Matter Waves 5 We were able to derive

Matter Waves 5 • We were able to derive the wave equations for waves in a string, a rod or for electromagnetic waves from a knowledge of the medium in which the waves propagated and of the propagating displacement itself. • In case, the velocity of the wave was closely linked to such physical parameters as tension, density, forces, spatial distributions, dielectric constant, and magnetic permeability • How to construct the wave equation for matter waves? (medium is not well defined) Ø We construct a formal equation that will meet very general requirements that matter often exhibits wavelike properties, such as electron diffraction. 1

The Schroedinger wave equation • What features must our wave equation include? (common feature between “particle” and “wave”) (5. 1) • If we assume that the matter wave equation has the harmonic solutions in one dimension as, Since, (5. 2) (Schroedinger time-dependent wave equation)

Schroedinger time-independent wave equation • If we are not concerned with the dynamic changes of energy state, but concerned with the question of what energy states are allowed in the presence of a particular potential energy V(x, y, z), so called stationary states of the system, the states can be expressed as (5. 3) • Substitution of the equation (5) into (4) leads to (5. 4) Schroedinger time-independent wave equation – E is constant, giving the allowed energy for a particular and V.

Schroedinger time-independent wave equation •

Free electron model of a confined electron “Particle in a box” • The Schrödinger equation becomes (5. 5) • The solution of the above equation has the form. (5. 6) • Substitution eq. (5. 6) into eq. (5. 5) results in 0 L

Free electron model of a confined electron • (5. 7) (5. 8) (5. 9) n: quantum number (5. 10) (note the similarity with (2. 31))

Free electron model of a confined electron • The appropriate wave functions for a particle in a one-dimensional box can be obtained, (5. 11) (5. 12) (5. 13)

Free electron model of a confined electron v. For large n (L in centimeters) when L=1 cm, E 1=3. 8 x 10 -15 e. V when L=0. 3 nm, E 1=4 e. V Wave Probability is function of x and varies with E Discrete En allowed ③ As wave picture reduces to classical picture. “correspondence principle”



• Since in either interpretation, is a probability, it must be “square integrable”. (5. 15) • When the wave function is multiplied by an appropriate constant A such that (5. 16) • The wave function is said to be normalized, and A is called the normalization constant. • A normalized wave function satisfies (5. 18)

