MATTER MATTER n n Anything that has mass
























































- Slides: 57

MATTER

MATTER n n Anything that has mass and volume (made up of “stuff” and takes up space) Exists in 4 states (phases) 1. 2. 3. 4. Solids Liquids Gases Plasmas

SOLID n Has a definite shape and a definite volume n Tightly packed particles

LIQUID n n n Has a definite volume but NO definite shape (takes the shape of its container) Particles are not held together as tightly Particles are free to move (flow around each other)


GAS n No definite shape or volume n Can move freely (expand) without any limits

PLASMA n High energy phase n Least common on Earth n Most common phase in the rest of the universe

MATTER MOVIE http: //www. abpischools. org. uk/page/modules/s olids-liquidsgases/slg 2. cfm? co. Site. Navigation_all. Topic=1

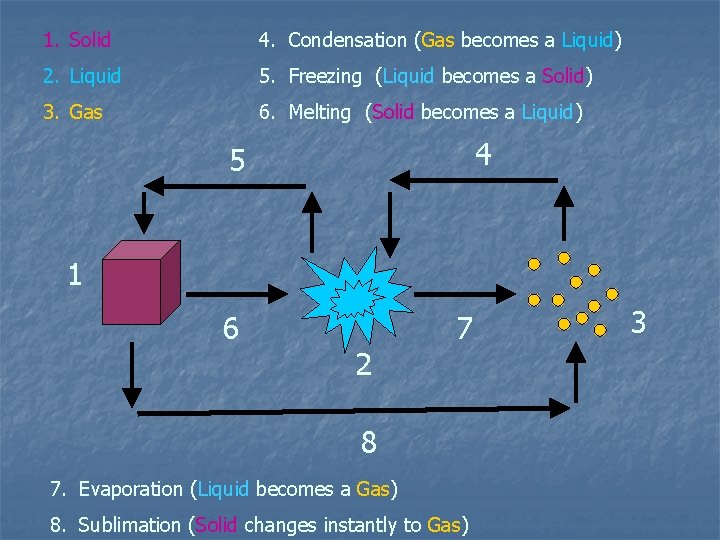
1. Solid 4. Condensation (Gas becomes a Liquid) 2. Liquid 5. Freezing (Liquid becomes a Solid) 3. Gas 6. Melting (Solid becomes a Liquid) 4 5 1 6 2 7 8 7. Evaporation (Liquid becomes a Gas) 8. Sublimation (Solid changes instantly to Gas) 3

Elements Mixtures & Compounds

Elements n n Substances that can’t be broken down into other substances Periodic Table n n Elements are listed on a chart in order of their characteristics Chemical Symbol 1 or 2 letters that stand for an element n Usually, but not always an abbreviation of the elements name n

Examples of Chemical Symbols Oxygen – O Helium – He Gold – Au Iron - Fe

Mixtures n Combination of 2 or more substances in which the substances keep their own properties n NOT chemically combined n NO definite amount of each ingredient n Examples: Milk, Kool-Aid, Pudding, Raisin Bran, Chocolate Chip Cookie Dough

Compounds n n 2 or more elements that have been chemically combined Has new properties that are different from the properties of the elements it’s made of (new substance) n Can be broken down by chemical means n Has a definite amount of each element

n Molecule n n Compounds Smallest part of a compound (can’t be any smaller unless it is broken apart into separate elements) Chemical Formula Used to show what elements and how many make up a compound n Examples: Na. Cl (sodium chloride) H 2 O (water) n Sodium Chlorine 2 Hydrogen Oxygen

Common Elements and Compounds in Earth Science Hydrogen Helium Carbon Nitrogen Oxygen Sodium Fluorine Aluminum Silicon Sulfur Chlorine Calcium Iron Nickel Magnesium Iridium H He C N O Na F Al Si S Cl Ca Fe Ni Mg Ir Lead Pb Radon Rn Uranium U Nitrate NO 3 Carbon Monoxide CO Carbon Dioxide CO 2 Cloro. Fluoro. Carbons CFC’s Ozone O 3 Water H 2 O Methane CH 4 Ammonia NH 3 Sulfuric Acid H 2 SO 4 Iron Oxide (Rust) Fe. O Calcium Carbonate (Limestone) Ca. CO 3 Silicon Dioxide (Quartz) Si. O 2 Sodium Chloride (Salt) Na. Cl

Elements Foldable Materials • • 1 Element Foldable 1 Pen/Pencil Scissors Colored Pencils: Red, Green, Yellow, Blue

STEP 1 Fold your foldable in ½. After folding in ½, your foldable should look like this

STEP 2 Open your foldable and cut ONLY on the dotted lines! DO NOT cut while folded!

STEP 3 Write in the information on the inside of your foldable as the information is shown in the next slides. H • Write in the symbol (use your colored pencils for this part) • Color Code the front (with the same color you used for the element symbol, make a star or shade in the area next to the element name) • Write in where the element is found (this will be shown in the ‘common example’ or in the ‘earth science connection’)

Hydrogen Common Example Hydrogen (H) Bomb Lightest Element H Earth Science Connection Hydrogen Fuel Most Common Element in Stars

Helium Common Example He Earth Science Connection Produced in the Sun Gas for Floating Objects

Carbon Common Example C Earth Science Connection Oil Coal Carbon Dating Natural Gas Coal Power

Nitrogen Common Example Dry Ice N Earth Science Connection 78% of Earth’s Atmosphere

Oxygen Common Example O Earth Science Connection Gas Used by Humans to Release Energy 21% of Earth’s Atmosphere

