Matter PURE SUBSTANCES AND MIXTURES Matter Chemistry is

Matter PURE SUBSTANCES AND MIXTURES

Matter Chemistry is the study of the composition of matter and the changes matter undergoes. Matter – anything that has mass and occupies space Examples: Table You Air

Matter can be divided into two smaller subgroups: Pure substances Matter Mixtures Pure Substances

Pure Substances Pure substances are broken into two smaller subcategories: Matter COMPOUNDS ELEMENTS Mixtures Pure Substances Compounds Elements

Element - simplest form of matter that has a unique set of properties. Elements are composed of ONE type of atom or molecule. Elements cannot be broken down into smaller substances. They are what they are! Gold (Au), Silver (Ag), Carbon (C), Oxygen (O 2) – each individual element

Compound - substance that contains two or more elements chemically bound together in a fixed proportion. CO 2, H 2 O, C 6 H 12 O 6 – More than one element

Compounds can be broken down into their components (ELEMENTS) by CHEMICAL means, but NOT physical. C 6 H 12 O 6 (+ Heat) H 2 O + Carbon H 2 O (+ Electricity) H 2 + O 2 BOLD = COMPOUND The properties of compounds are quite different from those of their component elements. Matter Mixtures Pure Substances Compounds Elements Can be separated CHEMICALLY

Mixtures The second large subgroup of matter are mixtures. Mixtures are composed of two or more elements or compounds physically combined. Mixtures can be separated PHYSICALLY into their components (PURE SUBSTANCES). Examples: Sugar and Water, Pasta and Water, M&M’s & Skittles. Matter Mixtures Can be separated PHYSICALLY Compounds Pure Substances Elements

Mixtures Two different types of mixtures: Heterogeneous Matter Homogeneous Mixtures Heterogeneous Can be separated PHYSICALLY Homogeneous Pure Substances Compounds Elements Can be separated CHEMICALLY

Heterogeneous Mixtures Heterogeneous mixtures composition that is NOT uniform throughout. You can see things in there! Chicken Noodle soup, dirt from your backyard, Sand in water, Concrete You would be able to physically pick apart the components of a heterogeneous mixture

Homogeneous Mixtures Homogeneous mixtures uniform composition. Same composition of elements or compounds throughout. Also Air, called solutions. Salt in Water (OCEANS!), Sugar in Water (Kool-aid)

Matter PURE SUBSTANCES AND MIXTURES

Matter Chemistry is the study of the composition of matter and the ________________. _________ occupies space Examples: ______________ – anything that has mass and
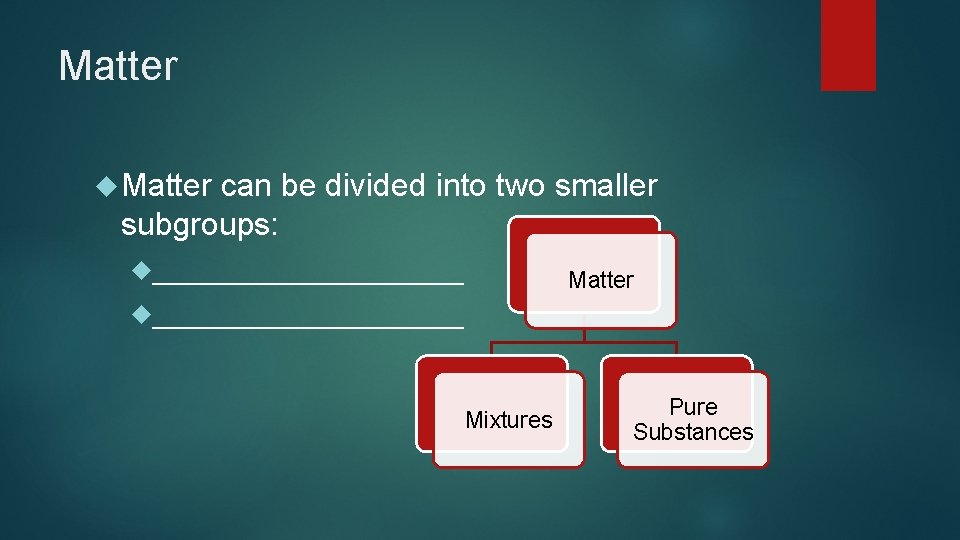
Matter can be divided into two smaller subgroups: __________ Matter __________ Mixtures Pure Substances

Pure Substances Pure substances are broken into two smaller subcategories: Matter __________ Mixtures Pure Substances Compounds Elements

Element __________ - simplest form of matter that has a unique set of properties. Elements are _____________________ _. Elements _____________________. They are what they are! Gold (Au), Silver (Ag), Carbon (C), Oxygen (O 2) –

Compound __________ - substance that contains two or more elements chemically bound together in a fixed proportion. CO 2, H 2 O, C 6 H 12 O 6 – More than one element

Compounds can be broken down into their components (ELEMENTS) by __________ means, but __________. C 6 H 12 O 6 H 2 O (+ Heat) H 2 O + Carbon (+ Electricity) H 2 + O 2 Matter Mixtures Pure Substances BOLD = COMPOUND Compounds The properties of __________ are quite different from those of their component __________. Elements Can be separated CHEMICALLY

Mixtures The second large subgroup of matter are mixtures. _________________________________ ______. Mixtures can be separated PHYSICALLY into their components (PURE SUBSTANCES). Examples: Sugar and Water, Pasta and Water, M&M’s & Skittles. Matter Mixtures Can be separated PHYSICALLY Compounds Pure Substances Elements

Mixtures Two different types of mixtures: __________ Matter __________ Mixtures Heterogeneous Can be separated PHYSICALLY Homogeneous Pure Substances Compounds Elements Can be separated CHEMICALLY

Heterogeneous Mixtures __________- composition that is NOT uniform throughout. You can see things in there! Chicken Noodle soup, dirt from your backyard, Sand in water, Concrete You would be able to physically pick apart the components of a heterogeneous mixture

Homogeneous Mixtures Homogeneous mixtures __________. Same composition of elements or compounds throughout. Also Air, called solutions. Salt in Water (OCEANS!), Sugar in Water (Kool-aid)
- Slides: 22