• To be used as a probability, the wave function must be normalized. (5. 17) (5. 14) • In spherical coordinates (5. 19) • Three important systems allow exact solutions. ① A free electron model of a confined electron ② Linear harmonic oscillator (F=-gx) ③
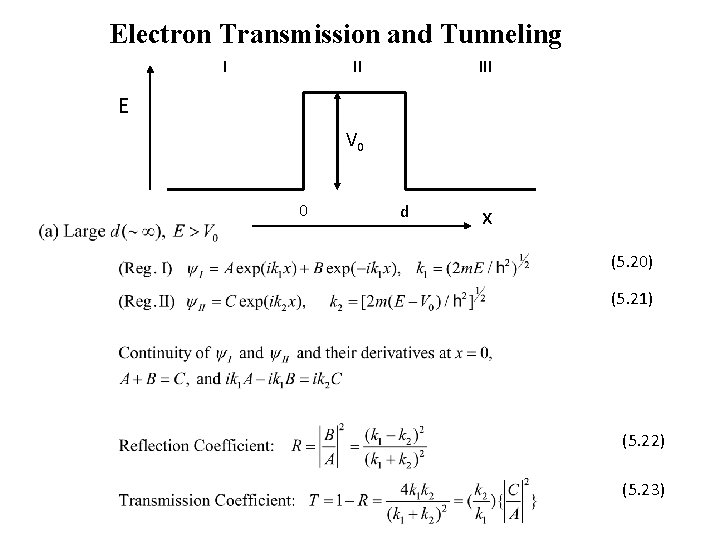
Electron Transmission and Tunneling I II III E V 0 0 d x (5. 20) (5. 21) (5. 22) (5. 23)
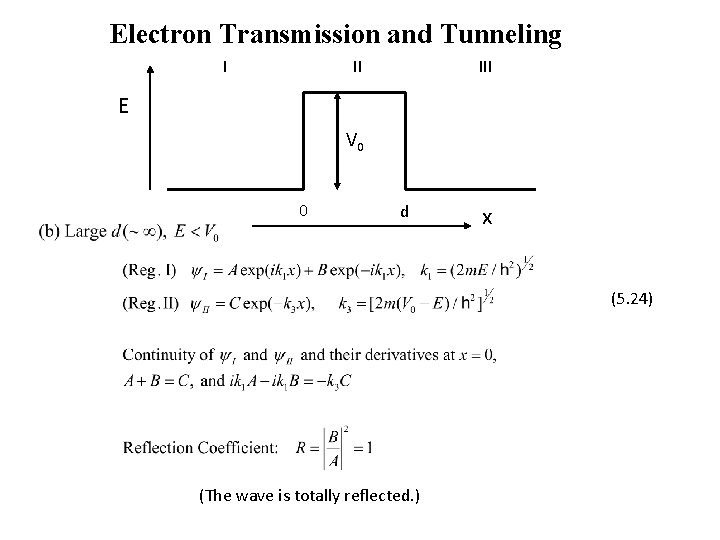
Electron Transmission and Tunneling I II III E V 0 0 d x (5. 24) (The wave is totally reflected. )
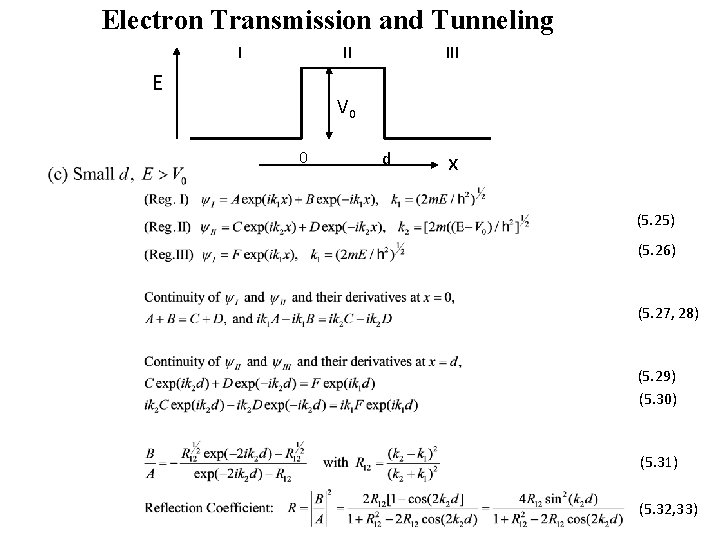
Electron Transmission and Tunneling I II E III V 0 0 d x (5. 25) (5. 26) (5. 27, 28) (5. 29) (5. 30) (5. 31) (5. 32, 33)
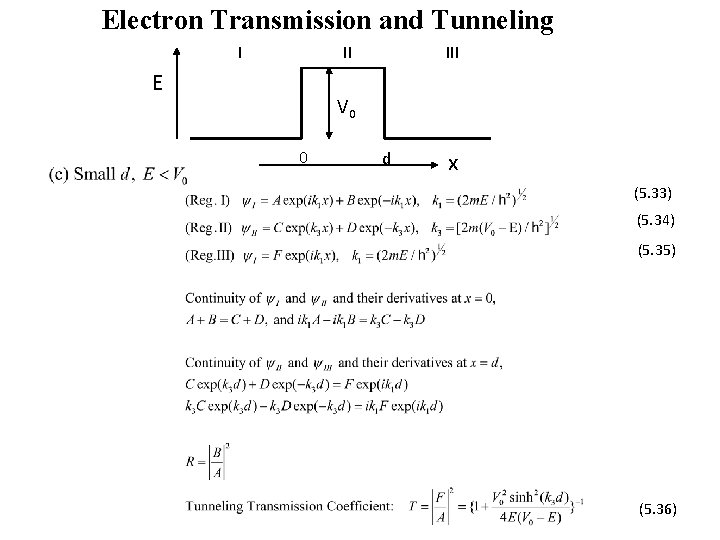
Electron Transmission and Tunneling I II E III V 0 0 d x (5. 33) (5. 34) (5. 35) (5. 36)

Linear harmonic oscillator Application of the wave picture to the harmonic oscillator leads to the conclusion of the quantization of energy into discrete amounts of wide significance for a variety of physical phenomena (5. 37) (5. 38, 39) (5. 40) (5. 41) f(x) is a polynomial in x that terminates after a finite number of terms in order to keep (x) finite and square integrable,

Linear harmonic oscillator -For the polynomial to terminate with the n-th power term, (5. 42) (5. 43) (5. 44) Wave motion can be represented as a collection of oscillators, each oscillator having one of the normal mode frequencies of the total system. Crystal vibrations and light can be described in terms of a classical wave formalism and also in terms of a quantum formalism. The quantum for crystal vibrations is called the phonon. The quantum for light is called the photon.