Sodium Common Example Element in Table Salt (Sodium Chloride) (Natrium) Na Earth Science Connection 6 th Most Common Element on Earth Part of the Ocean (Salt) Water

Fluorine Common Example Used to Strengthen Tooth Enamel F Earth Science Connection Part of a Cooling Agent Found in Older Appliances Negatively Affects Our Atmosphere

Aluminum Common Example Used in Thousands of Household Products Like Pop Cans and Sinks Al Earth Science Connection One of the Most Common Elements in Earth’s Crust

Silicon Common Example Si Earth Science Connection Most Common Mineral on Earth Used to Make Thousands of Products Like Kitchen Utensils, WII Remote Covers, Calk and Plastic Bracelets

Sulfur Common Example Found in Many Products Like Matches, Gunpowder and Fertilizer Rotten Egg Smell S Earth Science Connection Released in Volcanic Eruptions and Hot Springs Negatively Affects Our Atmosphere

Common Example Chlorine Cl Earth Science Connection Part of a Cooling Agent Found in Older Appliances Used to Disinfect Water Negatively Affects Our Atmosphere Element in Table Salt (Sodium Chloride) Part of the Ocean (Salt) Water

Common Example Calcium Ca Earth Science Connection Used by Our Bodies to Build Bones and Regulate Heartbeat Element in Limestone, the Most Common Rock on Earth

Iron Common Example Used in Many Metal Products Carries Oxygen Through Our Bodies (Ferrum) Fe Earth Science Connection Element in the Earth’s Core and Crust Magnetic

Common Example Used in Many Metal Products Element in Some Batteries Nickel Ni Earth Science Connection Element in the Earth’s Core

Magnesium Common Example Used By Our Bodies to Perform Over 300 Reactions in the Body Like Helping With Muscle Contraction Used in Many Metal Products Mg Earth Science Connection Common Element in the Earth’s Crust

Iridium Common Example Ir Earth Science Connection Used to Strengthen Platinum Comes From Asteroids and Comets

Lead (Plumbum) Common Example Pb Earth Science Connection Protects Against Radiation Used in Many Products Like Fishing Lures and Glass Pollutes Water and Poisonous if Ingested

Radon Common Example Sometimes Used for Cancer Radiation Treatments Rn Earth Science Connection Given Off Naturally By Bedrock Dangerous to Animals

Common Example Used to Create Nuclear Power Uranium U Earth Science Connection Common in Small Amounts in All Rocks

Compounds Foldable Materials • 1 Compound Foldable • 1 Pen/Pencil • Scissors

STEP 1 Fold your foldable in ½. After folding in ½, your foldable should look like this

STEP 2 Open your foldable and cut ONLY on the dotted lines! DO NOT cut while folded!

STEP 3 Write in the information on the inside of your foldable as the information is shown in the next slides. • Write in the symbol • Write in where the compound is found (these possibilities will be in PINK)

Color Coding n You may be using multiple colors on each compound to color code. Similar to what we used earlier, but you’re looking at the entire name. - GREEN- Only the FIRST letter is the symbol - YELLOW- The FIRST TWO letters is the symbol - RED- The Latin/Greek/Other name is abbreviated. - BLUE- The FIRST letter and another letter are used. n EXAMPLE: n n Carbon Monoxide- CO n Color code the SYMBOL ONLY. n

Carbon Monoxide CO Found in Automobile Exhaust Contains (1 atom) Oxygen Mon = 1 Contains Carbon Pollutes Our Atmosphere Colorless, Odorless, Tasteless, but highly toxic!

Ammonia Household Cleaner Compound in Animal Waste Pollutes Rivers and Lakes From Field Runoff, but Plants Need it to Grow NH 3 Contains Nitrogen Contains (3 atoms) of Hydrogen

Iron Oxide Rust Fe. O Contains Iron (Fe) Contains Oxygen Common Name is Rust Compound That Makes Dirt Red in Color

Ozone Layer in the Atmosphere Protects Us From Ultraviolet (UV) Radiation Given Off By Car Exhaust and a Pollutant at Ground Level O 3 Contains (3 atoms) of Oxygen

Carbon Dioxide Exhaled by Animals Given Off by the Burning of Fossil Fuels CO 2 Plants Use CO 2 Contains Carbon Contains (2 atoms) of Oxygen Di = 2 Too Much in the Atmosphere Causes Climate Change

Silicon Dioxide Si. O 2 Quartz: Most Common Mineral on Earth Contains Silicon Contains (2 atoms) of Oxygen Di = 2 Common Name is Quartz

Nitrate Pollutant Found in Animal Waste NO 3 Contains Nitrogen Pollutes Lakes and Rivers Contains (3 atoms) of Oxygen

Water Compound Needed by All Life H 2 O Contains (2 atoms) of Hydrogen Contains Oxygen Makes Up 75% of the Earth’s Surface

Sulfuric Acid Enters Our Atmosphere Through Automobiles and Factories H 2 SO 4 Contains (2 atoms) of Hydrogen Contains Sulfur Contains (4 atoms) of Oxygen Falls Back to Earth as Acid Rain

Chlorofluorocarbons CFC’s Used in the 1950’s 1990’s as a coolant and to make products such as styrofoam Destroys the earth’s Ozone layer

Calcium Carbonate Ca. CO 3 Contains Calcium Contains Carbon Contains (3 atoms) Oxygen Common Name is Limestone

Methane Too Much Pollutes the Atmosphere CH 4 Contains Carbon Contains (4 atoms) Hydrogen Natural Gas Given Off by the Earth and Animals

Sodium Chloride Na. Cl Contains Sodium (Na) Contains Chlorine Common Name is Table Salt