Matter and Energy Matteranything that occupies space and







































- Slides: 63

Matter and Energy • Matter—anything that occupies space and has mass (weight) • Energy—the ability to do work, it can be: • Chemical • Electrical • Mechanical • Radiant

Composition of Matter • Elements—fundamental units of matter • 96 percent of the body is made from four elements • Carbon (C) • Oxygen (O) • Hydrogen (H) • Nitrogen (N) • Atoms—building blocks of elements

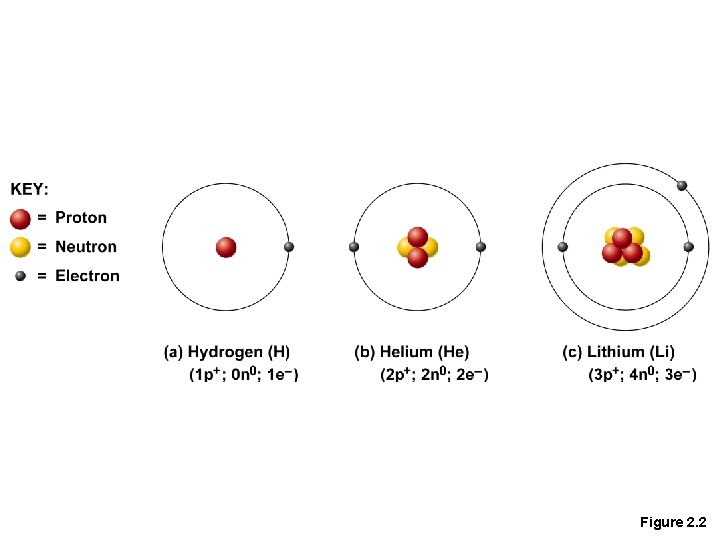
Subatomic Particles • Nucleus, contains: • Protons (p+) • Neutrons (n 0) • Orbiting the nucleus are: • Electrons (e–) • Number of protons equals numbers of electrons in an atom mostly.

Figure 2. 2

Identifying Elements • Atomic number— equal to the number of protons • Atomic mass number—sum of the protons and neutrons

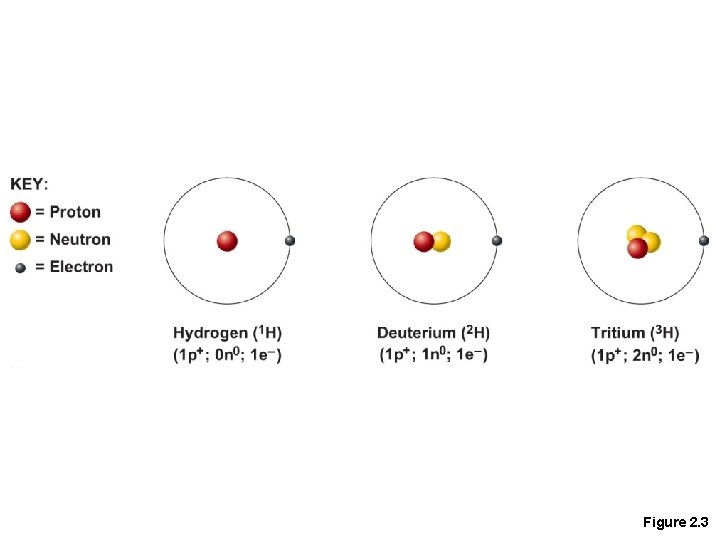
Isotopes and Atomic Weight • Isotopes • Atoms of the same element with the same number of protons and the same atomic number • Vary in number of neutrons

Figure 2. 3

Radioactivity • Radioisotope • Heavy isotope • Tends to be unstable • Decomposes to more stable isotope • Radioactivity—process of spontaneous atomic decay, used in treatment of cancer

Molecules and Compounds • Molecule—two or more atoms of the same elements combined chemically • Compound—two or more atoms of different elements combined chemically

Figure 2. 4

Chemical Reactions • Atoms are united by chemical bonds • Atoms dissociate from other atoms when chemical bonds are broken

Electrons and Bonding • Electrons occupy energy levels called electron shells • Electrons closest to the nucleus are most strongly attracted • Each shell has distinct properties • The number of electrons has an upper limit • Shells closest to the nucleus fill first

Electrons and Bonding • Bonding involves interactions between electrons in the outer shell (valence shell) • Full valence shells do not form bonds

