference EMR also acts like a particle; photoelectric effect, momentum

The de Broglie hypothesis states that particles of matter also had wavelengths and could behave as waves, just as photons did.

Matter Waves De Broglie stated that since EMR has momentum and acts like a wave, perhaps matter, which has momentum, also acts like a wave. • He used Compton’s momentum of EMR formula, p=h/λ and equated it to the formula for momentum of matter, p=mv


De Broglie wave equation De Broglie wavelength is more significant for small masses traveling at high speeds rather than large masses traveling at low speeds

Matter Waves Matter waves have the wavelength of

Matter Waves This was not a popular idea. In fact, de Broglie’s thesis was held up until Einstein reviewed his work and agreed with it. To prove the existence of such waves is very difficult because they are so small.

Example Calculate the wavelength of a 50 kg skier moving at 16 m/s.

Solution

This means what? This wavelength (8. 3 x 10 -37 m) is about a billion, trillion times smaller than a hydrogen atom! This wavelength is so small that it is completely unobservable.

Examples Calculate the wavelength of an electron moving at 1. 0 x 106 m/s.

Solution

What does this mean? This wavelength (7. 3 x 10 -10 m) is in about the same wavelength of x-rays. This is observable.

Eg) Determine the De Broglie wavelength for an alpha particle traveling at 0. 015 c.
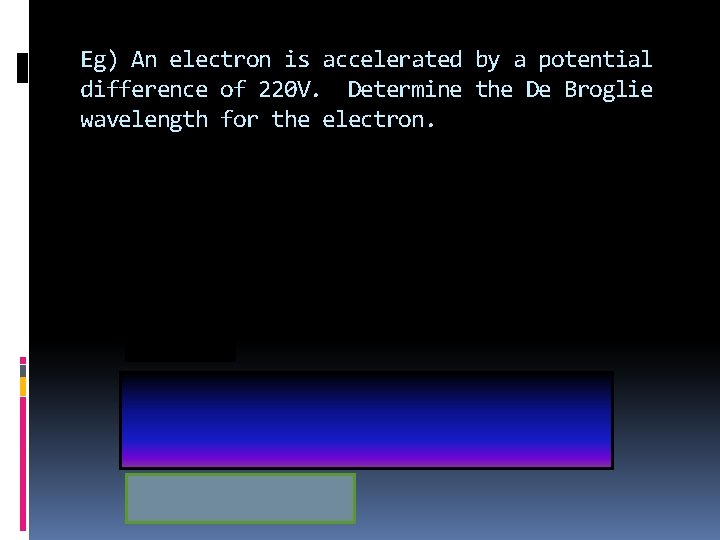
Eg) An electron is accelerated by a potential difference of 220 V. Determine the De Broglie wavelength for the electron.

Two years later, two American physicists demonstrated de Broglie’s experiment by performing experiments that showed interference patterns with electrons, as shown in this figure. (The 1925 experiment wasn’t actually a double slit experiment, but it showed the interference clearly. The double slit experiment with electrons was conducted in 1961. )

This behavior showed that whatever quantum law governed photons also governed particles. The wavelength of particles such as the electron is very small compared to the photon. For larger objects, the wavelength is even smaller still, quickly becoming so small as to become unnoticeable.

Story of six blind men
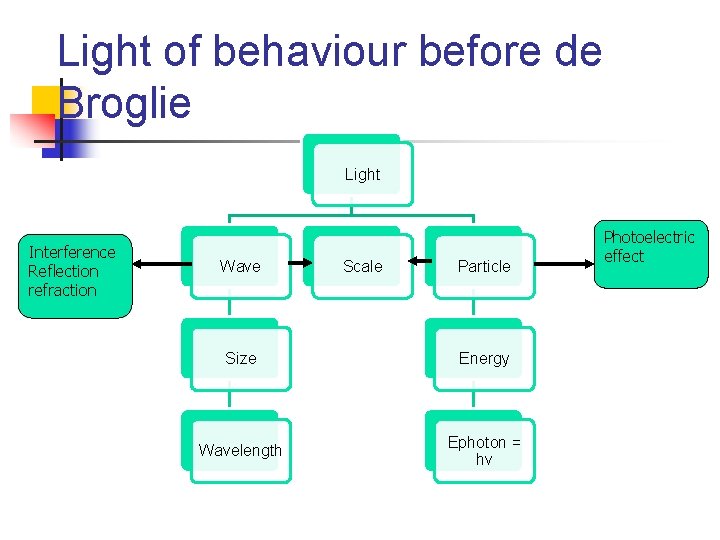
Light of behaviour before de Broglie Light Interference Reflection refraction Wave Scale Particle Size Energy Wavelength Ephoton = hv Photoelectric effect

Davisson-Germer Experiment • Soon after de Broglie’s idea was presented, Davisson and Germer observed evidence that beams of e¯ fired at the crystal lattice of metals diffract to produce nodes and antnodes. .

