MATTER BASICS Review Matter The Elements Matter is

MATTER BASICS Review

Matter & The Elements • Matter is anything that has mass and occupies space. • All matter is made up of elements • 118 elements, 92 exist in nature

Matter & The Elements • There are 6 elements you need to know for biology: • C – Carbon • H – Hydrogen • O – Oxygen • N – Nitrogen • P – Phosphorus • S – Sulfur
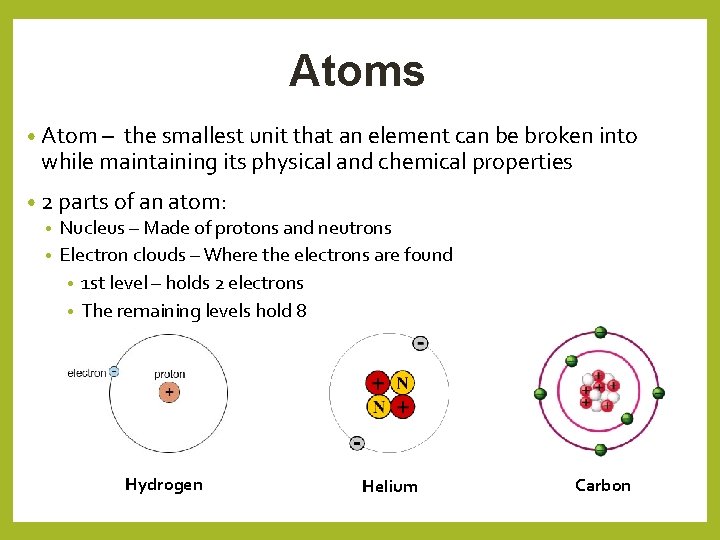
Atoms • Atom – the smallest unit that an element can be broken into while maintaining its physical and chemical properties • 2 parts of an atom: • Nucleus – Made of protons and neutrons • Electron clouds – Where the electrons are found • 1 st level – holds 2 electrons • The remaining levels hold 8 Hydrogen Helium Carbon
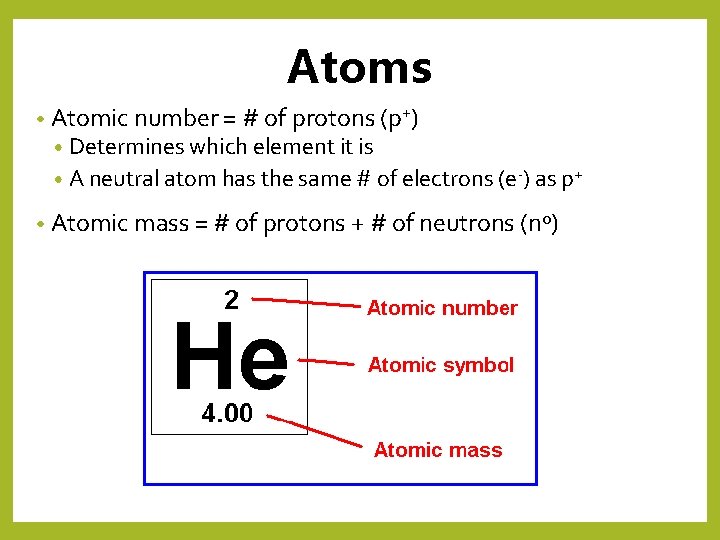
Atoms • Atomic number = # of protons (p+) • Determines which element it is • A neutral atom has the same # of electrons (e-) as p+ • Atomic mass = # of protons + # of neutrons (n 0)
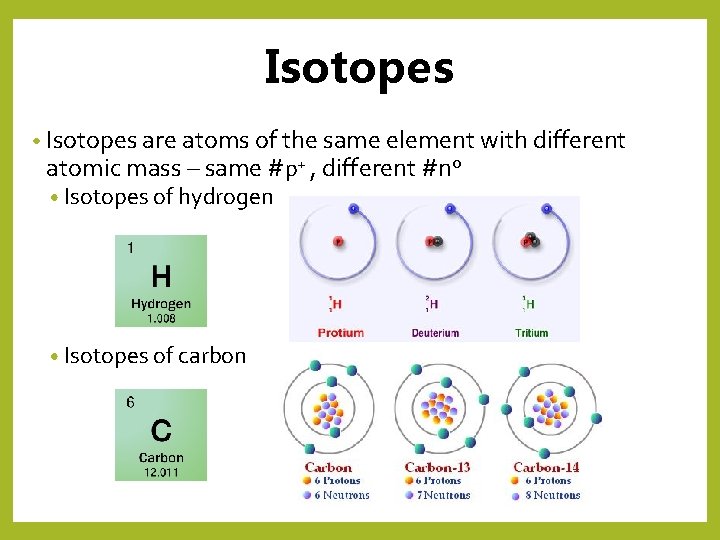
Isotopes • Isotopes are atoms of the same element with different atomic mass – same #p+ , different #n 0 • Isotopes of hydrogen • Isotopes of carbon
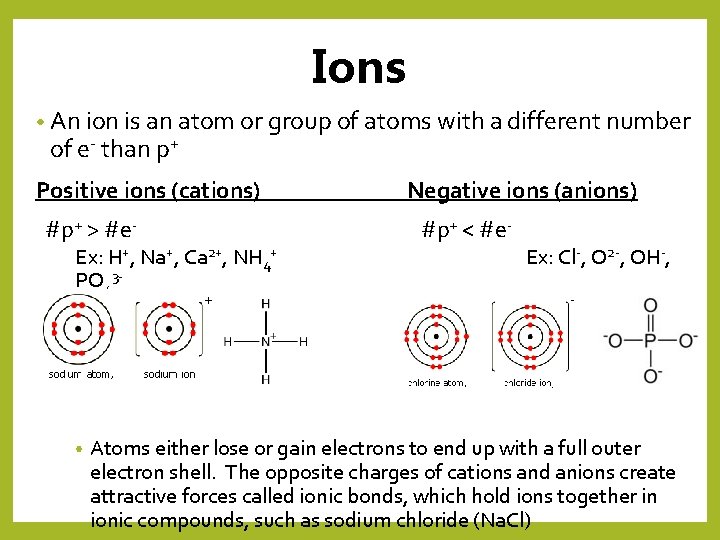
Ions • An ion is an atom or group of atoms with a different number of e- than p+ Positive ions (cations) #p+ > #e- Ex: H+, Na+, Ca 2+, NH 4+ PO 43 - • Negative ions (anions) #p+ < #e- Ex: Cl-, O 2 -, OH-, Atoms either lose or gain electrons to end up with a full outer electron shell. The opposite charges of cations and anions create attractive forces called ionic bonds, which hold ions together in ionic compounds, such as sodium chloride (Na. Cl)