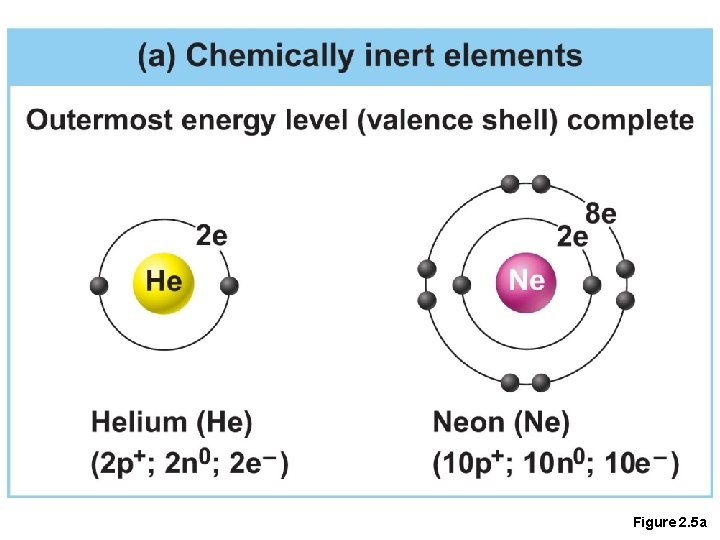
Inert Elements • Atoms are stable (inert) when the outermost shell is complete • How to fill the atom’s shells • Shell 1 can hold a maximum of 2 electrons • Shell 2 can hold a maximum of 8 electrons • Shell 3 can hold a maximum of 18 electrons

Inert Elements • Atoms will gain, lose, or share electrons to complete their outermost orbitals and reach a stable state • Rule of eights • Atoms are considered stable when their outermost orbital has 8 electrons • The exception to this rule of eights is Shell 1, which can only hold 2 electrons

Figure 2. 5 a

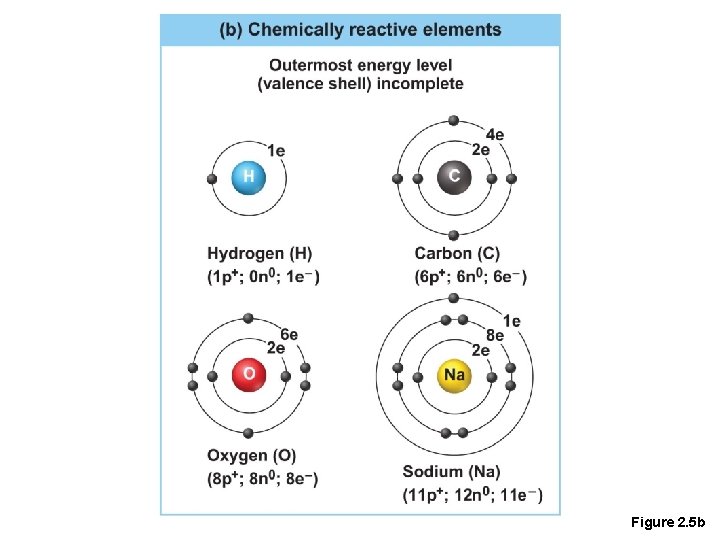
Reactive Elements • Valence shells are not full and are unstable • Tend to gain, lose, or share electrons • Allow for bond formation, which produces stable valence

Figure 2. 5 b

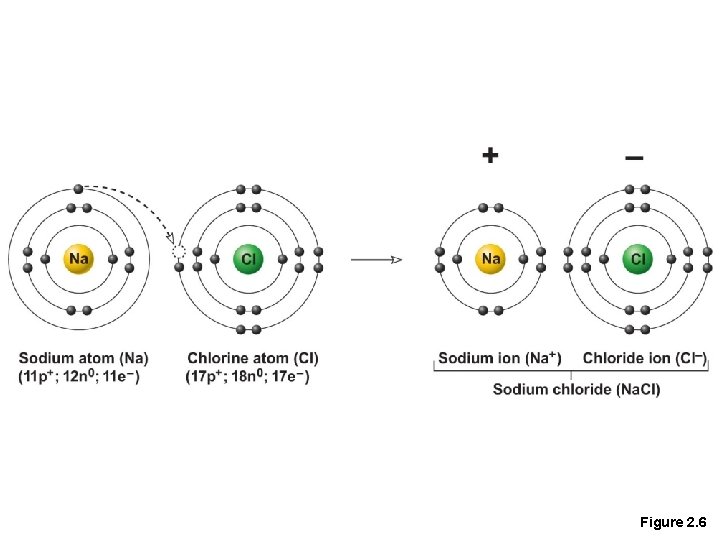
Chemical Bonds • 1. Ionic bonds • Atoms become stable through the transfer of electrons • Form when electrons are completely transferred from one atom to another they are weak bonds as they can easily dissociate in water • Ions • Result from the loss or gain of electrons • Anions are negative due to gain of electron(s) • Cations are positive due to loss of electron(s)1

