Matter PROPERTIES AND CHANGES CHAPTER 2 Four Phases
























- Slides: 27

Matter PROPERTIES AND CHANGES CHAPTER 2

Four Phases of Matter Solids 1. Have definite shape and volume Have strong intermolecular forces between particles. Therefore, the particles are very close to each other Usually the most dense phase In most cases incompressible

Four Phases of Matter Liquid 2. Liquids Have definite volume Takes the shape of the container “flow” and are incompressible The intermolecular forces are intermediate Weaker than solids, but stronger than gases � Therefore the densities are average �

Four Phases of Matter Gases �Gases Have no definite shape volume( dependent upon the container they fill) Very weak intermolecular forces so they have very low densities They are compressible and gases “diffuse”

Four Phases of Matter Plasma �Plasma Energy is very high due to extremely high temperatures. Atoms are stripped of their electrons They are “ionized” Moves quickly and unevenly Examples � Sun and lightening

Classification of Matter �Matter Anything that has mass and takes up space. �Pure substance Has a constant, invariable composition It’s identified by a formula or a symbol �Examples Water Helium

Classification of Matter �Compound Composed of two or more elements that cannot be decomposed by physical means Chemically combined Examples � Sodium chloride � Carbon dioxide

Classification of Matter �Element Simple substance that cannot be decomposed into simpler substances by any chemical change.

Classification of Matter �Mixtures Composed of 2 or more components that can be separated by physical means There are no chemical bonds between the separate parts The components or parts can vary in composition 2 types of mixtures

Classification of Matter �Homogeneous Mixture Sometimes called solutions Are evenly mixed Appears as one component even though there are 2 or more parts � Examples Plain jello Air Salt water

Types of mixtures �Heterogeneous mixture Unevenly mixed Can see separate components Examples � Dirt � Concrete � Muddy water

Heterogeneous Mixtures Colloid vs. Suspensions � 1) Colloids Particles are very small Do not settle out of solution Examples � Fog � Smoke � Whipped cream �Suspensions Particles are large Settle out of solution � Example: muddy water


Physical Separation of Mixtures Technique � 1. Filtration-to separate by size Use filter paper and funnel Used for homogeneous mixtures Ex. Muddy water

Physical Separation of Mixtures Technique � 2. Evaporation-boiling off liquid Used for homogeneous solutions Example: sea water � 3. Decant-to pour off liquid Used for homogeneous mixture

Physical Separation of Mixtures Technique � 4. Distillation-separate a mixture of liquids by boiling points How is this possible? � 5. Chromatography-to separate pigments from a homogenous solution Pigments separate because of difference in 1. 2. Mass Absorption rate

Physical Separation of Mixtures Technique � 6. Crystallization Formation of solid particles of a substance from a solution containing the dissolved substance. What does that mean? Example � Dissolve sugar into water � Place a string into solution � What forms around the string?


Conservation Laws �Law of Conservation of Matter is neither created or destroyed In reaction = � Amounts of reactants = amount of products made �Law of Conservation of Energy is neither created or destroyed BUT is rater it is transferred from one form to another

Physical Properties that don’t involve a change in the chemical identity of the substance Can measure these properties/characteristics without changing the identity Examples: � Melting point � Boiling point � Density � Electrical conductivity � solubility

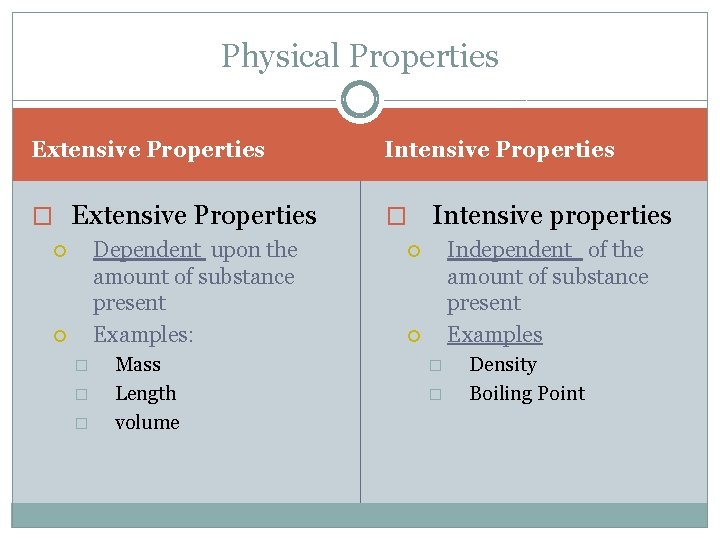
Physical Properties Extensive Properties Intensive Properties � Extensive Properties � Dependent upon the amount of substance present Examples: � � � Mass Length volume Intensive properties Independent of the amount of substance present Examples � � Density Boiling Point

Chemical Properties �Chemical Properties Refers to the way a substance undergoes a chemical reaction to form a new product. When you measure this property (characteristic) at least one new product is formed. �Examples: the ability to rot, rust, burn, ferment, grow, decay, sprout, combust

Physical Change �Physical change A process that will alter a substance’s appearance without changing its composition or idenity. �Any change that describes a 1. Phase Change � 2. Size Change � 3. Melting, freezing, evaporating, condensing Tearing, breaking, expanding, dissolving Shape Change � Hammering, stretching

Chemical Change �Chemical change A process that occurs that will change the identity and composition of the reactants �Examples: Burning, rusting, Exploding

Difference between properties and changes Properties Changes �Measure a process characteristic or trait.

Observations that Determine a Chemical change in a Lab �Gas is released Bubbles in liquids, smoke, odor �Heat is absorbed or released �Precipitate (solid) is formed in a homogeneous solution Start as a liquid, then you see “things” floating or settled on the bottom

Observations that Determine a Chemical change in a Lab �Definite color change occur Color is a ______ property The process of changing colors is a chemical change �Flash of light is released.