Matter and Particles of Light Quantum Theory Light





- Slides: 11

Matter and Particles of Light: Quantum Theory • Light (energy) and matter in motion behave both as • • • waves and particles Wave-Particle Duality - Quantum Theory Particles of light are called photons: E = hf = hc/l Photons of a specific wavelength l may be absorbed or emitted by atoms in matter Matter is made of different natural elements: lightest Hydrogen (1 proton), heaviest Uranium (92 protons) Smallest particle of an element is atom, made up of a nucleus (protons and neutrons), and orbiting electrons Electrons and protons attract as opposite electrical charges, NOT gravitationally like planets and Sun

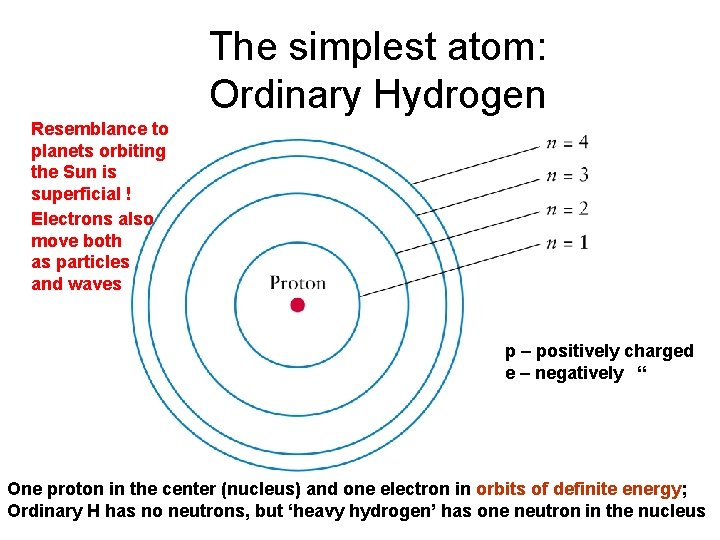
The simplest atom: Ordinary Hydrogen Resemblance to planets orbiting the Sun is superficial ! Electrons also move both as particles and waves p – positively charged e – negatively “ One proton in the center (nucleus) and one electron in orbits of definite energy; Ordinary H has no neutrons, but ‘heavy hydrogen’ has one neutron in the nucleus

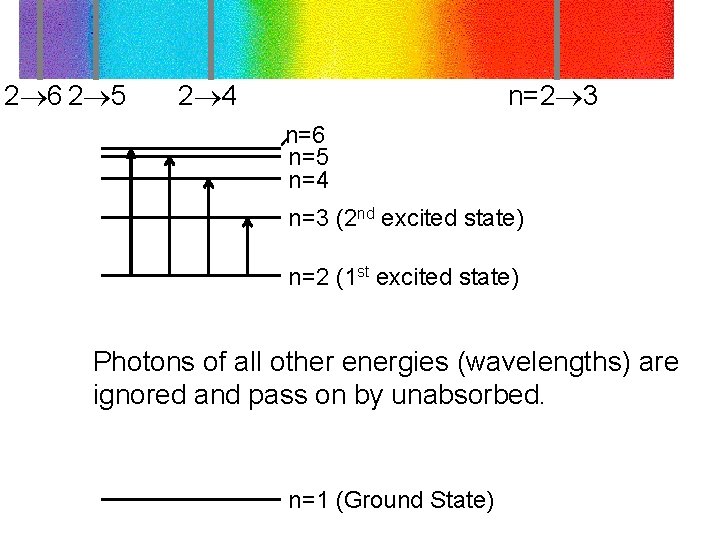
Absorption and emission of photons by H-atom An electron may absorb or emit light photons at specific wavelength Wavelength (n = 3 n = 2): 6562 Angstroms (RED Color) Energy of the photon must be exactly equal to the energy difference between the two ‘orbits’

file: ///E: /Univ 7 e/content/ch 05/0503002. html

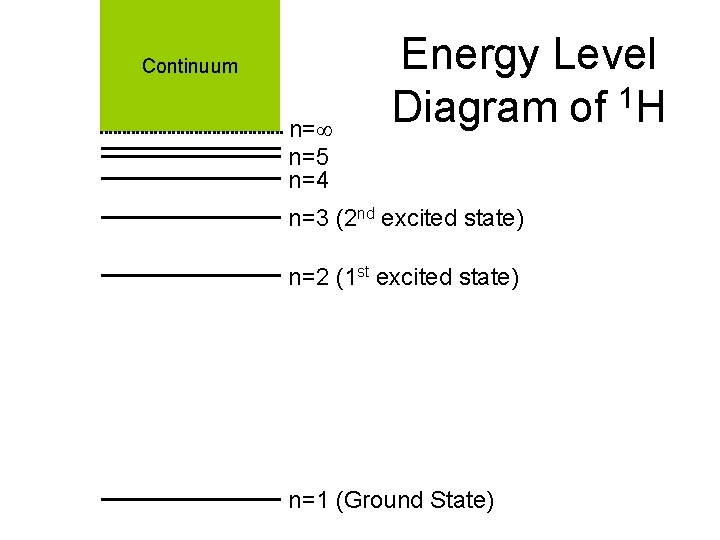
Continuum n= n=5 n=4 Energy Level Diagram of 1 H n=3 (2 nd excited state) n=2 (1 st excited state) n=1 (Ground State)