Figure 2. 6

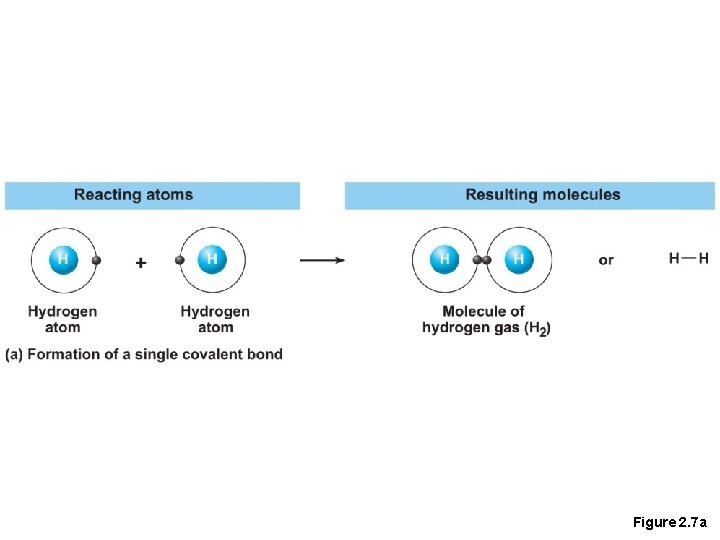
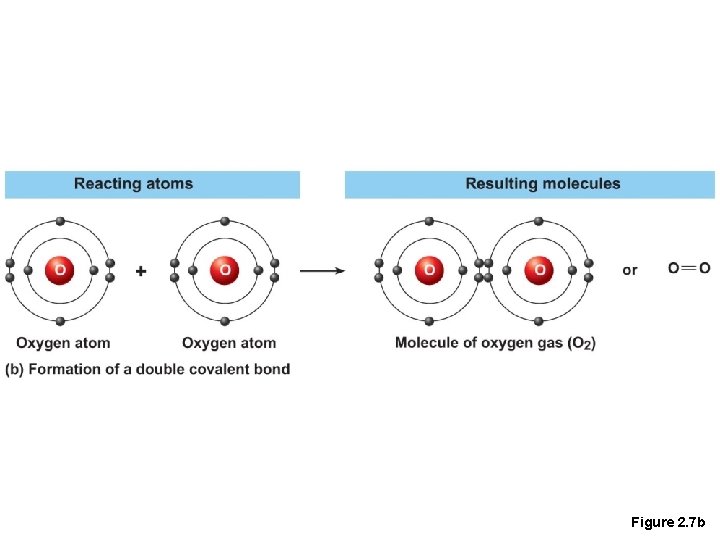
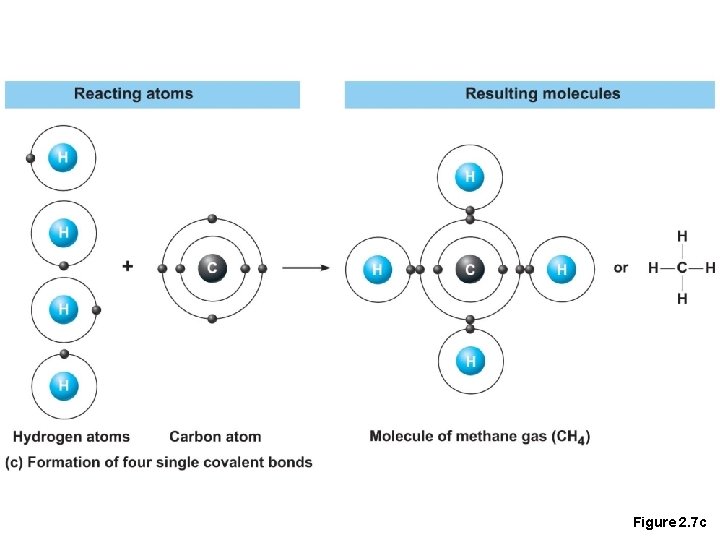
Chemical Bonds • 2. Covalent bonds • Atoms become stable through shared electrons, they are strong bonds as they never dissociate in water

Figure 2. 7 a

Figure 2. 7 b

Figure 2. 7 c

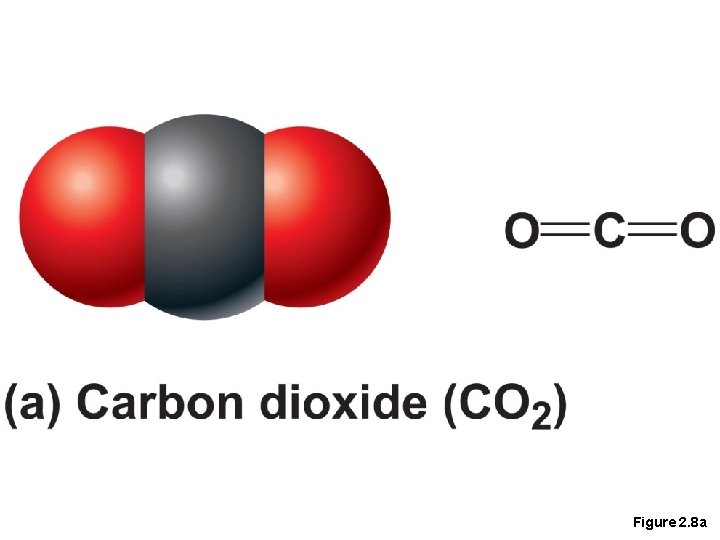
Covalent Bonds • Covalent bonds are either nonpolar or polar • Nonpolar • Electrons are shared equally between the atoms of the molecule • Electrically neutral as a molecule