Davisson and Germer Experiment n n The experiment was firstly performed in 1927 by the captioned scientists. This experiment demonstrated the wave nature of the electron, confirming the earlier hypothesis of de Broglie. Putting wave-particle duality on a firm experimental footing, it represented a major step forward in the development of quantum mechanics. 54 ke. V electron beam is normally incident to a nickel crystal (lattice spacing d = 0. 215 nm). Animation Davisson-Germer Exp. Modern Physics Chapter One 22
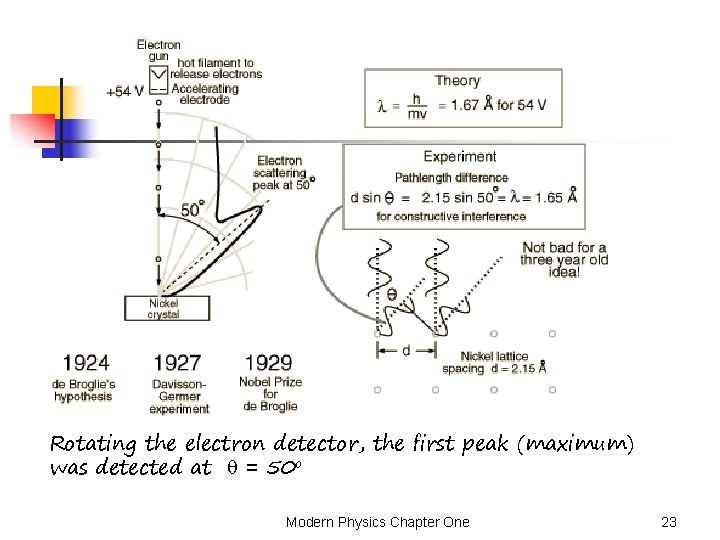
Rotating the electron detector, the first peak (maximum) was detected at = 50 o Modern Physics Chapter One 23
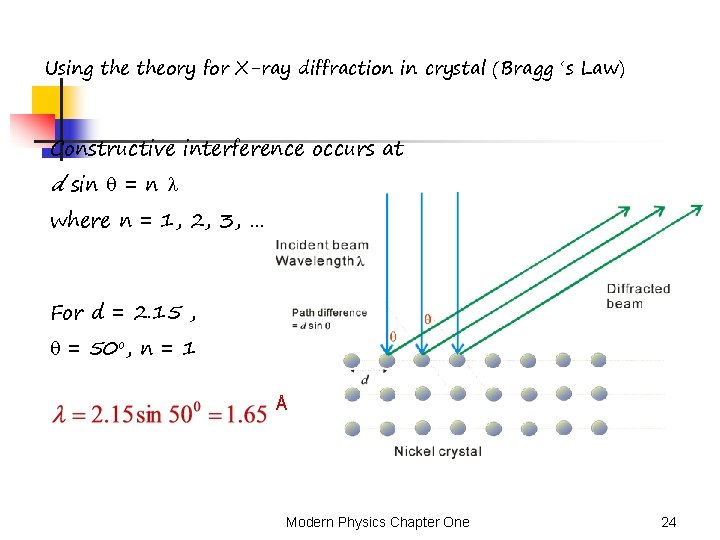
Using theory for X-ray diffraction in crystal (Bragg ‘s Law) Constructive interference occurs at d sin = n where n = 1, 2, 3, … For d = 2. 15 , = 50 o, n = 1 Å Modern Physics Chapter One 24

Momentum and wavelength of electron beam Rest energy of electron Eo Kinetic energy of electron K = q. V = 54 e. V<<Eo Classical (or non-relativistic) calculation is OK! 25

Wavelength of electron beam: Å Modern Physics Chapter One 26
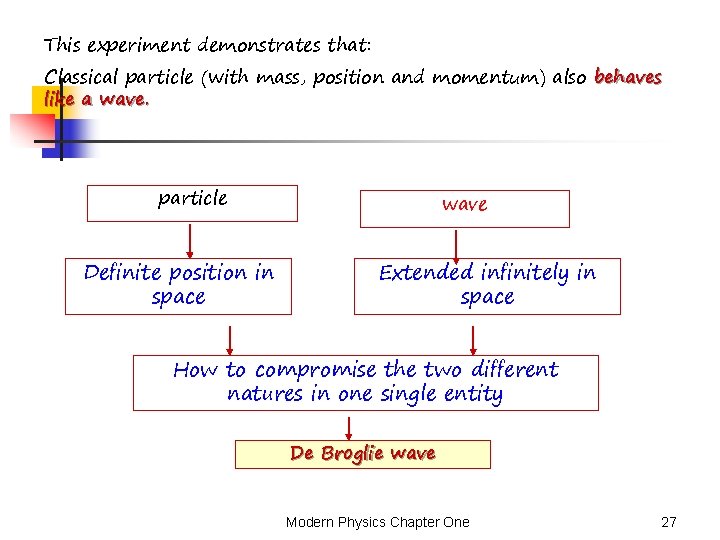
This experiment demonstrates that: Classical particle (with mass, position and momentum) also behaves like a wave. particle Definite position in space wave Extended infinitely in space How to compromise the two different natures in one single entity De Broglie wave Modern Physics Chapter One 27
- Slides: 27