Matter Measurements Claculations Classification Properties Measurements Significant Figures















- Slides: 16

Matter, Measurements, Claculations Classification, Properties, Measurements, Significant Figures, Matrix (Dimensional Analysis)

Chemistry Study of properties and behavior of matter.

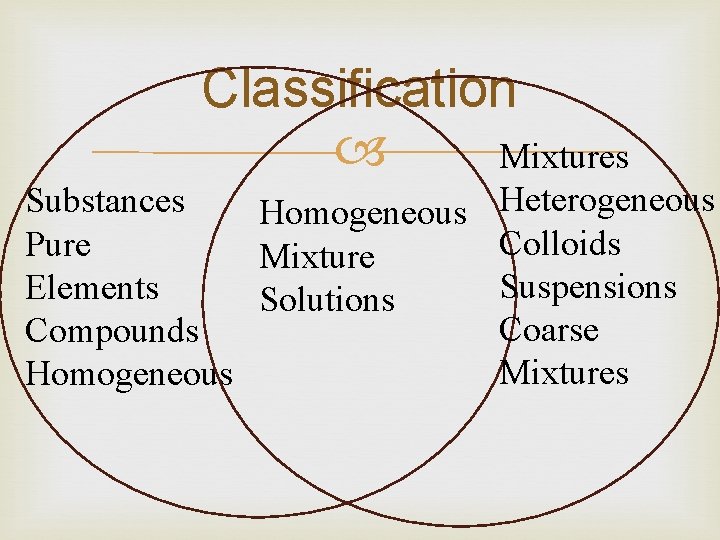
Classification Substances and mixtures

Classification Mixtures Heterogeneous Substances Homogeneous Colloids Pure Mixture Suspensions Elements Solutions Coarse Compounds Mixtures Homogeneous

How do you separate mixtures? Come up with at least five ways to separate different mixtures.

Properties - Intensive Physical Properties - Not dependent on the amount of matter - Temperature and melting point - Extensive Physical Properties - Dependent on the amount of matter - Mass and volume - Chemical Properties - Observable through a reaction

Differences between Theory and Scientific Law. - Theory - An explanation of a model that has predictive properties (use the scientific method) - Must be consistent with other theories - Scientific Law - A concise verbal statement or mathematical equation that summarizes a broad range of observations and experiences

Measurements are not exact. The last number is estimated. Since they are inexact, some uncertainty exists in all measurements.

Know the Base SI Units! Review the most common base units and the prefixes.

Temperature Physical property that determines the direction of heat flow.

Definitions - Definitions or counted numbers are exact. - There is no uncertainty in these numbers - There are 24 students in this class - That’s counted…. it has no uncertainty

Accuracy How close individual measurements agree with the “accepted” value.

Precision How close the measurements are to other measurements. Precise measurements have little “deviation” from each other. Some call this as having a small uncertainty from the average of the measurements.

Significant Figures They would not be called significant if they did not matter. Yes, they matter. Relearn what you had learned.

The Matrix Trust me. Chemistry was easier when you used this method. It will be easier in AP Chemistry if you keep using it. Do not be like Bilbo Baggins and disobey Gandalf in the Mirkwood Forest!

What are you doing standing around?