Figure 2. 8 a

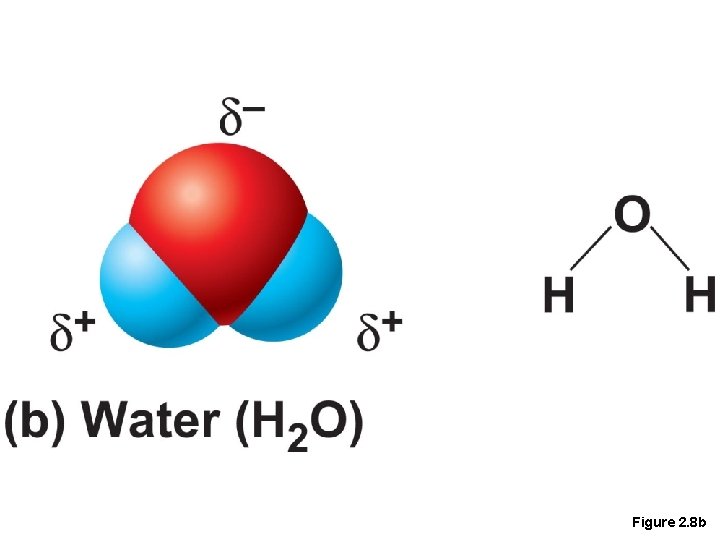
Covalent Bonds • Covalent bonds are either nonpolar or polar • Polar • Electrons are not shared equally between the atoms of the molecule • Have a positive and negative side or pole

Figure 2. 8 b

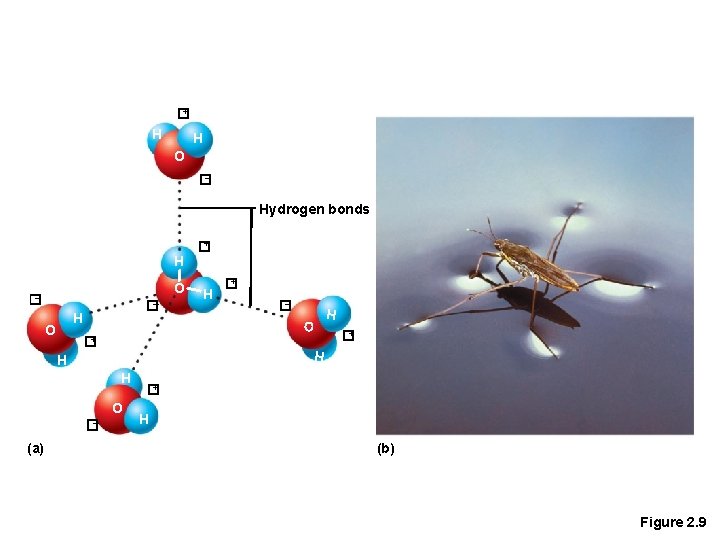
Chemical Bonds • 3. Hydrogen bonds • Weak chemical bonds • Hydrogen is attracted to the negative portion of polar molecule • Provides attraction between molecules


Biochemistry: Essentials for Life • Organic compounds • Contain carbon • Most are covalently bonded • Includes carbohydrates, lipids, proteins, nucleic acids • Inorganic compounds • Lack carbon • Tend to be simpler compounds • Includes water, salts, and some acids and bases

Important Inorganic Compounds • Water –Most abundant inorganic compound

Important Inorganic Compounds • Salts • Easily dissociate into ions in the presence of water • Vital to many body functions

Important Inorganic Compounds • 1. Acids • Release hydrogen ions (H+) • Are proton donors • 2. Bases • Release hydroxyl ions (OH–) • Are proton acceptors • Neutralization reaction • Acids and bases react to form water and a salt

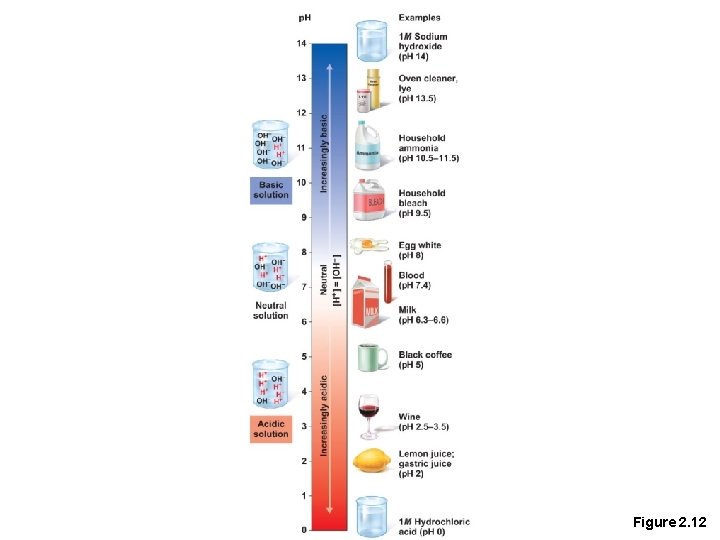
p. H • Measures relative concentration of hydrogen ions • p. H 7 = neutral • p. H below 7 = acidic • p. H above 7 = basic • Buffers—chemicals that can regulate p. H change