Covalent Bonds • In a covalent bond, neutral atoms share electrons to fill their outer shells. • Stronger than ionic bonds • The bonds in biological molecules are covalent • Carbon forms 4 covalent bonds, often with other carbon atoms!
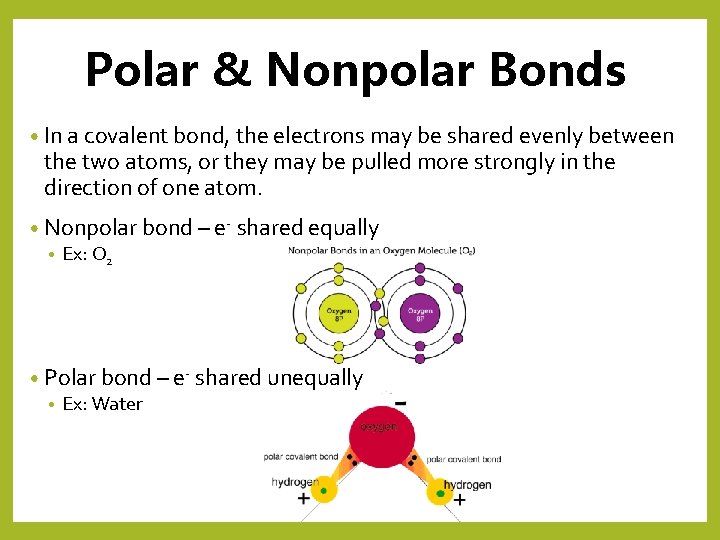
Polar & Nonpolar Bonds • In a covalent bond, the electrons may be shared evenly between the two atoms, or they may be pulled more strongly in the direction of one atom. • Nonpolar bond – e- shared equally • Ex: O 2 • Polar bond – e- shared unequally • Ex: Water

Compounds • Compounds are substances made of more than one element • Ionic compounds and molecular compounds • Forming compounds changes the physical and chemical properties of matter. • Sodium & chlorine are both toxic, but together they form common table salt • Ionic compounds dissociate (come apart) easily in water. • This is how acids and bases are formed. • More H+ ions = acid (p. H < 7), more OH- ions = base (p. H > 7) • Molecular compounds do not dissociate in water.

Chemical Equations • Chemical reactions are written as equations using chemical symbols and formulas. • Must be balanced (# of each type of atom must be the same on both sides)
- Slides: 11