Matter Anything that has a mass and a














- Slides: 18

Matter • Anything that has a mass and a volume

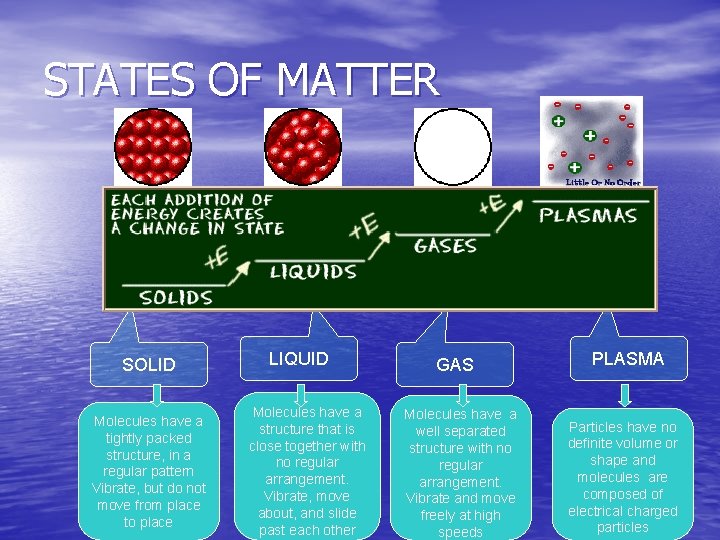
STATES OF MATTER • The Four States of Matter • Solid • Liquid • Gas • Plasma • Four States

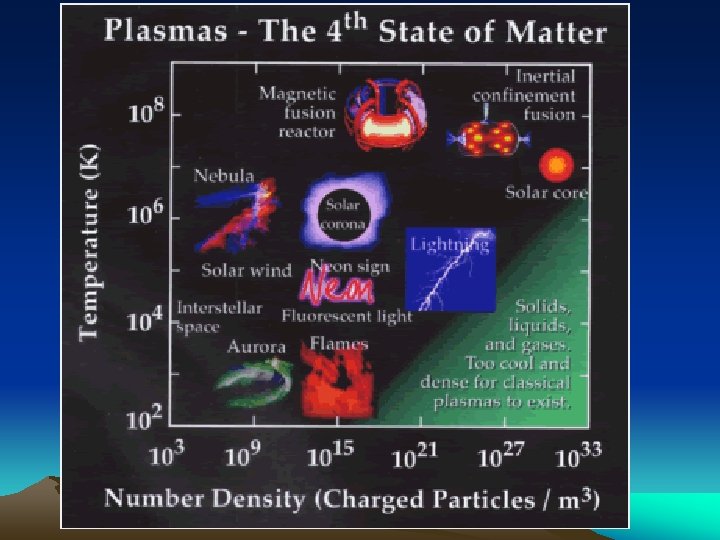
STATES OF MATTER SOLID Molecules have a tightly packed structure, in a regular pattern Vibrate, but do not move from place to place LIQUID Molecules have a structure that is close together with no regular arrangement. Vibrate, move about, and slide past each other GAS Molecules have a well separated structure with no regular arrangement. Vibrate and move freely at high speeds PLASMA Particles have no definite volume or shape and molecules are composed of electrical charged particles

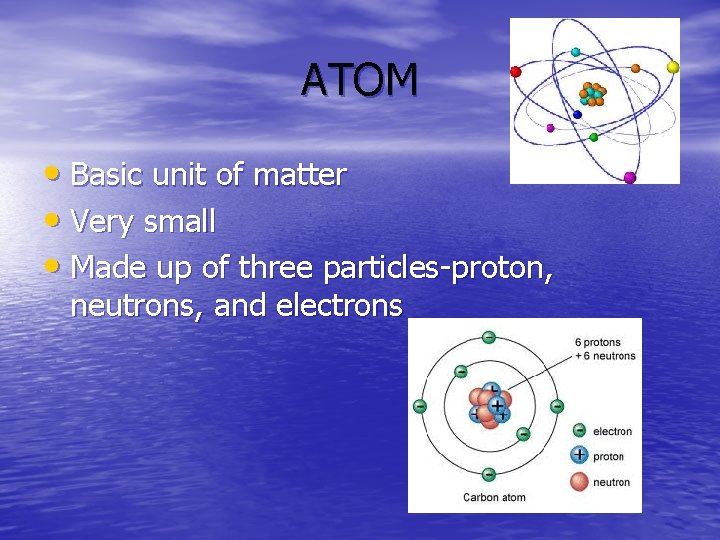
ATOM • Basic unit of matter • Very small • Made up of three particles-proton, neutrons, and electrons

STATES OF MATTER Ø Based upon particle arrangement Ø Based upon energy of particles Ø Based upon distance between particles

Kinetic Theory of Matter It states that all particles are in continual random motion which are molecules or atoms.

ABSOLUTE ZERO • Theoretical temperature at which atoms would not be moving • It is -273. 15 degrees celsius or -459. 68 Fahrenheit

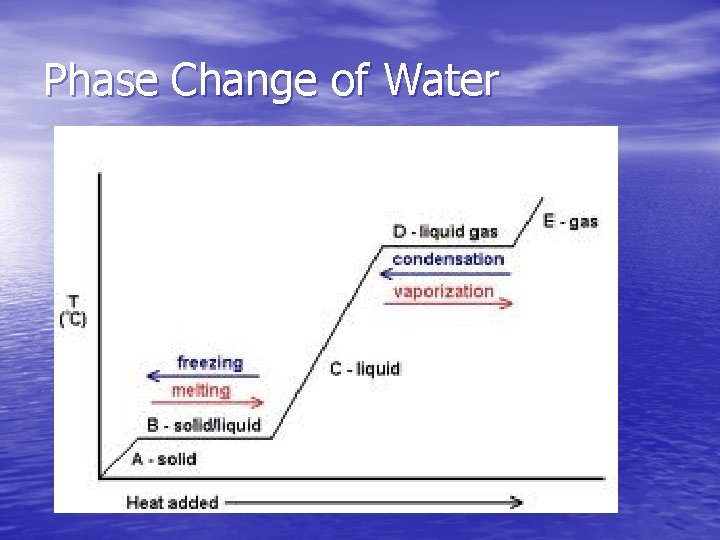
PHASE CHANGES Description of Phase Change Solid to liquid Term for Phase Change Melting Liquid to Freezing solid Heat Movement During Phase Change Heat goes into the solid as it melts. Heat leaves the liquid as it freezes.

PHASE CHANGES Description of Phase Change Term for Phase Change Heat Movement During Phase Change Vaporization, which includes Heat goes into the Liquid to gas boiling and liquid as it vaporizes. evaporation Heat leaves the gas Gas to liquid Condensation as it condenses. Heat goes into the Solid to gas Sublimation solid as it sublimates.

2. Lightning


Phase Changes Occur when we take away or add energy from a substance.

Phase Change of Water

Heat Is a measurement of the energy from all particles that are in a substance (potential and kinetic).

Temperature • Is a number that is related to the motions of the molecules of a substance.

Calorie-a unit of heat • A calorie is the amount of heat energy it takes to raise the temperature of 1 gram of water 1 degree Celsius.

Pressure • The amount of force exerted on the surface of an object

Viscosity