Figure 2. 12

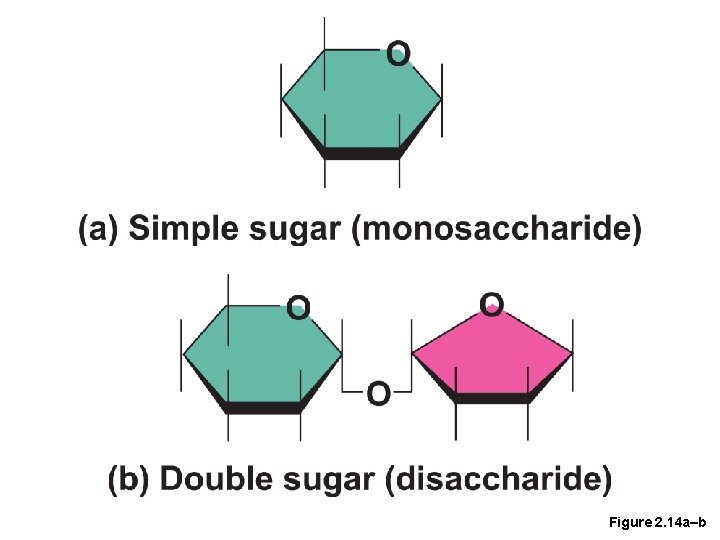
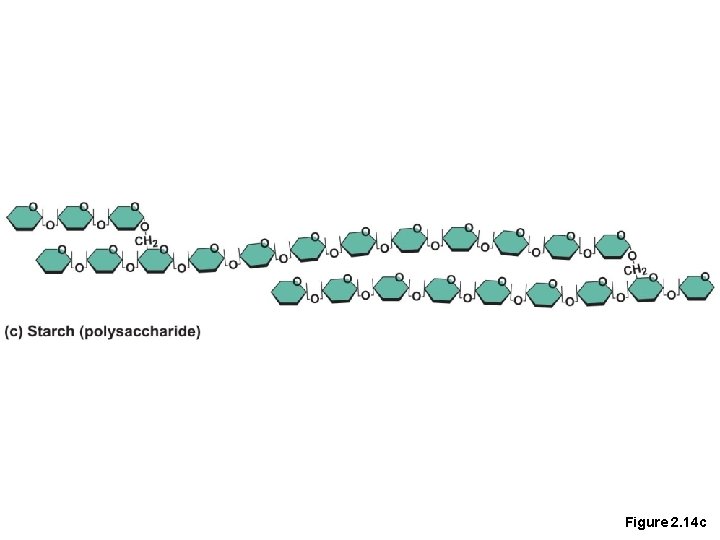
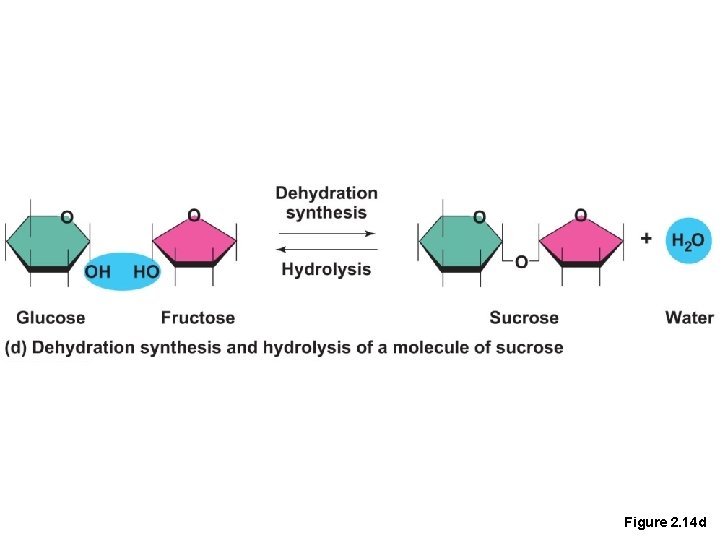
Important Organic Compounds • Carbohydrates • Contain carbon, hydrogen, and oxygen • Include sugars and starches • Classified according to size • Monosaccharides—simple sugars(glucose) • Disaccharides—two simple sugars joined by dehydration synthesis(sucrose) • Polysaccharides—long-branching chains of linked simple sugars(starch)

Figure 2. 14 a–b

Figure 2. 14 c

Figure 2. 14 d

Important Organic Compounds • Lipids • Contain carbon, hydrogen, and oxygen • Carbon and hydrogen outnumber oxygen • Insoluble in water