Linear Harmonic Oscillator-Phonon Statistics • Crystal vibrations: There are two kinds of “quantization”; The limitation of the allowed frequencies in a finite crystal to the normal modes by the boundary conditions. (classical atoms and classical waves; , only specific wavelengths, m 2 L, and their corresponding frequencies, m, are allowed) The limitation of the allowed, energies in one of specific mode of an oscillator. (quantum mechanical; we consider the individual atoms are now “particles” with wavelike properties. The energy in a vibration with a particular allowed mode (state), n, must be of the form Both limitations must be satisfied by the vibration in a crystal, i. e. ,

Linear Harmonic Oscillator-Phonon Statistics • Suppose that there are N different modes possible, and let the index m indicate the m-th mode with frequency wm. • What is the average number of phonons expected in lattice waves with frequency wm in equilibrium at a temperature T? energy corresponding to the n-th state of the m-th mode, i. e. the state with n phonons, each with energy of (1) Calculate the probability that there is a vibration with energy Emn and frequency wm at T. (2) Calculate the average energy for the m-th mode at T (3) Divide this average energy by the energy of single phonon (1)

Linear Harmonic Oscillator-Phonon Statistics (2) average energy of the mth mode (3) average number of phonon with ωm Applies to both phonons and photons
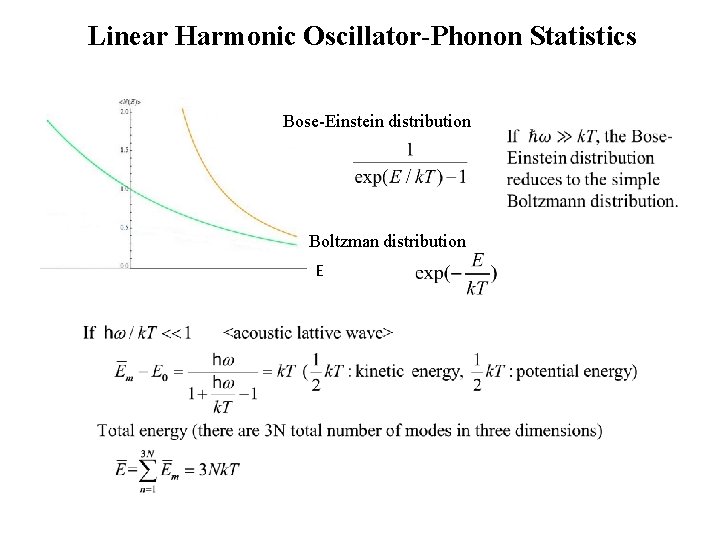
Linear Harmonic Oscillator-Phonon Statistics Bose-Einstein distribution Boltzman distribution E

The Hydrogen atom • A single electron bound to a single proton by a Coulomb attraction force. • An understanding of the solution for the hydrogen atom allow at least a qualitative understanding of elementary atomic structure. • The Schroedinger equation for the hydrogen atom is where the potential energy is given by For spherical coordinates,

The Hydrogen atom • The resulting differential equations for each of the variables are where A and B are the constants of separation • Angular parts [the equations for ( ) & (θ)] are independent of V(r)and are the same for all central force-field systems. : expressible in “spherical harmonics” and do not for the free hydrogen atom affect the allowed energies for the system. • There are three quantum numbers because there are three kinds of variables. Quantization is expected because an electron is confined physically.

The solutions of Φ(ϕ), Θ(θ) Φ(ϕ) In order for Φ(ϕ) to be single valued, Φ(ϕ+2π)= Φ(ϕ) Energy in a free hydrogen atom does not depend on ml The energy may depend on ml in the presence of magnetic field – Zeeman effect Θ(θ) - Involves polynomials in sinq and cosq - Θ(θ) to be finite, a polynomial must be terminated after a finite number of terms, which introduce another quantum number l
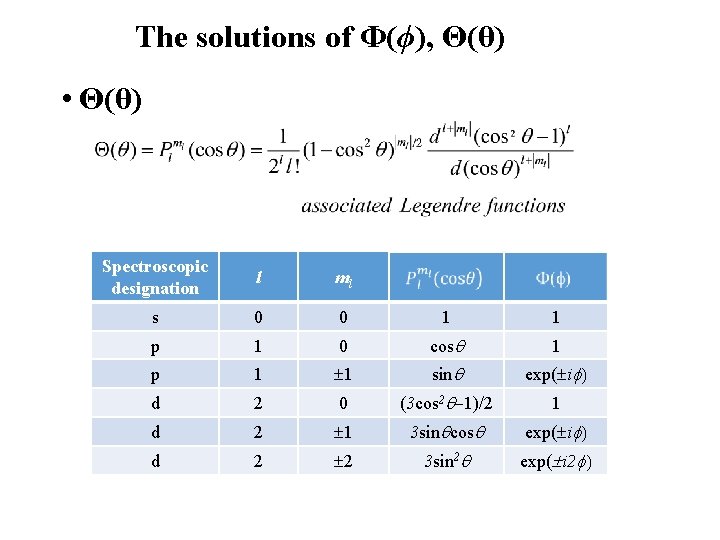
The solutions of Φ(ϕ), Θ(θ) • Θ(θ) Spectroscopic designation l ml s 0 0 1 1 p 1 0 cos 1 p 1 1 sin exp( i d 2 0 (3 cos 2 1)/2 1 d 2 1 3 sin cos exp( i d 2 2 3 sin 2 exp( i 2

