Matter Matter Stuff that objects are made of

Matter

Matter • Stuff that objects are made of • Anything that has mass and volume • 3 Types – Elements – Compounds – Mixtures

Phases of Matter • Solids • Liquids • Gases • Animation

Solids • • Definite Shape and Volume Strong Intermolecular Forces Particles are close together Particles are arranged in neat, orderly lines • Particles do move – shake, vibrate – Animation • High melting and boiling points • Designated by (s) Examples: – Au(s), Fe(s) , H 2 O(s)
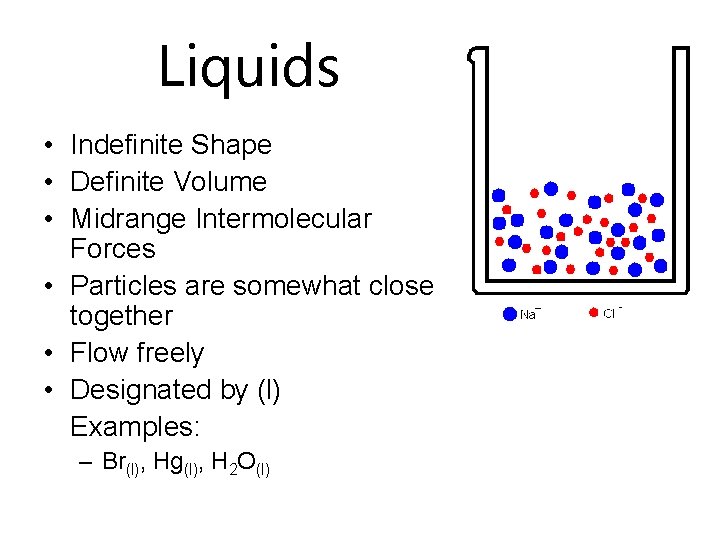
Liquids • Indefinite Shape • Definite Volume • Midrange Intermolecular Forces • Particles are somewhat close together • Flow freely • Designated by (l) Examples: – Br(l), Hg(l), H 2 O(l)

Gases • NO definite Shape or Volume – Particles will spread out to fill the shape and volume of a container • • • Weak Intermolecular Forces Particles are far apart Particles are very disorganized Particles quickly move all over Low melting and boiling points Designated by (g) Examples: – Ne(g), O 2(g) , H 2 O(g)
Melting and Boiling Points • Use Reference Table S to find the melting and boiling points of a solid, a liquid, and a gas Element Solid Liquid Gas Melting Point Boiling Point

• Write a general statement about how boiling point relates to phase.

Physical Properties – Characteristics that can be observed without changing the identity of the substance (no new substance is formed) • Examples: Color, Hardness, melting point Physical Changes – Changes that occur without changing the identity of the substance • Examples: Cutting, Ripping, Phase Changes

Chemical Properties – Characteristics that can. NOT be observed without changing the identity of the substance (a new substance is formed) • Examples: Reactivity, Flammability Chemical Changes – A change that results in a new substance • Examples: Burning, Rusting • Physical/Chemical Change Song

Physical/Chemical Examples Indicate if each of the following is physical or chemical 1. 2. 3. 4. 5. 6. 7. 8. The boiling point of water is 100 o. C. Antacids will neutralize stomach acid. Diamonds will cut glass. Most metals conduct electricity. The density of NH 3 is 0. 77 g/L. Solution A and B are reacted together and a precipitate (solid) is formed. 2 H 2(g) + O 2(g) → 2 H 2 O(l) → H 2 O(g)

Classification of Matter § All matter can be classified as one of the following: – Elements – Compounds – Mixtures

§ All matter can be described as: – Homogeneous – uniform throughout – Heterogeneous – different throughout § Classify the following as homogeneous or heterogeneous 1. 2. 3. 4. 5. 6. A glass of water Italian salad dressing Turkey and gravy Milk Orange juice with pulp Orange juice without pulp

Substances Ø Type of Matter – Element, Compound Ø All samples have identical properties and composition l They are homogeneous

Elements n n n Composed entirely of atoms with the same atomic number Cannot be decomposed Found on the Periodic Table Begin with a capital letter Homogeneous Examples: Cu, Fe, O 2, Ne – Diatomic Element = contains two of the same atoms (O 2, H 2)

Compounds n n n Composed of at least 2 different elements Elements are chemically combined (bonded) Homogeneous

Compounds n Can be decomposed (by chemical means) into 2 or more elements – Electrolysis, Reactions n n The properties of the component elements are usually very different from the properties of the compound Combined in a definite ratio – In Sodium Chloride (Na. Cl) the Na and Cl must combine in a 1: 1 ratio n Examples: Mg. O, H 2 O, Na. Cl – Binary Compound = contains 2 different elements
• Compare and contrast elements and compounds. Elements Both Compounds

Mixtures n n Combination of 2 or more substances No chemical change occurs (No bond is formed)

Mixtures Parts retain their individual properties n Can be separated by physical means n – Filtering, Magnetism, Distillation, Density n Can combine in varying ratios – The amount of salt in salt water can vary

Mixtures n Can be heterogeneous – Concrete, Dirt, Chocolate Chip Cookie n Can be homogeneous – Gases (Air) – Aqueous Solutions – a solution made with water n Na. Cl(aq) = salt is dissolved in water

Law of Conservation of Matter • Matter cannot be created or destroyed

Law of Conservation of Mass • Mass cannot be created or destroyed • The mass before a reaction must be equal to the mass after the reaction

Law of Conservation of Energy • Energy cannot be created or destroyed • The amount of energy remains constant, however it may transfer forms http: //www. youtube. com/watch? v=uy. N 9 y 0 BEMqc

Exothermic Reactions • Heat is released • Heat is a product • Surroundings will feel warm

Endothermic Reactions • Heat is absorbed • Heat is a reactant • Surroundings will feel cold
- Slides: 26