2 6 2 5 2 4 n=2 3 n=6 n=5 n=4 n=3 (2 nd excited state) n=2 (1 st excited state) Photons of all other energies (wavelengths) are ignored and pass on by unabsorbed. n=1 (Ground State)

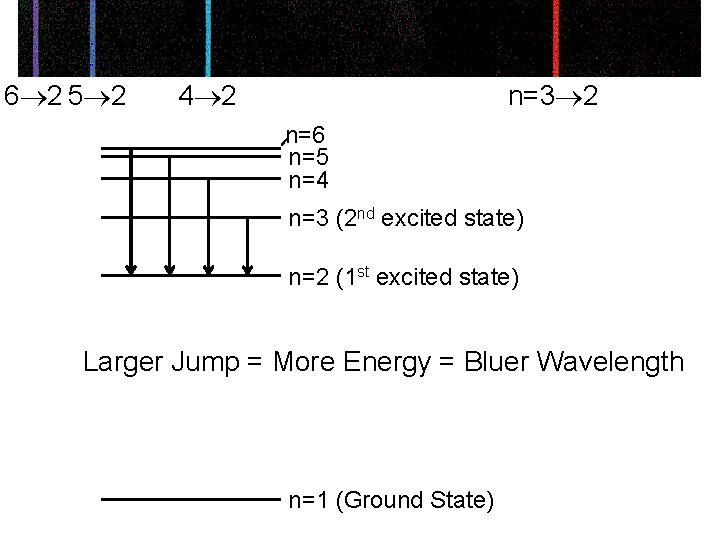
6 2 5 2 4 2 n=3 2 n=6 n=5 n=4 n=3 (2 nd excited state) n=2 (1 st excited state) Larger Jump = More Energy = Bluer Wavelength n=1 (Ground State)

Energy, Frequency, Wavelength • Light particles ‘photons’ have a unique wavelength • The more ‘energetic’ a wave, the higher its frequency, or lower its wavelength • Planck’s Law: Photon energy (‘quantum’) is E=hf=h/l ‘h’ is the Planck’s constant This ‘quantum’ of energy must be equal to the difference in energies between two electron orbits, for either absorption or emission by an atom

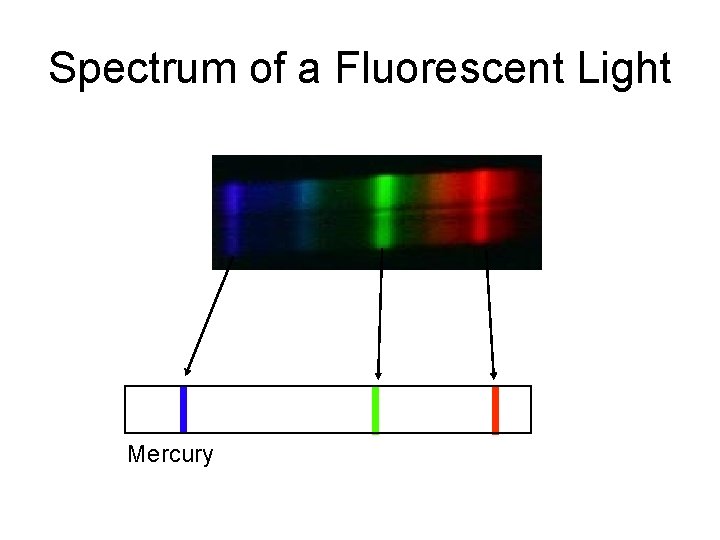
Spectrum of a Fluorescent Light Mercury

Characteristic spectra of elements Each element has a unique set of spectral lines, thus enabling its identification in the source. Observations of spectra of different elements in a source (planet, star, galaxy etc. ) yields its chemical composition

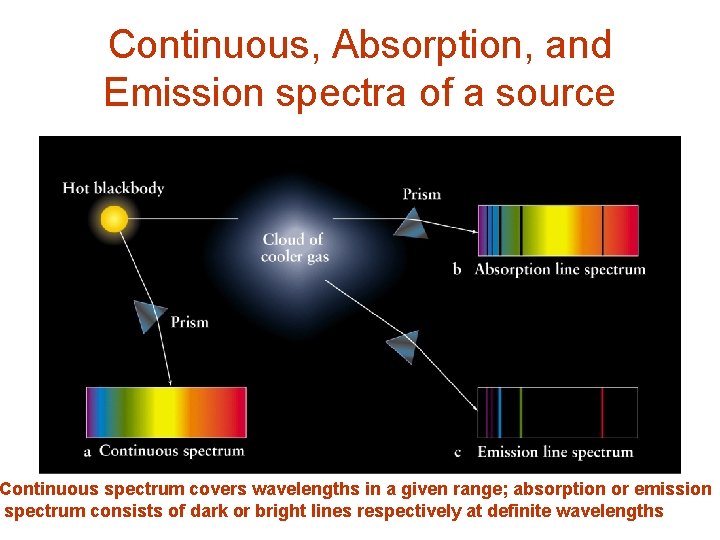
Continuous, Absorption, and Emission spectra of a source Continuous spectrum covers wavelengths in a given range; absorption or emission spectrum consists of dark or bright lines respectively at definite wavelengths