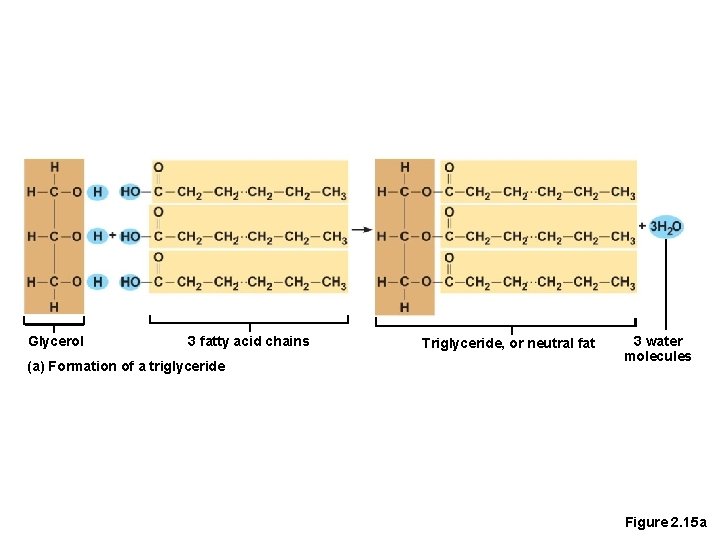
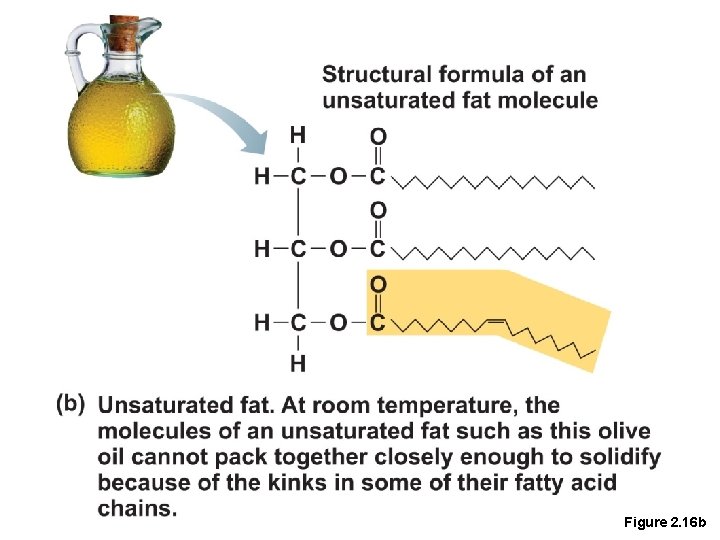
Lipids • Common lipids in the human body • Neutral fats (triglycerides) • Found in fat deposits • Source of stored energy • Composed of three fatty acids and one glycerol molecule • Saturated fatty acids contain only single covalent bonds(as butter, solid at room temp. , dangerous) • Unsaturated fatty acids contain one or more double covalent bonds(olive oil, liquid, safe)

Glycerol 3 fatty acid chains (a) Formation of a triglyceride Triglyceride, or neutral fat 3 water molecules Figure 2. 15 a

Figure 2. 16 a

Figure 2. 16 b

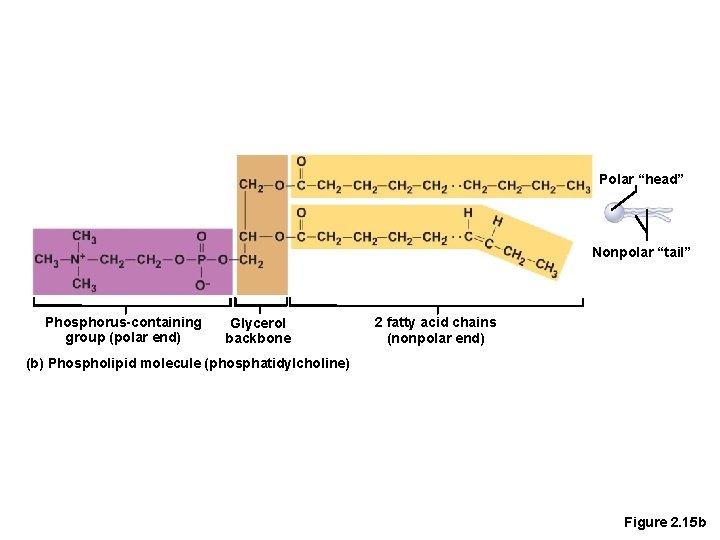
Lipids • Common lipids in the human body (continued) • Phospholipids • Contain two fatty acids rather than three • Form cell membranes

Polar “head” Nonpolar “tail” Phosphorus-containing group (polar end) Glycerol backbone 2 fatty acid chains (nonpolar end) (b) Phospholipid molecule (phosphatidylcholine) Figure 2. 15 b

Lipids • Common lipids in the human body (continued) • Steroids • Include cholesterol, bile salts, vitamin D, and some hormones • Cholesterol is the basis for all steroids made in the body

Important Organic Compounds • Proteins • Account for over half of the body’s organic matter • Provide for construction materials for body tissues • Play a vital role in cell function • Act as enzymes, hormones, and antibodies • Contain carbon, oxygen, hydrogen, nitrogen, and sometimes sulfur • Built from amino acids

Proteins • Amino acid structure • Contain an amine group (NH 2) • Contain an acid group (COOH) • Vary only by R groups