The solution for R(r) • For R(r), the differential equation becomes • The solution of the above equation can once again be written in the form of a polynomial that must be terminated after a finite number of terms in order for R(r) to be finite and well behaved. • The termination conditions introduce third quantum n, so called the principal quantum number n, called the principal quantum number, because in the hydrogen atom the energy depends only on the value of n. (Bohr radius) Total solution:

Energy of the hydrogen atom • Allowed energies in the hydrogen atom • The ground state of the hydrogen atom corresponds to, • Spin: a kinds of intrinsic angular momentum called electron spin - spin quantum number - z component of spin momentum is The total wave function can then be designated by four quantum numbers:

Energy of the hydrogen atom (Lyman series)

Orbital of hydrogen atom (Variation of the radial wave function)
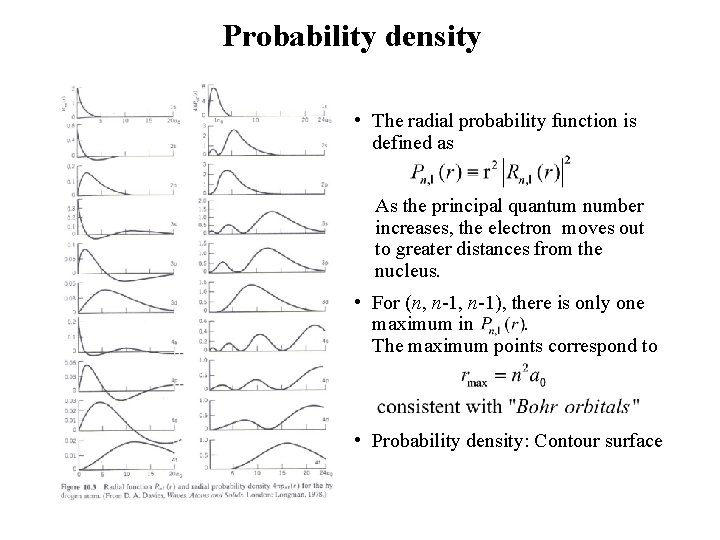
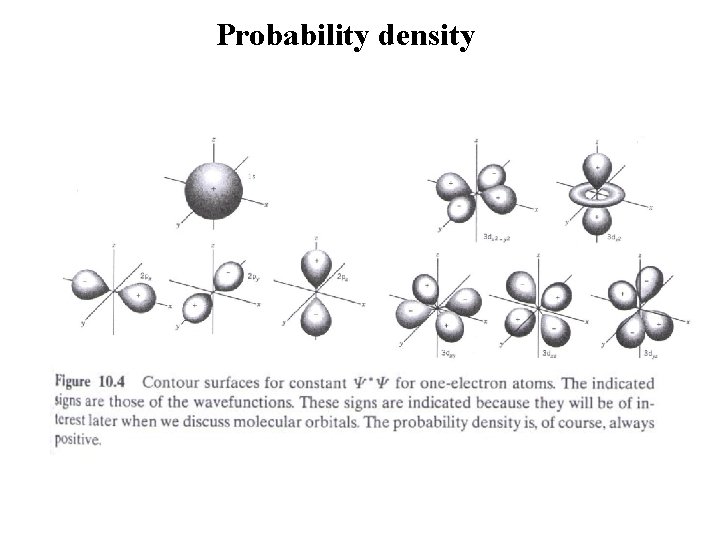
Probability density • The radial probability function is defined as As the principal quantum number increases, the electron moves out to greater distances from the nucleus. • For (n, n-1), there is only one maximum in . The maximum points correspond to • Probability density: Contour surface

Orbital of hydrogen atom ② 2 p state: p states have 3 orbital states. with spin, p states have 6 possible states. all the state have the same energy in hydrogen atom. State with different spatial distributions but with the same energy in said to as “degenerate”. A p state has a three fold degeneracy. The angular distribution for the p-state is given by (not a function of and is therefore symmetric about the z-axis. )
Probability density

The Periodic Table • (periodic table)

Angular momentum of the hydrogen atom • The angular momentum of a hydrogen atom can only have the values • The z component of the angular momentum is expressed in terms of the magnetic quantum number ml, - The possible orientations of the angular momentum vectors of a hydrogen atom with are
- Slides: 35