Matter can change phases permanently or temporarily Temporary
















- Slides: 18

Matter can change phases permanently or temporarily. • Temporary changes are called PHYSICAL changes. • Permanent changes are called CHEMICAL changes.

Physical Changes

Structure of water Structure of ice Structure of steam

Physical Changes When a physical property (like the phase of water) is altered we called it a PHYSICAL CHANGE In other words: A physical change involves a change in physical properties (shape, size, density, etc. ) It takes a physical change to change water to steam, or ice to liquid.

Physical Changes • Physical Changes: only the phase changes, the substance does not. • Physical changes usually change the size or shape of the substance.

Physical Changes Examples of physical changes include: Melting Point The ice cream is melted but it is still ice cream. It still tastes like ice cream.

Physical Changes Examples of physical changes include: Shape The shape of the wood changes when it is made into a desk, but it is still wood.

Physical Changes Examples of physical changes include: Freezing Point The glass of water is frozen, but it is still water (H 20)

Physical Changes can be caused by changes in energy.

Physical Changes can be caused by changes in energy.

chemical Changes

Chemical Changes Chemical Change: a change that creates a NEW material. (A change in chemical properties) The original materials are changed into something different. Typically irreversible

Chemical Changes Common examples of chemical changes that you may be somewhat familiar with are; digestion, respiration, photosynthesis, burning, and decomposition.

Chemical Changes Examples of chemical changes include: burning Once the wood is burned, it is changed forever.

Chemical Changes Examples of chemical changes include: rusting The rusty spoon cannot be changed back.

Chemical Changes Examples of chemical changes include: cooking Once the egg is fried, it can not be changed back to the raw egg.

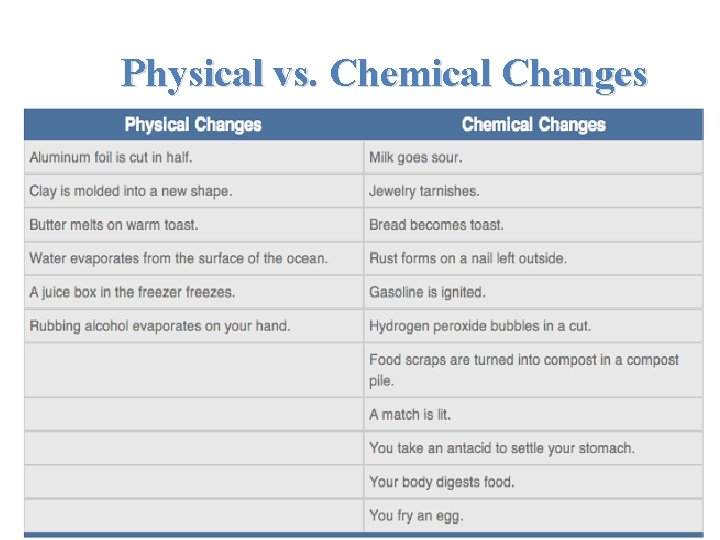
Physical vs. Chemical Changes

Let’s Summarize • All matter has physical properties and chemical properties. • Physical properties can change, but the structure of the matter does NOT change • Chemical properties lead the way to chemical changes. • Chemical changes make new substances.