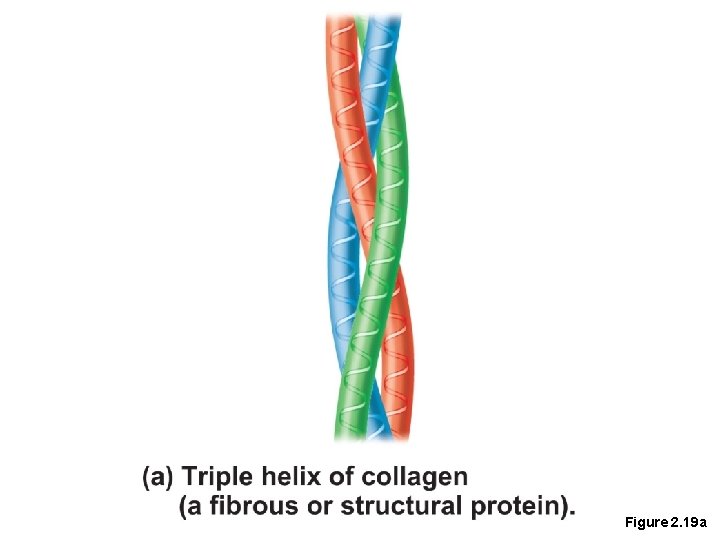
Proteins • Fibrous proteins • Also known as structural proteins • Appear in body structures • Examples include collagen and keratin • Stable

Figure 2. 19 a

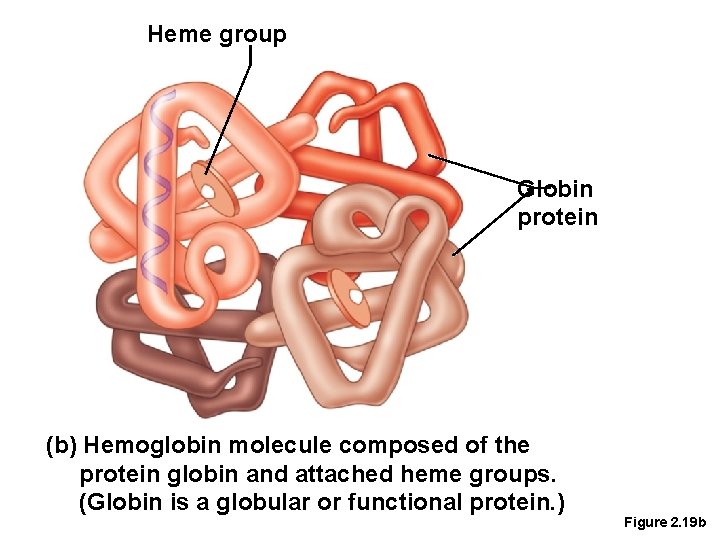
Proteins • Globular proteins • Also known as functional proteins • Function as antibodies or enzymes • Can be denatured

Heme group Globin protein (b) Hemoglobin molecule composed of the protein globin and attached heme groups. (Globin is a globular or functional protein. ) Figure 2. 19 b

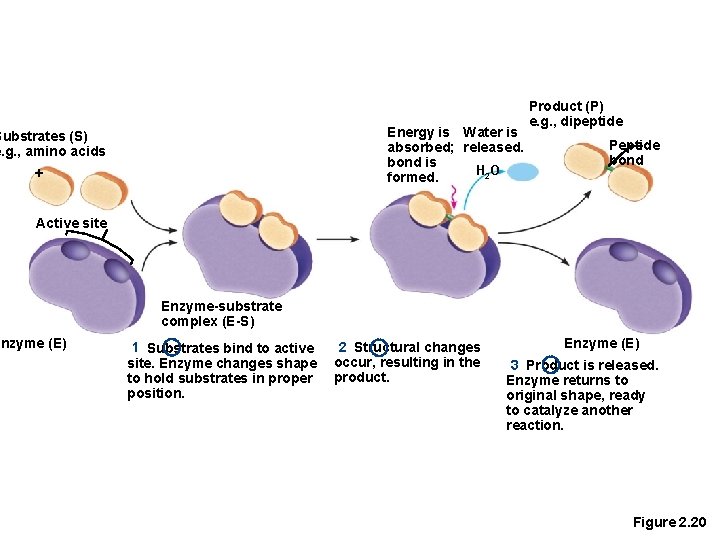
Enzymes • Act as biological catalysts • Increase the rate of chemical reactions • Bind to substrates at an active site

Energy is Water is absorbed; released. bond is H 2 O formed. Substrates (S) e. g. , amino acids + Product (P) e. g. , dipeptide Peptide bond Active site Enzyme (E) Enzyme-substrate complex (E-S) 1 Substrates bind to active site. Enzyme changes shape to hold substrates in proper position. 2 Structural changes occur, resulting in the product. Enzyme (E) 3 Product is released. Enzyme returns to original shape, ready to catalyze another reaction. Figure 2. 20

