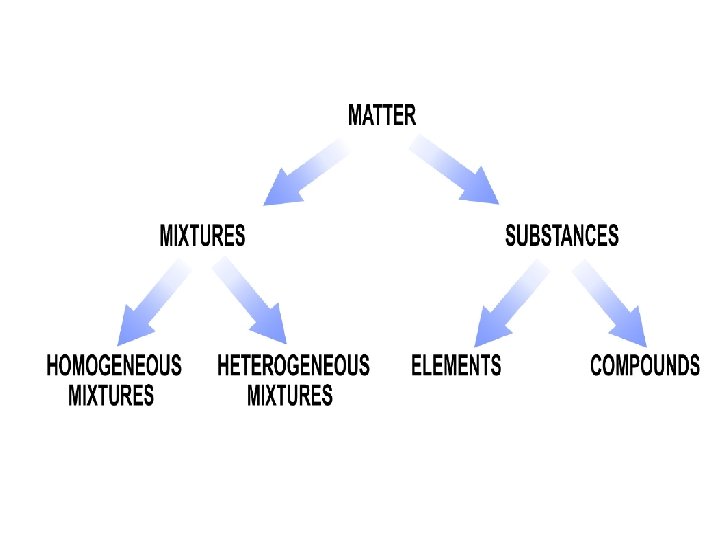
Matter Notes Matter Mixtures 2 Types of Mixtures













- Slides: 16

Matter Notes Matter: Mixtures: 2 Types of Mixtures 1. Homogeneous Mixture 2. Heterogeneous Mixture Separating Mixtures Substances: 2 Types of Substances: 1. Elements 2. Compounds Summary Explanation Illustration

Chapter 16 Section 1


Matter • Matter is used to describe anything that has mass and takes up space. • Matter is characterized (described) by different characteristics (ex. Boiling point, Melting point, hardness, elasticity)



Mixtures • Mixtures contain more than one type of matter • Two Types of Mixtures: – Homogeneous Mixtures – Heterogeneous Mixtures

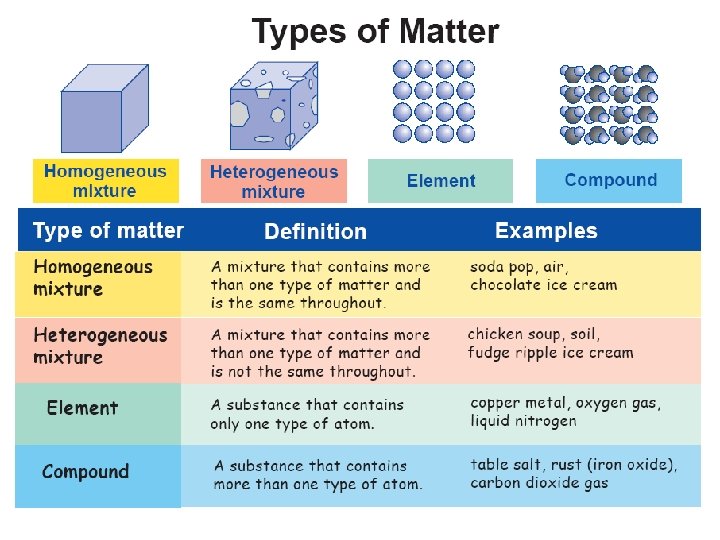
Homogeneous Mixtures • A mixture that is the same throughout. (In other words, all samples of a homogeneous mixture are the same. ) • Ex. Unopened Soda, Brass

Heterogeneous Mixtures • A mixture that is not the same throughout. (In other words, each sample of a heterogeneous mixture is not the same. ) • Ex. Chicken Noodle Soup, Snack Mix

Separating Mixtures • All mixtures can be separated into different types of matter by physical means such as sorting, filtering, heating or cooling. • Physical separation does not change the characteristics of each component. • Ex. Pulling out all of the pretzels from the Snack Mix


Substances • Substances are types of matter that cannot be separated by physical means. • The two types of substances are: –Elements –Compounds

Elements • Elements contain only one kind of matter. • Elements can be found on the periodic table of elements. • Ex. Silver

Compounds • Compounds contain more than one kind of matter. • The different kind of matter found in the compound cannot be separated without changing the substance.


Separating mixtures into substances is a very important part of scientific work. Medical researchers try to isolate the substances in plants that may help heal diseases. Forensic scientists try to match evidence from the scene of a crime with substances found with a suspect. Nutritionists evaluate the amount of carbohydrates, fats, proteins, vitamins, and minerals in various foods.