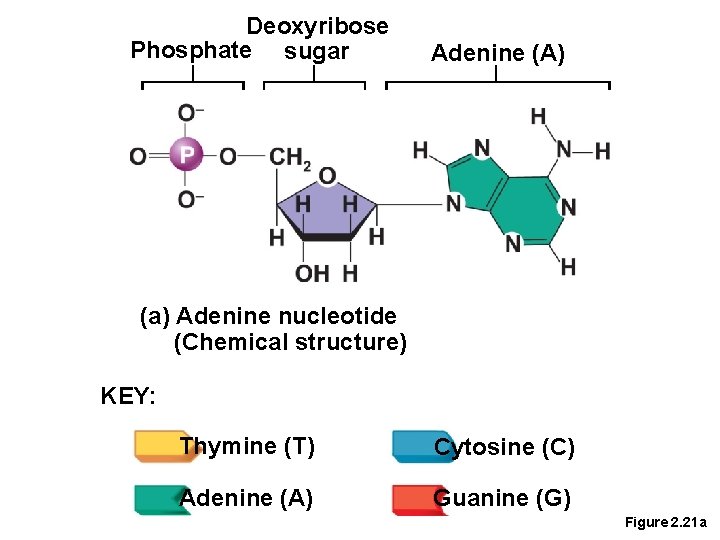
Important Organic Compounds • Nucleic Acids • Built from nucleotides • Pentose or ribose (5 carbon) sugar • A phosphate group • A nitrogenous base • A = Adenine • G = Guanine • C = Cytosine • T = Thymine • U = Uracil.

Deoxyribose Phosphate sugar Adenine (A) (a) Adenine nucleotide (Chemical structure) KEY: Thymine (T) Cytosine (C) Adenine (A) Guanine (G) Figure 2. 21 a

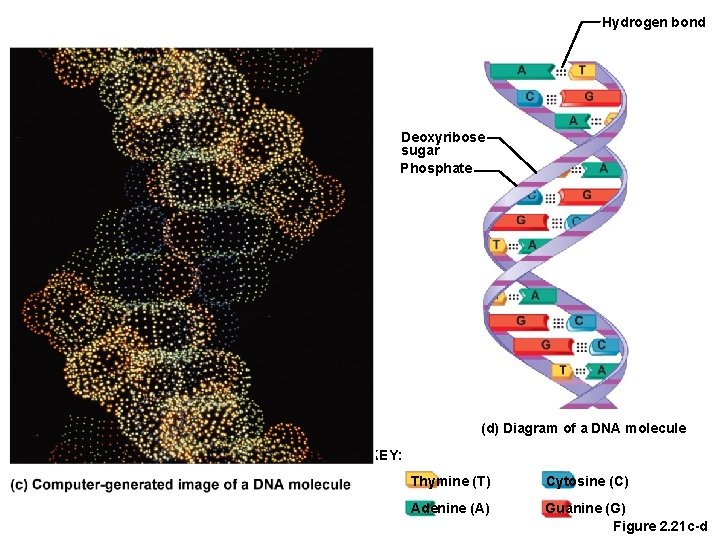
Nucleic Acids • Deoxyribonucleic acid (DNA) • The genetic material found within the cell’s nucleus • Provides instructions for every protein in the body • Organized by complimentary bases to form a double-stranded helix • Contains the sugar deoxyribose and the bases adenine, thymine, cytosine, and guanine • Replicates before cell division

Hydrogen bond Deoxyribose sugar Phosphate (d) Diagram of a DNA molecule KEY: Thymine (T) Cytosine (C) Adenine (A) Guanine (G) Figure 2. 21 c-d

Nucleic Acids • Ribonucleic acid (RNA) • Carries out DNA’s instructions for protein synthesis • Created from a template of DNA • Organized by complimentary bases to form a single-stranded helix • Contains the sugar ribose and the bases adenine, uracil, cytosine, and guanine • Three varieties are messenger, transfer, and ribosomal RNA

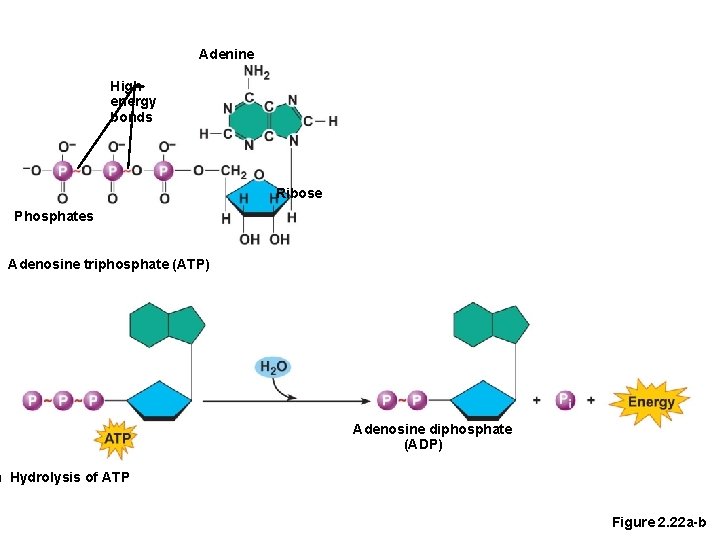
Important Organic Compounds • Adenosine triphosphate (ATP) • Composed of a nucleotide built from ribose sugar, adenine base, and three phosphate groups • Chemical energy used by all cells • Energy is released by breaking high energy phosphate bond • ATP is replenished by oxidation of food fuels

Adenine High energy bonds Ribose Phosphates ) Adenosine triphosphate (ATP) Adenosine diphosphate (ADP) ) Hydrolysis of ATP Figure 2. 22 a-b